- LOGIN
- MemberShip
- 2026-04-24 06:16:47
- Company
- IgA nephropathy drug Vanrafia soon to be introduced to Korea
- by Eo, Yun-Ho Mar 05, 2026 05:30pm
- Vanrafia, a new treatment for the rare kidney disease IgA nephropathy, may soon enter the Korean market.According to industry sources, Novartis Korea recently submitted a marketing authorization application to the Ministry of Food and Drug Safety (MFDS) for Vanrafia (atrasentan), a treatment for adults with primary IgA nephropathy (IgAN) with a urinary protein-to-creatinine ratio (UPCR) of 1.5 g/g or higher.Vanrafia was designated an orphan drug in Korea in August last year and previously received accelerated approval from the U.S. Food and Drug Administration (FDA).The drug is a once-daily oral non-steroidal therapy that can be used in combination with supportive treatment, including renin–angiotensin system (RAS) inhibitors, and may also be used in combination with sodium-glucose cotransporter-2 (SGLT-2) inhibitors.Vanrafia demonstrated efficacy in an interim analysis of the Phase III ALIGN study. However, the study has not yet confirmed whether the drug can slow the decline of kidney function in IgAN patients.In the ALIGN study, patients receiving Vanrafia in combination with RAS inhibitors experienced a clinically meaningful and statistically significant 36.1% reduction in proteinuria compared with the placebo group. This effect was observed as early as 6 weeks and was sustained for 36 weeks.The effect of Vanrafia on UPCR was consistent across subgroups in the primary study cohort, including differences in age, sex, race, estimated glomerular filtration rate (eGFR), and baseline proteinuria.IgA nephropathy is a progressive, rare autoimmune kidney disease in which the immune system attacks the kidneys, often causing glomerular inflammation and proteinuria.Up to 50% of IgAN patients with persistent proteinuria progress to kidney failure within 10 to 20 years after diagnosis, eventually requiring maintenance dialysis or kidney transplantation. Treatment responses can vary among patients.
- Policy
- MFDS ‘GMP revocation system reform and ongoing litigation are separate matters’
- by Lee, Tak-Sun Mar 04, 2026 05:41pm
- AI-generated imageAs the Ministry of Food and Drug Safety (MFDS) moves forward with legislative revisions to the GMP certification revocation system in cooperation with the National Assembly, it has clarified that the changes will not apply to pharmaceutical companies that previously had their GMP certifications revoked.The ministry also drew a clear line regarding companies currently engaged in litigation, stating that the institutional reform is separate from ongoing lawsuits.According to the MFDS on the 3rd, the proposed amendment to the Pharmaceutical Affairs Act, jointly introduced on the 27th of last month by Rep. Jong-heon Baek (People Power Party) and Rep. Mi-hwa Seo (Democratic Party of Korea), reflects findings from last year’s research on improving the GMP certification revocation system.The GMP revocation system, implemented in December 2022, is a so-called “one-strike out” rule under which the MFDS revokes GMP certification for manufacturing sites that seriously violate Good Manufacturing Practice standards or repeatedly falsify manufacturing and quality records.The MFDS revokes GMP certification or changes compliance determinations if they were obtained through ‘false or fraudulent’ means or if manufacturing/quality management records were ‘repeatedly’ falsified or incorrectly documented.Additionally, it issues a ‘correction order’ if there is a risk of quality impact due to the lack of detailed standards or procedures for GMP compliance.The industry has previously argued that penalties under this system are excessively harsh, while also pointing out that criteria such as ‘repeated’ within the legally stipulated regulations lack clarity.Therefore, this amendment to the Pharmaceutical Affairs Act introduces intermediate regulations that allow for the suspension of GMP certification for up to 6 months and corrective orders to be issued, depending on the severity of the violation, when simple errors occur in GMP record-keeping or when there are minor issues in GMP compliance or operation.Furthermore, a new regulation has been established stipulating that pharmaceutical companies that fail to create or retain GMP records will have their certification immediately revoked, similar to cases of false or fraudulent certification. This aims to prevent deliberate failure to prepare GMP records to evade certification revocation.Since the revocation system took effect, GMP certification revocations were notified