- LOGIN
- MemberShip
- 2026-04-24 02:52:26
- Company
- Oral CSU drug ‘Rhapsido’ nears approval in KOR
- by Eo, Yun-Ho Apr 02, 2026 08:46am
- The oral urticaria drug ‘Rhapsido’ is nearing commercialization in Korea.According to industry sources, Novartis Korea’s oral BTK inhibitor Rhapsido (remibrutinib) is expected to receive marketing authorization from the Ministry of Food and Drug Safety next month (May).Rhapsido is an oral targeted therapy that inhibits Bruton’s tyrosine kinase (BTK), a key pathway in the pathophysiology of CSU, thereby blocking the release of histamine and inflammatory mediators.The drug was approved in the United States last September for the treatment of adult CSU patients whose symptoms persist despite second-generation H1 antihistamines.CSU is a disease characterized by severe symptoms and unpredictable exacerbations, making diagnosis and management difficult. It is known to arise from immune dysregulation. In CSU patients, the immune system can be activated via allergic (IgE) or autoimmune (IgG) pathways.This leads to specific immune cells activating the BTK protein. Once activated, BTK triggers the release of histamine and other pro-inflammatory mediators, causing red, swollen, and itchy hives.The most notable feature of Rhapsido is that it is an oral drug (taken twice daily). Until now, treatment options for patients unresponsive to first-line antihistamines have been largely limited to the injectable biologic Xolair (omalizumab). The arrival of Rapsido opens a new option, an oral targeted therapy.The drug demonstrated efficacy in the Phase III REMIX-1 and REMIX-2 studies. Results showed Rhapsido demonstrated superiority over placebo in improving itch severity (ISS7), hive severity (HSS7), and total urticaria activity score (UAS7) starting from Week 2. Approximately one-third of patients achieved complete remission (defined as zero itch and zero hives) by Week 12.Beyond CSU, Novartis is also expanding clinical development of Rhapsido across a range of immune-mediated diseases, including chronic inducible urticaria (CIndU), hidradenitis suppurativa (HS), food allergy, and multiple sclerosis.
- Company
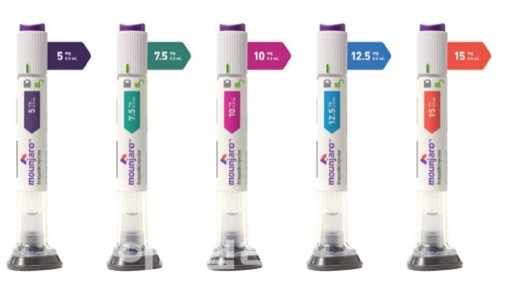
- Lilly Korea sales, 194%↑ from a year earlier…'Mounjaro' effect
- by Son, Hyung Min Apr 02, 2026 08:46am
- Eli Lilly Korea's sales structure in South Korea is undergoing a rapid, substantial changes. Following the launch of the GLP-1 class blockbuster 'Mounjaro,' the company’s financial performance has surged, completely restructuring its growth model, which previously centered on its core products.According to the Financial Supervisory Service, Eli Lilly Korea’s sales last year reached KRW 482.1 billion, a 193.6% increase compared to the previous year. During the same period, operating profit rose by 259.2%, jumping from KRW 10.3 billion in 2024 to KRW 37.1 billion last year. Eli Lilly Korea's sales performance by year (unit: KRW 100 million)Previously, the company's sales relied on oncology drugs such as ‘Verzenio (abemaciclib)’ and ‘Cyramza (ramucirumab),’ as well as the SGLT-2 inhibitor ‘Jardiance (empagliflozin)’ and the biologic ‘Taltz (ixekizumab).’ Due to a lack of new blockbuster entries, sales had stalled below KRW 200 billion from 2021 to 2024.However, this structure changed abruptly with the emergence of ‘Mounjaro (tirzepatide).’ Mounjaro secured outstanding growth momentum by rapidly expanding beyond diabetes into the obesity treatment market.Mounjaro acts on both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. Through this dual action, it stimulates insulin secretion, improves insulin resistance, and decreases glucagon secretion, thereby lowering both fasting and postprandial blood glucose levels.In patients with diabetes and obesity, the "incretin effect" is typically diminished, primarily due to reduced GLP-1 secretion and impaired GIP action. As GLP-1 and GIP are key hormones responsible for approximately two-thirds of the postprandial insulin response, the dual-axis mechanism of Mounjaro stands out as a significant clinical advantage.Mounjaro's first indication was secured in June 2023 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. In August 2024, the indication was expanded for chronic weight management. By expanding its scope to include patients with obesity or overweight patients with weight-related comorbidities, Mounjaro successfully became positioned as an obesity treatment.Released in the domestic market last August, Mounjaro quickly secured position in the market. According to market research firm IQVIA, Mounjaro recorded KRW 28.4 billion in sales in Q3 of last year, which then surged to KRW 187.1 billion in Q4, easily surpassing KRW 100 billion in quarterly sales for a single item. During the same period, it outpaced its competitor 'Wegovy (semaglutide),' rapidly increasing its market dominance.A similar trend is observed in the global market. As of Q2 of last year, Mounjaro's global sales exceeded those of Wegovy, marking a significant turning point in the battle for leadership in the obesity treatment market.Active Development of Multi-Mechanism GLP-1 SuccessorsLilly is also continuously strengthening its GLP-1-based portfolio. Currently, the oral GLP-1 agonist 'orforglipron' is undergoing regulatory approval processes in more than 40 countries, with a New Drug Application (NDA) for type 2 diabetes expected in the U.S. by the end of this year.Unlike Mounjaro, orforglipron is a single GLP-1 mechanism but distinguishes itself as an oral medication. Notably, it does not require fasting after administration and, as a small-molecule-based drug, has lower production costs, suggesting high market scalability. In clinical trials, it has shown superior results compared to competitors in both HbA1c reduction and weight loss.Lilly's diabetes and obesity treatment 'Mounjaro'Next-generation pipelines are also under development. For instance, 'retatrutide,' a triple agonist that simultaneously targets GLP-1, GIP, and glucagon (GCG). Currently, no triple-action obesity drug has been approved, and retatrutide, which is in Phase 3 clinical trials, is considered the closest to commercialization.According to recently released Phase 3 study results, retatrutide met primary endpoints by demonstrating significant improvements in HbA1c and weight loss compared with placebo. Lilly is considering strategies to expand retatrutide's indications beyond obesity to various chronic diseases, including diabetes and liver disease.Additionally, 'eloralintide,' which targets both GLP-1 and amylin receptors, has entered global Phase 3 trials. This drug mimics the action of the amylin hormone secreted by the pancreas to act directly on the brain, thereby increasing satiety and suppressing food intake.
- Company
- Global pharmas cautiously welcome pricing reform
- by Son, Hyung Min Apr 01, 2026 08:15am
- “Domestic firms are crying, multinational firms are smiling.” This is the prevailing assessment of Korea’s latest drug pricing reform.With the reform centered on lowering prices of generic drugs now finalized, Korea’s drug pricing structure is approaching a turning point. While the government has proposed reallocating savings toward rewarding innovative drugs and improving patient access, the industry is also voicing cautious views that outcomes will depend on the system’s effectiveness and execution.According to industry sources, the Ministry of Health and Welfare finalized the drug pricing reform plan on the 26th through the Health Insurance Policy Deliberation Committee (HIPDC). The reform focuses on lowering the pricing benchmark for generic and off-patent drugs from the current 53.55% to around 45%.This reform is regarded as significant not simply as a price cut, but as a structural reorganization aimed at strengthening incentives for innovative drugs using the savings generated.The key lies in resource reallocation. The strategy is to use the funds secured by adjusting the generic-centric pricing structure to lower barriers to reimbursement for new drugs through measures such as ▲faster reimbursement listing of treatments for rare and severe diseases, ▲introduction of flexible drug pricing contracts, and ▲raising the ICER (incremental cost-effectiveness ratio) threshold.This is interpreted as a response to long-standing criticism that Korea’s pricing system has focused excessively on cost containment, limiting access to innovative therapies.The Ministry of Health and Welfare also defined the reform as a structural transformation of the pricing system.The Ministry stated, “By advancing the drug pricing system to the level of major countries, we can enhance public access to treatment and coverage while r reducing drug expenditure burdens. Establishing a compensation system for research and development and efforts to ensure the stable supply of essential medicines will serve as a momentum for the pharmaceutical and biotech industries to take the leap forward.”Global pharmas express “cautious optimism”…System design is keyGlobal pharmaceutical companies are generally welcoming the reform. Given that patient access and reimbursement rates for new drugs in Korea have lagged behind major countries, there is an expectation that savings from generic price cuts could improve access to innovative therapies.The Korea Research-based Pharmaceutical Industry Association (KRPIA), which is primarily composed of multinational pharmaceutical companies, also offered a positive assessment.The association stated, “he policy reflects a commitment to reflecting the value of innovative new drugs and enhancing patient access. It is crucial whether the reform’s intent is actually realized through system design and implementation,” thereby emphasizing the importance of policy implementation.This expectation is also supported by data. According to PhRMA’s ‘2023 Global Access to New Medicines Report,’ among the 460 new drugs covered by health insurance worldwide from 2012 to 2021, South Korea’s coverage rate was 22%, falling below the G20 (28%) and OECD (29%) averages.For innovative cancer drugs, the rate was 23%, and for rare disease treatments, it was just 12%, both significantly lower than the G20 and OECD averages, respectively.However, global pharmaceutical companies are also expressing conditional caution. While the entry environment may improve, there are concerns that requirements for demonstrating value during reimbursement listing have become more stringent.An official from a global pharmaceutical company noted, “It is positive in terms of improving access to new drugs and strengthening clinical value-based evaluation. However, the strengthened post-listing price control raises concerns about predictability.”Another official from a global pharmaceutical company agreed with the direction but raised questions about its implementation.The official said, “The changes in government perception toward rare and severe diseases are significant. Even drugs under the pilot approval-evaluation-negotiation linkage program are facing reimbursement delays, so concrete execution plans are urgently needed.”Regarding the increase in the ICER threshold, the official emphasized, “The magnitude of the increase is more important than the direction itself. If implementation is delayed under the pretext of policy research, it will just become another waiting period for patients.”Institutional issues surrounding global pharmaceutical companies also remain unresolved. Industry feedback regarding the revision of certification criteria for innovative pharmaceutical companies was, “only the name has changed, with limited substantive improvement.”Apart from the fact that bonus points are awarded for certain factors such as attracting foreign capital, joint research, and open innovation, the assessment is that the industry’s long-standing demand for the inclusion of achievements in attracting headquarter-level R&D investment has not been sufficiently reflected. Furthermore, the fact that indicators, such as the scale of pharmaceutical exports, which are difficult for multinational companies to meet, remain unchanged, poses disadvantages.While some companies are already expanding cooperation with the government and joint research with domestic firms with certification in mind, the extent to which these efforts will be reflected in evaluations remains uncertain.An industry official stated, “To foster the domestic biotech ecosystem, collaboration with global pharma is essential. Since the role of domestic branches is crucial in attracting headquarter-level R&D investment, these characteristics need to be reflected in the design of the system.”Another industry official noted, “The new system is already affecting future pipeline processes. Multinational companies have been strengthening preparations to obtain the Innovative Pharmaceutical Company certification,” suggesting that this reform is bringing about changes in actual business strategies in practice.