to 8 companies as of last October, beginning with Hutecs Korea. Of these, 5 companies are currently pursuing administrative litigation, while penalties for 3 have been finalized.An MFDS official stated, “In principle, the amendment to the Pharmaceutical Affairs Act cannot be applied retroactively. Companies currently involved in litigation may submit opinions regarding the amendment, but legislative changes and lawsuits are separate matters.”This suggests that the MFDS intends to continue defending its position in court against companies that have filed lawsuits.The MFDS official stated, “We understand that once this revised Pharmaceutical Affairs Act is promulgated, it is expected to take effect one year later. Accordingly, we plan to proceed with revising the details in accordance with the legal provisions through amendments to the Prime Ministerial Decree (Regulations on the Safety of Pharmaceuticals).”The reason opposition lawmaker Jong-heon Baek of the People Power Party joined the MFDS in pursuing this amendment is that, after Baek spearheaded and passed the initial Pharmaceutical Affairs Act amendment addressing the revocation of GMP compliance certifications, the industry flooded the MFDS with requests for further revisions.In response, Rep. Baek’s office requested that the MFDS conduct research on potential improvements, ultimately leading to the current legislative proposal. The amendment is jointly sponsored by Rep. Mi-hwa Seo of the Democratic Party of Korea, reflecting bipartisan support for refining the system.
- Company
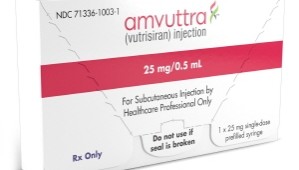
- Amvuttra for multiple neuropathy now prescribed at hospitals
- by Eo, Yun-Ho Mar 04, 2026 05:41pm
- A RNAi-based drug, 'Amvuttra,' currently seeking reimbursement listing, has entered the prescription area of general hospitals.According to industry sources, the new drug Amvuttra, a treatment for hereditary transthyretin-mediated amyloidosis with polyneuropathy (hATTR-PN), introduced to the Korean market by Medison Pharma Korea, has passed the drug committees (DC) of tertiary general hospitals, including Seoul Samsung Medical Center and Sinchon Severance Hospital.Medison Pharma is currently negotiating with the National Health Insurance Service (NHIS) for the drug price. Accordingly, once this drug is listed, it is expected to proceed quickly to prescription. Amvutra previously passed the Health Insurance Review and Assessment Service's (HIRA) Drug Reimbursement Evaluation Committee in December last year.Furthermore, Medison Pharma is currently operating Amvuttra's Expanded Access Program (EAP) for patients.Amvutra was designated as an orphan drug by the Ministry of Food and Drug Safety (MFDS) in November 2023 and received final approval in November last year.Amvutra is administered by subcutaneous injection once every 3 months. This drug targets and silences specific messenger RNA (mRNA), thereby blocking the production of both wild-type and mutant transthyretin (TTR) protein.The efficacy of Amvutra was demonstrated through the HELIOS-A Phase 3 study. The Phase 3 trial enrolled 164 patients with hATTR-PN and polyneuropathy across 22 countries. These patients were randomly assigned to either the Amvutra group (122 patients), receiving 25mg via subcutaneous injection every three months, or the 'Onpattro (patisiran)' group (42 patients), receiving 0.3mg/kg via intravenous injection every three weeks.Amvutra's efficacy was assessed by comparing its data with placebo data from the APOLLO study, which evaluated the efficacy and safety of Onpattro in a patient population similar to that in HELIOS-A.As a result, during the 9-month treatment period, the Amvutra group experienced less severe neurological impairment and showed improved quality of life compared to the placebo group. In the 10-meter walk test, which assesses a patient's walking speed and motor ability, the time taken by the vutrisiran group showed almost no change. NT-proBNP, a biomarker used to evaluate cardiac function, also showed improvement.Meanwhile, hATTR-PN, which affects approximately 1 in 100,000 people, is a disease caused by mutations in the transthyretin gene. It is characterized by systemic multiple autonomic neuropathies, including cardiac, gastrointestinal, and ophthalmic. Vyndaqel stabilizes the transthyretin protein.Generally, symptoms such as pain, paresthesia, and paralysis begin in the lower extremity nerves, where abnormal proteins tend to accumulate, eventually spreading to the upper body and affecting other organs, such as the heart, kidneys, and eyes. Life expectancy is 7 to 12 years on average from the onset of symptoms.