- Policy
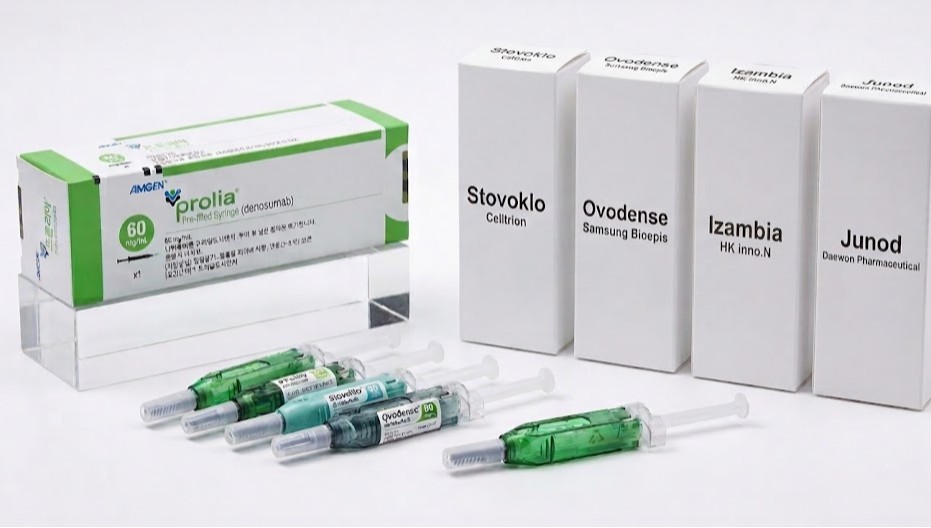
- Daewon Pharm joins the Prolia biosimilar competition
- by Lee, Tak-Sun Apr 01, 2026 08:15am
- AI-generated image (Prolia, Stoboclo, Obodence, Izambia, Junod)A series of biosimilars with the same active ingredient as Amgen’s osteoporosis drug Prolia are being approved. With Daewon Pharm joining the race, 5 products have now received marketing authorization in Korea.Prolia is an osteoporosis treatment administered as a subcutaneous injection once every 6 months, offering strong bone density improvement and fracture prevention effects.In Korea, it has dominated the market with annual sales of approximately KRW 180 billion through copromotion with Chong Kun Dang.On the 31st, the Ministry of Food and Drug Safety approved Daewon Pharma’s Prolia biosimilar, ‘Junod Prefilled Syringe.’This product is indicated for: ▲ the treatment of postmenopausal women with osteoporosis, ▲ the treatment of men with osteoporosis to increase bone density, ▲ the treatment of glucocorticoid-induced osteoporosis, ▲ the treatment of bone loss in patients with non-metastatic prostate cancer receiving androgen deprivation therapy, and ▲ the treatment of bone loss in women with breast cancer receiving adjuvant aromatase inhibitor therapy.It is administered via subcutaneous injection 6 six months in the upper arm, upper thigh, or abdomen, identical to Amgen’s Prolia. The product demonstrated equivalence to Prolia through Phase I and Phase III trials.Juno is a biosimilar developed by Gedeon Richter, headquartered in Budapest, Hungary.Gedeon Richter is a multinational pharmaceutical company with a global presence.In addition to Junod, Daewon Pharm has also introduced the Forteo biosimilar ‘Terrosa Inj’ through an agreement with Gedeon Richter. Terrosa was developed by Richter-Helm Biotec, a joint venture between Gedeon Richter and the German company Helm.With Daewon Pharm obtaining approval for Junod, there are now 5 Prolia biosimilars available in the Korean market.Celltrion’s ‘Stoboclo Prefilled Syringe’ was the first biosimilar approved in November 2024, followed by Samsung Bioepis’ ‘Obodence Prefilled Syringe,’ Meditip’s ‘Meditip Denosumab Prefilled Syringe,’ and HK inno.N’s ‘Izambia Prefilled Syringe,’ all of which have obtained marketing approval.Meditip Denosumab Prefilled Syringe was approved in Korea through a regulatory affairs agency, and Novartis has been identified as the contract manufacturer. There is a possibility that Novartis will handle domestic sales in the future.HK inno.N’s Izambia was developed by Spain-based mAbxience. HK inno.N has also signed a contract with mAbxience to introduce a nivolumab (brand name: Opdivo) biosimilar in addition to this denosumab biosimilar.Meanwhile, Celltrion’s Stoboclo is being co-marketed by Daewoong Pharmaceutical, and Samba’s Obodence is being co-marketed by Hanmi Pharmaceutical.The denosumab biosimilar market is rapidly becoming a battleground among major domestic pharmaceutical companies.Currently, Celltrion’s Stoboclo and Samsung Bioepis’ Obodence are listed for reimbursement. Full-scale competition is expected once the remaining approved products are also listed. All patents for Prolia listed with the MFDS expired as of March 17 last year.
- InterView
- [Reporter's View] Let's focus on building 'solid pharmas'
- by Lee, Jeong-Hwan Apr 01, 2026 08:15am
- Why does the conflict between the Ministry of Health and Welfare (MOHW) and the pharmaceutical industry over drug pricing reforms repeat the same patterns? The MOHW finalized a reform plan at the Health Insurance Policy Review Committee, held in March, focusing on reducing generic drug prices while providing incentives for innovative pharmaceutical companies and those contributing to the supply of unstable essential medicines. However, the differences in position between the two parties do not seem to be clearly resolved.The MOHW promised to exchange information and gather opinions to design a reform plan that could also be favorable to the pharmaceutical industry, addressing the National Assembly, the media, and the industry. However, the prevailing assessment is that these efforts remained insufficient and incomplete.Of course, it is impossible to devise an administration or policy that satisfies everyone 100%. The MOHW's stance during deliberations on this drug-pricing reform was unsatisfactory.The MOHW's challenge is understandable. Lee Hyung-hoon, the 2nd Vice Minister of Health and Welfare and Chairman of the Health Insurance Policy Review Committee, expressed the hardship during the process of establishing the reform plan. Lee noted that it is not easy to achieve both the conflicting tasks of ensuring health insurance efficiency and fostering the pharmaceutical industry. Lee's sincere statement received positive responses from reporters, offering a chance to experience and empathize with the difficulties of operating executive branch policy.Despite this, the MOHW must seek better ways to engage the pharmaceutical industry when devising future drug pricing reforms. This is to minimize the aftereffects of exhaustive clashes between the government and industry, while improving the likelihood of a soft policy landing.Health economists point out that the MOHW has not made sufficient effort to design a drug-pricing system that balances equity between the domestic and multinational, foreign-capital pharmaceutical industries.Furthermore, they note that it is difficult to discern the MOHW's philosophy and specific direction for managing National Health Insurance finances. They stated that the administrative goals are so blunt and ambiguous that it is difficult to predict them. The criticism is that if the MOHW were to reveal its inner thoughts transparently, proper consultation would be possible.The MOHW emphasized the "structural improvement toward innovative new drugs" as the justification for this drug pricing reform and as a mechanism to appeal that it is inevitable to cut the prices of already-listed generics.While the direction might not be wrong, the slope was too steep. South Korea's pharmaceutical industry is still centered on generics. The quality and self-sufficiency rate of generics are high. In reality, hundreds of items are licensed, prescribed, and distributed for a single active ingredient.On the other hand, only 41 domestically developed new drugs have obtained marketing authorization. The first domestically developed new drug was Sunpla (SK Pharma), authorized in July 1999, and the most recent domestic new drug is Xcopri (Dong-A ST), which received its license in November 2025, making it the 41st. Among these, no new drug has achieved blockbuster-level sales.To demand that the domestic pharmaceutical industry, which has birthed 41 new drugs over 25 years and is still evaluated as being at a "high school level" or a "twenty-year-old" who has just removed the label of a minor, instantly stop manufacturing and selling generics to produce blockbuster results is an excessively harsh demand.The reform plan should have reflected more of the industry's concerns about how much new drug Research and Development (R&D) will be revitalized by this drug pricing reform, how negative it will be for the supply of price-stabilized medicines, and the potential causal link to employment stability.In MOHW data on the direction of drug pricing reform, one word stood out: "solid pharmaceutical companies."The MOHW expressed its ambition to use this reform as a turning point to cultivate and support robust pharmaceutical companies that are sincere about new drug development and the stable supply of essential medicines, rather than being buried only in generic sales competition.To achieve this ambition, the MOHW must set transparent, clear standards for what constitutes a "solid pharmaceutical company" and develop evaluation indicators. A process involving a public-private consultative body is necessary. This means that active administration and two-way communication are absolutely required to align the "zero point" regarding "robustness" with pharmaceutical companies.During the process of establishing drug pricing reform plan, many "solid" pharmaceutical companies that have secured R&D ratios criticized the government, saying, "It is doubtful whether the MOHW or the MFDS has the standards to distinguish innovative pharmaceutical companies that are sincere about industrial development and public health, or if they even have the evaluation data."The point was that government administration capable of distinguishing "paper companies" that rely on consigned generic production as their core revenue source from "real" pharmaceutical companies has not been properly carried out until now.In a situation where there is zero trust in whether the government has the criteria to distinguish the wheat from the chaff or the administrative power to do so, the MOHW suddenly pushed a reform plan to cut generic prices by up to 40% while favoring companies that have proven innovation. The industry voiced that they were told to just "sign it," which led to frustration and even fear.When promoting the reform plan, the MOHW must conduct follow-up oversight to develop a comprehensive report card that clearly distinguishes robust pharmaceutical companies and provides tailored support.The MOHW now faces the difficult task of communicating closely with the pharmaceutical industry to find and resolve the blind spots in government administration regarding the resolution of the multi-item generic structure, while recognizing that high-quality generics are the heart of K-Pharmaceuticals and the "two legs" that will support the future of industry and employment.We anticipate MOHW administration that identifies field-level clues to fundamentally shift the policy paradigm away from simply cutting generic prices. This would be catching two birds with one stone: health insurance cost savings and new drug development.