- InterView
- [Reporter’s View] Polypharmacy management no longer a pilot project
- by Jung, Heung-Jun Mar 04, 2026 05:41pm
- Launched in 2018 as the “Proper Medication Use Support Program,” the National Health Insurance Service’s polypharmacy management project has now entered its ninth year.Although it remains designated as a pilot program, the time has come for the government to make the decision and transition it into a full-scale program.With the Integrated Care Support Act taking effect this year, the government must expand community-based medical and care services. This is a critical window to formally incorporate the polypharmacy management program, which has long remained outside the institutional mainstream.Elderly individuals who require integrated care services often take multiple medications due to frequent hospitalizations or outpatient visits. To achieve a truly home-centered, integrated care model, proper management must begin at an earlier stage.In other words, minimizing medication-related issues that arise (or may arise) as elderly patients move between medical institutions and home is essential to improving the overall quality of home-based services.For this reason, the hospital-based polypharmacy management model should be prioritized for transition to a full-scale program. Since 2020, participation and patient coverage have steadily expanded, while multidisciplinary collaboration among physicians, pharmacists, and nurses has become well established.Because the program operates through inpatient/discharge and outpatient models, it can significantly contribute to managing patients returning to their residences after receiving medical services.If the support is integrated care for elderly patients who have already received primary management, local governments can provide the service relatively easily. It also enables a smoother link between hospital-based and community-based models.The effectiveness of the polypharmacy management program has been sufficiently demonstrated over the past 9 years. Research results show not only a reduction in the risk of adverse drug reactions but also cost savings.Considering the financial benefits from reduced patient readmissions and additional outpatient visits, the budget required for full-scale implementation would not be substantial.At a time when the sustainability of the national health insurance system is becoming increasingly important, strengthening polypharmacy management can serve as a safeguard against unnecessary spending. While expanding participating institutions remains important, the program should now be scaled into a nationwide service through full implementation.A pilot program is meant to test effectiveness and reduce trial and error. After 9 years of validation, it is now time to transition to full-scale implementation, starting with the hospital model and gradually incorporating insurance funding.