- Company
- NMOSD drug ‘Uplizna’ fails reimbursement again in KOR
- by Eo, Yun-Ho Apr 01, 2026 08:15am
- Uplizna, a new drug for neuromyelitis optica spectrum disorder (NMOSD), has failed to secure reimbursement listing in its second attempt.According to Dailypharm coverage, Mitsubishi Tanabe Pharma Korea’s pricing negotiations with the National Health Insurance Service (NHIS) for Uplizna (inebilizumab), a treatment used to treat adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are positive for anti-Aquaporin-4 (AQP4) antibodies, have ultimately collapsed.Although both sides made efforts to continue discussions by extending the negotiation period, it is understood that disagreements arose over adjustments to the expenditure cap.Consequently, it is expected that Uplizna, which had been gaining expectation as a new treatment option for NMOSD, will face significant challenges in establishing a practical prescription environment in the domestic market for the time being.Uplizna had also previously halted its listing process last October at the pricing negotiation stage due to supply-related issues.At the time, the company had accepted the “below the evaluated amount” condition set by the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee and entered price negotiations. However, the parties failed to reach an agreement within the 60-day negotiation period. Subsequently, HIRA attempted to enter into extended negotiations, but the talks could not begin as the pharmaceutical company was unable to ensure domestic supply.Supply-related issues for Uplizna remain unresolved. The drug was originally developed by Amgen, while Tanabe holds commercialization rights for Korea and other Asian countries through a licensing agreement.It remains to be seen what steps Uplizna will take next after failing its second attempt to secure reimbursement coverage in Korea.NMOSD occurs when AQP4 autoantibodies, a disease-specific biomarker produced by B cells, bind to AQP4, a target antigen present on glial cells in the central nervous system, and activate the immune responses, causing nerve damage.Uplizna is an anti-CD19 human monoclonal antibody that selectively binds to CD19, a B-cell-specific surface antigen, depleting B cells that produce AQP4 antibodies, thereby preventing disease relapse.The safety and efficacy of Uplizna were demonstrated in the N-MOmentum study, which evaluated the use of Uplizna monthly in 230 patients without the use of concomitant immunosuppressive agents.Study results showed that 89% of patients treated with Uplizna did not experience a relapse during 197 days of follow-up, resulting in a 77.3% reduction in the risk of relapse compared to placebo. Safety evaluations of Uplizna also showed comparable rates of adverse events to the placebo group.
- Company
- Patient & Consumer Alliance says "Structural reform needed for drug pricing reform"
- by Son, Hyung Min Mar 31, 2026 08:45am
- Patient & Consumer Alliance for Healthcare Rights (PCA) provided both positive evaluations and concerns over structural limitations regarding the recent drug pricing system reform. The drug price cut is a significant change; However, they argue that a breakthrough reform remains difficult as long as the market remains centered on rebate-driven structures.On the 27th, the PCA stated as such after the Health Insurance Policy Review Committee approved the "Measures to Improve the National Health Insurance Drug Pricing System" the previous day.The Patient & Consumer Alliance for Healthcare Rights (PCA) was officially launched on the 24th joined by four public citizens and patient organizations, including the Consumers Union of Korea, the Korea Alliance of Patient Organizations, Citizens' Movement for Consumers, and the Korean Organization for Rare Diseases, with the goal of shifting the current "government-led and provider-centric" medical structure toward a patient- and consumer-centered one (photo= Korean Organization for Rare Diseases).Previously, the Ministry of Health and Welfare (MOHW) finalized a plan to lower the drug price calculation rate for generics and off-patent drugs from the current 53.55% to 45%. The PCA evaluated this measure as "meaningful progress, marking the first reform of the generic drug pricing structure in 14 years."However, the PCA stated that price reductions alone are insufficient to eradicate the practice of rebates.The PCA stated, "Drug prices are not commensurate with rebates and that rebate practices will persist as long as the unfair competitive structure across licensing and distribution remains intact."They also raised concerns about the tiered price adjustment structure and the newly introduced "New Innovative Pharmaceutical Company" system included in the reform.According to the PCA, "The grace period of up to 10 years delays market restructuring," adding, "New Innovative Pharmaceutical Company" system lacks clear criteria and evaluation methods, potentially allowing companies without innovative capabilities to be incorporated into the support system.Furthermore, the PCA identified potential issues with the government's policies supporting the pharmaceutical and biotech industries. They argued that if this continues, with savings from price cuts being reinvested in industrial support, it could result in using taxpayer money to sustain companies with uncertain competitiveness.The PCA further stated, "The scale and performance of investments into pharmaceutical R&D over the 26 years since the separation of prescribing and dispensing have never been fully disclosed to the public," adding, "Discussions on additional support should be provided only after independent performance evaluations are made public."Issues were raised regarding the sales structures of certain companies within the Korean pharmaceutical market.The PCA pointed out, "There may be so-called bogus pharmaceutical companies that maintain market presence through rebate-driven sales via Contract Sales Organizations (CSOs) without possessing production facilities or research capabilities," and argued that "These companies undermine the competitive foundation for legitimate companies."The PCA stated, "If this structure persists even after lowering drug prices, companies relying on rebates, rather than those with true competitiveness, will maintain their market status," and adding, "The key is the normalization of the competitive order rather than just price reduction."The PCA stressed that government support is necessary to improve the pharmaceutical market structure. They called for the immediate launch of a 'Pharmaceutical Market Fair Trade Task Force (TF)' involving the Ministry of Health and Welfare, the Ministry of Food and Drug Safety, and the Fair Trade Commission to promote: ▲Eradicating rebates and strengthening CSO management ▲Establishing exit criteria for pharmaceutical companies without production capabilities ▲Disclosing performance results of financial support for pharmaceutical R&D.The PCA stated, "Drug price reduction is only the beginning, and as long as the rebate structure remains, patients may not receive benefits," adding, "The government must prioritize creating a foundation for fair competition before returning financial savings to the industry."The PCA concluded, "We hope this pricing reform serves as a starting point for reform leading to a fair pharmaceutical market," and stated, "The alliance would continuously monitor the implementation process." The PCA emphasized that government support is necessary to improve the pharmaceutical market structure.