- Policy
- Pharma exports surpassed $10 billion threshold
- by Lee, Jeong-Hwan Mar 04, 2026 05:41pm
- Lee Hyung-hoon, Second Vice Minister of MOHWIn 2025, South Korea's bio-health industry export sales reached a record high of KRW 27.9 billion. The Ministry of Health and Welfare (MOHW) met with exporting companies to listen to industry voices and gather opinions on support measures.Notably, pharmaceutical exports surpassed the $10 billion threshold for the first-time last year, recording $10.4 billion.This figure represents an approximately ten-fold increase over the past ten years as biopharmaceuticals, which account for 62.6% of pharmaceutical exports, expanded their export markets, focusing on the United States and Europe.Lee Hyung-hoon, Second Vice Minister of MOHW, held a corporate briefing on the activation of bio-health industry exports at City Tower in Seoul at 2:00 PM on the 3rd. Lee encouraged the efforts of export company officials and promised to strengthen support.After sharing industry trends, Vice Minister Lee sought government support measures by listening to difficulties in the field.Among pharmaceutical companies, Samsung Biologics, GC Biopharma, HK inno.N, and Alteogen attended. In the medical device field, CGBio, Vuno, Meerecompany, and Wontech attended.The briefing began with presentations on the 2026 outlook and export activation support plans, followed by officials from each exporting company presenting the difficulties they face and proposed policy improvement measures. It concluded with a free discussion for all attendees.◆2025 export performance=Last year, the export value of the bio-health industry, including pharmaceuticals, biotech, medical devices, and cosmetics, reached $27.9 billion, an increase of 10.3% compared to the previous year, despite uncertain conditions such as tariffs. This recorded an all-time high and ranked 8th among South Korea's major industries.Specifically, in terms of last year's performance by major industry, semiconductors ranked first with $173.4 billion, followed by automobiles at $72.0 billion, general machinery at $46.9 billion, petroleum products at $45.5 billion, petrochemicals at $42.5 billion, ships at $31.8 billion, steel at $30.3 billion, and bio-health at $27.9 billion.Notably, pharmaceuticals surpassed $10 billion for the first time, recording $10.4 billion in exports and achieving their highest-ever performance.This is the result of biopharmaceuticals, which account for 62.6% of pharmaceutical exports, expanding their markets in the United States and Europe and increasing approximately tenfold over the past decade.By country, the United States, Switzerland, and Hungary were the major countries for exports, accounting for 39.5% of total exports and showing steady increases centered on advanced markets.The export value of biopharmaceuticals grew from $670 million in 2015 to $3.49 billion in 2020, and further increased approximately tenfold to $6.52 billion last year.Medical devices, including in-vitro diagnostic devices, shifted back into a recovery trend, and general medical devices also made progress with steady growth. By country, the United States, China, and Japan accounted for 33.1% of total exports, maintaining the top three positions.◆2026 export outlook=The MOHW announced an export target for the bio-health industry of $30.4 billion, a 9.1% increase compared to last year.This year's goals are $11.7 billion for pharmaceuticals (+12.4%), $6.2 billion for medical devices (+2.7%), and $12.5 billion for cosmetics (+9.5%).In pharmaceuticals, domestic biopharmaceuticals are expected to lead export growth, driven by the global expansion of the biopharmaceutical market and South Korea's position as the world's number one in Contract Development and Manufacturing Organization (CDMO) capacity.This is the outcome of the current status for biosimilar development following the patent expirations of 58 original biopharmaceuticals through 2032, and the encouragement of biosimilar prescriptions in the United States and the EU.In medical devices, export expansion is expected in advanced and emerging markets for ultrasound imaging diagnostic devices and X-ray equipment incorporating AI technology, as they increase diagnostic accuracy and efficiency, in line with trends in aging populations and the AI transition.To support export activation, the MOHW will invest KRW 233.8 billion, a 3.5-fold increase from last year, to overcome export uncertainties and actively foster the bio-health industry through investment promotion, supply chain strengthening, overseas regulatory response, consulting, marketing, and the establishment of local bases. ◆Pharmaceutical and bio field=The MOHW will strengthen competitiveness by creating an investment environment. Based on industry suggestions and analysis of past performance, the ministry will promote improvements to the innovative pharmaceutical company certification system for companies with excellent R&D investment and continue to form a KRW 1 trillion mega-fund to ensure that domestic drug pipeline-based new drugs reach global commercialization.Specifically, a new specialized fund of KRW 150 billion will be established this year to support Phase 3 clinical