- Opinion
- "Joint bioequivalence licensing system is driving abundant generics"
- by Lee, Jeong-Hwan Mar 31, 2026 08:45am
- Director Lee Jae HyunWhile the government announced plans to divert from the multi-item generic structure and improve the pharmaceutical industry’s trend by focusing on new drugs through drug pricing reforms, criticism has emerged that there is a need to innovate by diagnosing blind spots in the "approval system" beyond pricing alone.In Korea, it has been pointed out that the so-called '1+3 system,' under which the government grants marketing authorization for generics based on a single joint bioequivalence (BE) study conducted by one contract manufacturer and up to three consigned pharmaceutical companies, must be discarded.Furthermore, it has been suggested that, for the long-term development of the domestic pharmaceutical industry, the government must make a policy decision to allow South Korea's world-class clinical physician workforce to enter new drug development.In an interview on the 29th at the Sungkyunkwan University Center for Pharmaceutical Regulatory Sciences in Yeongdeungpo, Seoul, Director Lee Jae Hyun (Professor at Sungkyunkwan University College of Pharmacy) emphasized, "Why we are seeing hundreds of generics for a single active ingredient today is not due to high drug prices, but because of the joint bioequivalence licensing system."Regarding the '1+3 system,' which grants generic marketing authorization even to companies that did not conduct in-house BE tests but instead outsourced them to a contract manufacturer, Lee evaluated it as a "policy with no global precedent."Lee questioned whether a company can truly be called a "pharmaceutical company" if it simply purchases BE test data conducted by others, obtains authorization for identical twin generics, and profits by releasing them to the market after merely changing the brand name and packaging.Lee noted, "Although it was reduced to '1+3' (from the previous unlimited structure), the concept of joint BE licensing is preposterous. Even a '1+1' system, where only one consigned license is granted per contract manufacturer, makes no sense. Lee stated, "This is the fundamental cause of generic proliferation, which undermines the fundamentals of regulatory science. Why do drugs with identical shapes, ingredients, and dosages have different manufacturers, brand names, and packaging?"Lee emphasized, "If one set of BE data exists, marketing authorization should be granted only to that specific pharmaceutical company. As long as this joint BE licensing system remains in place, a structure in which generic substitution is hindered, and generics proliferate, will persist. We should not allow companies to outsource production just by hanging up a 'pharmaceutical company' sign. We must abolish these mismanaged licensing and joint BE policies."Lee explained, "If the licensing policy is corrected, drug price management can also be designed much more rationally. A policy could be designed that grants 'branded generic' rights only to the original and the first generic, while all other generics are authorized as 'no-brand generics' using the ingredient name rather than a brand name," adding, "Policies to increase generic utilization could become possible, and the controversy over INN (International Nonproprietary Name) prescribing would disappear, as the product name itself would be authorized as the ingredient name."Director Lee also believes that to develop Korea-Blockbuster new drugs, Korea’s abundant clinical physician workforce must be funneled into new drug development, where basic science is essential.While the stages of new drug development include discovery, demonstrating efficacy and safety through clinical trials, and obtaining marketing authorization, Lee suggests establishing policies to recruit physicians, one of Korea's greatest strengths, in the actual development of new drugs.Director Lee suggested, "New drugs are not developed by government pricing policies or pharmaceutical companies. They emerge from the advancement of a country's life sciences and the continuous increase in research on new drug substances," adding, "I believe the cultivation of talent for developing new drugs ultimately lies in the utilization of the physician workforce. Implementing administration that actively utilizes Korea's globally powerful resource in the clinical trial field is the shortcut to becoming a powerhouse in new drugs."Lee stated, "We must allow doctors to engage in new drug activities through policies such as designating university hospitals as new drug development centers, providing military service benefits to participating doctors, and expanding national-level support for new drug ventures and startups within those institutions. It is difficult to develop new drugs solely within the pharmaceutical industry or regulatory science. A policy decision is needed to use doctors as the foundation for blockbuster new drugs."Lee added, "Furthermore, we need to consider amending the outdated Pharmaceutical Affairs Act. Korea's Pharmaceutical Affairs Act has not been changed from a generic-centered legal structure since the 1950s. While a pharmaceutical company is defined as one that manufactures and produces drugs, there is no system where a new drug developer can receive product approval as a pharmaceutical company."Lee concluded, "We need to create a Pharmaceutical Affairs Act to manage pharmaceutical personnel, have a Drug Safety Management Act handle synthetic drugs and new drug approvals, and separate biological products from synthetic drugs through a "Biopharmaceutical Act" or an "Advanced Biologics Act," adding, "This is a matter that requires a shift in the government's regulatory paradigm. It is time to consider shifting the manufacturer-centered Pharmaceutical Affairs Act into a basic act and separately operating laws for drug distribution management, synthetic drug licensing, and biological products."