trials, which require the most capital during new drug development, and more than KRW 1 trillion in national health and medical R&D will catalyze expanding private R&D investment.The MOHW will also revitalize the industrial ecosystem by strengthening the pharmaceutical supply chain. To prepare for supply chain crises, the ministry will prepare an integrated stabilization response system, including securing raw materials and parts, supporting production facilities for drugs with unstable supply, and supporting the stockpiling of core medicines.First, it will support the stabilization of new biomaterials) and expand support for diversifying new raw material purchases (10 companies, KRW 1.5 billion). It will also support the production of drugs with unstable supply (4 companies, KRW 3.8 billion), the stockpiling of new core medicines (5 companies, KRW 500 million), and the advancement of manufacturing for new promising export medicines (15 companies, KRW 8.1 billion).To strengthen support for global entry and export diversification, the goal is to move beyond the generic and biosimilar stages that have been the center of the pharmaceutical industry and enter the global market, with a focus on innovative new drugs.The ministry plans to strengthen full-cycle support, including 'open innovation' based on cooperation, 'consulting,' the operation of domestic accelerator programs (inbound), and key global market entry (outbound).The "K-Biopharma Next Bridge" project will be introduced to facilitate collaboration between domestic companies with promising technologies and global leading companies with high interest in the Korean market. Open innovation growth programs will be operated one-by-one, and the provision of information on regulatory changes by country and consulting for patent, legal, tax, customs, and distribution channel diversification will be strengthened.Domestically, an accelerator program will be operated to increase the commercialization success rate of technology-based startups, support for companies occupying the Boston CIC to establish a bridgehead for US market entry will be expanded from 30 to 40 companies, and various networking activities to secure global sales channels will be strengthened through export consultations, the dispatch of market pioneer groups, and support for participating in global medical societies.◆Medical device =An innovation ecosystem will be created to strengthen competitiveness, centered on the Medical Device Comprehensive Support Center, which covers the entire cycle from R&D to commercialization and overseas entry. Utilizing the Medical Device Comprehensive Information System and MDCC, the MOHW will strengthen customized consulting support for companies and provide more professional information.The Medical Device Consulting Council (MDCC) consists of 202 external experts in eight fields, including licensing and systems, R&D and clinical trials, overseas entry, commercialization, and legal and accounting.Furthermore, the ministry will strengthen technology verification and market linkage for domestic companies through the global open innovation program "New Impact Korea," which promotes cooperation with global companies, hospitals, and investment institutions.By holding "MedTech Insight," which systematically provides industry trends, regulations, and market information, the ministry will support the development of strategies aligned with field demand and lay the foundation for sustainable growth in the domestic medical device industry.To increase the export potential of the domestic medical device industry, new projects to strengthen competitiveness will be expanded.Support will be provided for the commercialization of innovative technologies and the strengthening of the competitiveness of the next-generation surgical robot industry, including support for the rapid commercialization of AI-applied products and the construction of an AI-based surgical robot innovation lab from 2026 to 2030.Additionally, the MOHW will support clinical trials and real-world evaluations through professional consulting and medical staff matching by operating six hospital-based demonstration support centers this year, thereby facilitating the acquisition of clinical evidence and market entry in domestic and overseas markets.To strengthen market-tailored export support, specialized support for major overseas markets will be increased. To facilitate local-based global entry cooperation, the ministry will support 10 companies in the US bio cluster in Houston with company occupancy and provide up to KRW 200 million per company annually to cover regulatory response costs essential for overseas market entry.The MOHW will also help secure stable sales channels by supporting marketing and auxiliary export costs amid uncertainties in the international supply chain.Furthermore, from this year, the "Medical Device Global Education and Training Support" project for overseas medical staff will be promoted to expand experience with key Korean products through linked education with overseas medical institutions, training facilities, and international medical societies, thereby strengthening product reliability and the foundation for market expansion.
- Policy
- GMP “one-strike” rule to be eased… suspension introduced
- by Lee, Jeong-Hwan Mar 03, 2026 09:21am
- The National Assembly and the government are launching a comprehensive overhaul of the “one-strike out” (immediate revocation) system for pharmaceutical GMP (Good Manufacturing Practice) compliance violations.This approach introduces an intermediate regulatory step: adding a ‘GMP suspension’ penalty before immediate revocation, depending on the severity of violations related to the manufacturing record. The suspension period can be set for up to 6 months.At the same time, a new provision will mandate immediate revocation of GMP certification for companies that fail to prepare, properly retain, or intentionally discard GMP records, equating such conduct with obtaining certification through false or fraudulent means.The reform aims to address concerns that the current system, which focuses heavily on the ‘repeated nature’ of GMP manufacturing record violations by pharmaceutical companies, has led to some unreasonable penalties.On the 27th, Rep. Jongheon Baek of the People Power Party and Rep. Mi-hwa Seo of the Democratic Party of Korea jointly introduced amendments to the Pharmaceutical Affairs Act to rationalize GMP regulations.The bipartisan push for legislation to improve the GMP one-strike-out system was influenced by the Ministry of Food and Drug Safety (MFDS) commissioning and completing a research project based on industry complaints.Previously, amid incidents involving unauthorized manufacturing and falsified GMP records, the MFDS and the National Assembly strengthened GMP sanctions to prevent recurrence.As a result, the one-strike-out system for GMP compliance violations of pharmaceutical companies was implemented in December 2022 following National Assembly legislation.However, since the law's enactment, pharmaceutical companies whose GMP certifications were revoked by the MFDS have repeatedly filed administrative lawsuits challenging the decisions. As shortcomings in the system became apparent, calls for improvement emerged from both the pharmaceutical industry and the National Assembly.Complaints frequently arose that immediate revocation of GMP certification not only halts a pharmaceutical company's drug production lines but also causes significant operational damage to contract manufacturers tied to the penalized company, necessitating pre-revocation regulatory measures.Specifically, pharmaceutical companies criticized the current GMP one-strike-out system as excessive punishment. They argued it applies the criterion of ‘repeated false or erroneous record-keeping’ without considering the severity of the violation (such as falsifying manufacturing records), its impact on human health, or the presence of intent.Pharmaceutical companies pointed out that focusing solely on repetition for GMP certification revocation can result in lighter penalties for serious violations compared to minor infractions and does not adequately account for intent.Consequently, some companies reportedly resorted to intentionally not preparing or discarding GMP records to avoid revocation triggered by repeated documentation errors.In response, the MFDS initiated a research project on the GMP compliance certification system and advanced supplementary legislation in collaboration with the National Assembly.The core revision introduces ‘GMP certification suspension’ as an intermediate sanction between corrective orders and full revocation. This penalty will be applied based on the severity of the GMP violation and its impact on drug quality.Notably, companies that fail to prepare or retain GMP records will face immediate revocation, targeting attempts to evade penalties through deliberate non-documentation or record destruction.Additionally, a new intermediate regulatory measure was established, allowing for a 6-month suspension of GMP certification for relatively minor GMP violations.The MFDS and lawmakers Jong-heon Baek and Mi-hwa Seo, who proposed the bill, plan to collaborate on advancing GMP certification revocation regulations through swift legislation.
- Policy
- Will pharma be able to request 'records of administrative sanctions' during acquisition?