- Policy
- Premiums for incrementally modified drugs remain uncertain
- by Jung, Heung-Jun Mar 31, 2026 08:45am
- As the government has finalized the overall framework of the drug pricing system reform, discussions are expected to move into detailed areas, including premiums for incrementally modified drugs.The core of this drug pricing system reform is the innovative level of companies. However, decisions regarding product-level premium measures, such as those for incrementally modified drugs, have been left out.According to industry sources on the 27th, since the premium rate for incrementally modified drugs was not clearly determined at this Health Insurance Policy Deliberation Committee meeting, follow-up discussions are expected to continue.The reform plan discussed at the Health Insurance Policy Deliberation Committee meeting last November had tentatively decided to maintain the current premium system for “incrementally modified drugs, incrementally modified combination drugs, and biosimilars.”However, this wording was deleted at the latest HIPDC meeting. With the addition of a new “quasi-innovative company” category, the drug price premium scheme has been significantly revised. The disclosed premium preferential measures consist solely of a 60% premium for innovative drugs, a 50% premium for quasi-innovative drugs, and preferential treatment for pharmaceutical companies and drugs that ensure supply stability.Under the current pricing calculation system, incrementally modified drugs receive premiums on top of the base price. For salt/formulation changes after original patent expiry, the price is set at 70% with a premium from the base price (53.55%). The price is set at 77% with a premium from the base price (58.9%). The premium is generally applied for 1 year after listing, but can be extended up to 3 years if there are three or fewer generic manufacturers listed for reimbursment.The key question is whether the government will maintain these premium rates. Since the base calculation rate has been lowered, there is a possibility that the premium rates will also be readjusted. If the government attempts to lower the premium rates for incrementally modified drugs in line with the reduced base rate, strong industry backlash is expected. The prevailing view in the industry is that if the government attempts to adjust the premium rate as well, it will dampen the motivation to develop incrementally modified drugs.There is also the possibility of reforming premiums for incrementally modified combination drugs. Currently, their price is calculated as the sum of 53.55% of each component drug’s pre-patent-expiry price. In this case, innovative pharmaceutical companies receive preferential treatment with a 68% sum, while general pharmaceutical companies receive a 59.5% sum.Given that drug price premium tiers are currently divided into innovative, quasi-innovative, and non-innovative categories, changes are needed in the calculation rates for incrementally modified drugs as well.In particular, since the “quasi-innovative” category is a newly established preferential pricing bracket, discussions must also address whether to apply differential premium rates within this category.Industry insiders agreed that, as the basic calculation rates and drug price premiums had not been finalized until now, concrete discussions must begin immediately.A pricing manager at a pharmaceutical company stated, “Until now, the focus was on establishing the overall framework, so detailed discussions were not possible. Now, discussions on incrementally modified drug premium rates must begin.”Another industry official added, “The content announced at this Health Insurance Review and Assessment Service (HIRA) meeting only covers the broad framework, so significant detailed adjustments are needed. The same applies to the premium rate for incrementally modified drugs. While each pharmaceutical company may focus on different aspects of the reform, since the base calculation rate has decreased, it is necessary to ensure that premiums are maintained as much as possible.”
- Opinion
- ‘Revisiting Xeljanz safety concerns based on accumulated long-term data’
- by Son, Hyung Min Mar 31, 2026 08:45am
- Eun-mi Song, Professor of Gastroenterology and Hepatology, Ewha Womans University Seoul HospitalAs treatment strategies for ulcerative colitis shift toward maintaining long-term remission, the criteria for selecting treatment options are also changing.In particular, as Janus kinase (JAK) inhibitors establish themselves as key treatment options alongside biologics, safety concerns surrounding Pfizer’s ‘Xeljanz (tofacitinib),’ including major adverse cardiovascular events (MACE) and thrombosis, have been consistently raised.Amid this context, as results from domestic cohort studies involving Korean patients accumulate, there is a growing movement to reassess its safety in real-world clinical settings.Professor Eun-mi Song of the Department of Gastroenterology at Ewha Womans University Seoul Hospital, who led this study, recently told Daily Pharm, “Initially, there were significant concerns about side effects due to the mechanistic characteristics of JAK inhibitors. Looking at actual clinical data, contrary to expectations, the safety profile is comparable to that of existing biologics.”Ulcerative colitis is a disease characterized by chronic inflammation of the colon's mucosa, with recurring symptoms including diarrhea, bloody stools, and abdominal pain. Unlike acute colitis, it is a chronic condition with no clear cause that involves repeated cycles of remission and relapse, requiring many patients to continue treatment for the rest of their lives.The number of patients in Korea is also rapidly increasing. Due to Westernized dietary habits and environmental changes, prevalence is rising, particularly among younger patients aged 20–40. As the number of patients in this age group increases, which is highly active in society, the need for long-term disease management also grows.At the same time, the treatment landscape is evolving. While a step-up strategy, gradually increasing treatment intensity, was previously the norm, an ‘accelerated step-up’ strategy, which adjusts treatment intensity more quickly based on the patient’s condition, is now being applied in clinical practice. Treatment goals are also evolving beyond simple symptom relief toward the fundamental suppression of inflammation, such as achieving endoscopic remission.The problem lies in the high recurrence rate. Since more than 80% of patients experience recurrence and some progress to severe disease, the continuity of treatment, which maintains stable suppression of inflammation even after initial remission, is identified as a key factor determining long-term prognosis.Accordingly, efforts are ongoing to establish strategies for maintaining long-term remission while also validating drug safety in real-world clinical settings.In particular, JAK inhibitors have faced persistent safety concerns since their introduction, including risks of MACE, thrombosis, infections, and malignancies.However, it has been pointed out that these risks were primarily derived from data on rheumatoid arthritis, which involves a large proportion of elderly patients, and that there are limitations to applying them directly to the ulcerative colitis patient population, which has a relatively high proportion of younger patients.Furthermore, in actual clinical practice, the patterns of adverse reactions may vary depending on patient comorbidities, age, and concomitant therapies, reinforcing the need for the collection of real-world data from domestic patient populations.Against this backdrop, a large-scale population-based cohort analysis was conducted in Korea. Using data from the National Health Insurance Service (NHIS), the study compared the risk of serious adverse events (SAEs) between the Xeljanz group (521 patients) and the TNF inhibitor group (1,295 patients) in patients with moderate-to-severe ulcerative colitis from May 2019 to April 2022.Analysis revealed that the overall incidence rate of SAEs was 4.41 per 100 person-years in the Xeljanz group and 5.33 in the TNF inhibitor group, showing no statistically significant difference between the two treatment groups. In particular, no differences were observed between the groups in the risk of thromboembolism, opportunistic infections such as herpes zoster and tuberculosis, or malignancies.Professor Song stated, “The occurrence of complications is influenced more by individual risk factors, such as the patient’s age or underlying conditions, than by the drug itself. If treatment and monitoring are conducted in conjunction with consideration of each patient’s risk level, Xeljanz is a viable long-term treatment option.”Q. Given the reimbursement criteria, the top-down approach seems ideal, but the step-up approach still predominates in real life.In Korea, ulcerative colitis treatment is moving toward an ‘accelerated step-up’ approach, which is a practical compromise between top-down and traditional step-up strategies. This involves closely monitoring patient response and rapidly escalating to more potent therapies when initial treatments are insufficient, enabling early remission.In the past, treatment typically began with 5-aminosalicylic acid (5-ASA) agents, followed by sequential use of immunomodulators in a stepwise approach; however, in recent practice, steroids or immunomodulators are used from the outset in patients with severe symptoms. In particular, if the disease continues to worsen despite this initial intervention, biologics or small-molecule agents (JAK inhibitors) such as Xeljanz are introduced early on, in accordance with domestic health insurance reimbursement criteria.Q. When considering switching, what specific criteria are used to make the change?Disease severity is the primary factor. Physicians assess symptom severity, endoscopic inflammation, and laboratory results to evaluate disease status. The first criterion is selecting the treatment with the highest expected efficacy based on severity, followed by safety considerations.The criteria for selecting ulcerative colitis treatments have evolved to comprehensively consider not only the patient’s clinical characteristics but also safety in relation to comorbidities, as well as the patient’s individual preferences and lifestyle patterns. While treatment options were limited in the past, the recent introduction of new drugs with diverse administration routes and schedules has made it possible to design sophisticated treatment plans tailored to each patient’s specific situation.Q. How was the Xeljanz cohort study conducted?This large-scale, population-based cohort study of Korean ulcerative colitis patients was designed to directly compare Xeljanz with TNF inhibitors, which have been in clinical use for a relatively long period and are considered to have an established safety profile, as the control group.The study results confirmed that the safety profile of Xeljanz is comparable to that of existing biologics, namely TNF inhibitors. Initially, due to its mechanism of action, it was anticipated that the Xeljanz group would have a relatively higher risk of viral infections or thrombosis, however, actual analysis revealed no statistically significant difference in the incidence of serious adverse events between the two treatment groups.However, a major limitation of this study is that specific data on initial drug dosing were not obtained during the study, preventing precise analysis of dose-dependent safety and efficacy differences. Previous studies, such as ORAL Surveillance, have already suggested that dosage differences in JAK inhibitors can have a significant impact on safety outcomes, and there remains a possibility that this study could also reveal differences in clinical outcomes based on dosage.Q. Despite the periodic release of safety data on Xeljanz, concerns regarding risks still persist.Even among healthcare professionals, there is a vague fear of complications when prescribing JAK inhibitors like Xeljanz. However, clinical data reported domestically and internationally to date show that these concerns do not translate into actual risks. In conclusion, it has been confirmed that effectively controlling the inflammatory state early on with Xeljanz, which has a potent and rapid effect, actually contributes to reducing the risk of disease-related complications and ensuring long-term patient safety.Furthermore, compared to Western populations, the absolute probability and incidence of thrombosis in Asian patient groups have been observed to be relatively lower. While Western populations exhibit higher thrombosis rates due to factors such as larger body frames and a higher proportion of obese individuals, Asian populations show a similar trend of increased risk compared to the general population, yet the absolute number of cases tends to be lower.Previous safety warnings regarding JAK inhibitors were primarily based on data from rheumatoid arthritis, which involves a large number of elderly patients in their 50s and 60s. However, since the ulcerative colitis patient population consists mostly of younger individuals, the risks of thrombosis and other complications, which were raised in the rheumatoid arthritis data dominated by elderly patients, were found to be relatively lower.Q. Do clinical experiences in actual prescribing practice show similar patterns to study findings in terms of efficacy and safety?Combined results from domestic multicenter studies and real-world clinical experience indicate that the safety concerns raised during the early stages of the introduction of JAK inhibitors, including Xeljanz, do not pose a significant problem in real-world clinical settings. In particular, regarding herpes zoster, which was expected to carry a high risk based on the mechanism of action, no serious safety issues as previously feared emerged, thanks to thorough preemptive vaccination of healthcare providers and close monitoring. On the contrary, the greatest strength of Xeljanz perceived in clinical practice was its very rapid and potent efficacy, providing immediate therapeutic benefits to patients in urgent need of rapid symptom improvement.Q. What are the limitations of the current treatment environment, including reimbursement?While Western practice emphasizes top-down strategies for improved long-term outcomes, Korea faces practical constraints regarding the application of early, potent treatment due to the National Health Insurance system and financial limitations.Thus, greater flexibility in enabling early use of potent therapies, especially for severe patients, is considered essential for improving treatment outcomes. Furthermore, recent accumulated data have demonstrated that switching between different JAK inhibitors yields clinically significant efficacy. Consequently, it is anticipated that switching between different JAK inhibitors, even if a patient shows an inadequate response to a specific JAK inhibitor, may provide meaningful clinical benefits, offering additional options for patients who do not respond adequately to a specific agent.Q. What is the clinical significance of JAK inhibitors in ulcerative colitis?Despite their relatively recent introduction, small-molecule therapies have become a core pillar, playing a central role in the treatment of ulcerative colitis. While there was once a vague apprehension regarding these treatments even among clinicians, the experience and data shared by professors who have prescribed drugs such as Xeljanz in actual clinical practice confirmed that the risk of complications, which had been a concern, was lower than expected and manageable.In particular, for moderate-to-severe patients who struggled with the burden of even daily outings due to recurring cycles of symptom improvement and flare-ups, oral small-molecule agents, which offer high convenience, have become a practical alternative that dramatically improves quality of life. With the recent expansion of available treatment options to 3 or more, led by the introduction of Xeljanz, and ongoing new drug development, it is crucial for patients to maintain hope and work closely with healthcare professionals to establish a treatment strategy optimized for their individual needs in order to maintain long-term remission.