- by Lee, Jeong-Hwan Mar 03, 2026 09:21am
- A bill is being developed to grant pharmaceutical manufacturers that intend to acquire an existing pharmaceutical company the authority to request and receive records of administrative sanctions imposed on the previous owner (the former operator) by administrative authorities.This legislation aims to address the issue that the current law lacks a legal basis for an assignee to verify the assignor's history of administrative sanctions.Once the bill is passed, cases in which an assignee suffers unfair losses, such as managerial damage, by acquiring a pharmaceutical company without knowledge of existing administrative sanctions are expected to disappear.On February 27, 2026, Representative Park Hyeong-soo of the People Power Party proposed a partial amendment to the Pharmaceutical Affairs Act containing these details.Under the current law, when a new operator succeeds to the status of a former operator through business transfer, inheritance, or merger between corporations, the effects of administrative sanctions on the former operator are transferred to the assignee, except for cases of good faith acquisition.In other words, if a pharmaceutical company with finalized administrative sanctions is purchased, the responsibility and effects of those sanctions are transferred to the assignee rather than the assignor.Rep. Park raised concerns that if a pharmaceutical company is acquired without knowledge of any administrative sanction history, the assignee is forced to fulfill the existing sanctions, which can sometimes lead to a situation worse than not having acquired the company at all.Accordingly, Rep. Park introduced a bill that allows a person seeking to succeed to a pharmaceutical manufacturing business (a person intending to acquire a pharmaceutical company) to request the history of past administrative sanctions and details of administrative sanctions currently in progress from the Minister of Health and Welfare (MOHW), the Minister of Food and Drug Safety (MFDS), provincial governors, or heads of local governments (mayors, governors, or district heads).Rep. Park explained, "This bill allows assignees to check the records of administrative sanctions against the previous operator in advance, enabling them to make more rational decisions regarding the acquisition," adding, "The Anti-Corruption and Civil Rights Commission has also recommended establishing procedures to verify administrative sanctions, as cases of attempting to transfer businesses to evade administrative sanctions have become frequent."
- Policy
- Reimb for switching between Evrysdi and Spinraza allowed in Korea
- by Jung, Heung-Jun Mar 03, 2026 09:21am
- Beginning in March, the scope for switching between spinal muscular atrophy (SMA) treatments Evrysdi and Spinraza will be expanded with reimbursement.With bidirectional switching now permitted between injectable and oral therapies, prescriptions can be adjusted based on changes in patient condition. Previously, only a single switch from Spinraza to Evrysdi had been allowed.AI-generated ImageOn the 27th, the Health Insurance Review and Assessment Service (HIRA) announced updated reimbursement criteria for SMA therapies, effective March 1, following revisions to the Ministry of Health and Welfare’s reimbursement standards.Until now, switching or combination therapy between SMA treatments had not been covered. Under the revised policy, a one-time conditional switch will be permitted between the injectable Spinraza and the oral formulations Evrysdi Dry Syrup and Evrysdi Tab.Switching is allowed if clinical improvement has been confirmed during treatment, the discontinuation criteria are not met, and there is a valid clinical reason. This applies not only to switching from an injectable to an oral medication but also from an oral medication to an injectable. Patients can even switch from an injectable to an oral medication and then return to an injectable.The change enhances treatment accessibility by allowing reimbursement coverage when medication adjustments are necessary based on a patient’s condition.However, patient motor function assessment criteria have become more detailed. Previously, assessment tools were applied uniformly based on whether the patient was under or over 24 months of age.Starting in March, assessment tools will instead be differentiated based on whether the patient can sit, among other refinements. This will apply to assessments conducted after implementation.With the new reimbursement listing of Evrysdi Tab in March, long-term prescription limits have also been established. The maximum prescription duration for Evrysdi Tab is 56 days.Roche Korea’s Evrysdi Tab 5mg is scheduled for reimbursement listing next month at KRW 793,333, adding a tablet formulation alongside the existing dry syrup.With the addition of a new formulation, the wording has been revised to refer to ‘Evrysdi Dry Syrup, etc.’ in prior review procedures and reimbursement criteria to reflect the expanded formulations.
- Product
- Wegopro, Mounjung? Warning over supplements resembling medicines
- by Kim JiEun Mar 03, 2026 09:21am
- The Korean Pharmaceutical Association (President Young-hee Kwon) has urged consumers to exercise caution regarding health functional foods and general food products that may be confused with prescription medicines.The KPA emphasized, “As the distribution of general foods that are classified as other processed goods with names and appearances similar to pharmaceuticals increases in the market, there is a growing risk of treatment delays, misuse, and other health harms. This is not merely a matter of product choice but an issue directly linked to public health and safety, requiring special caution.”The association noted that cases have recently been identified in which health functional foods and general foods are being sold with names and appearances similar to prescription drugs.In particular, products using names reminiscent of diabetes or obesity treatments are being distributed, raising concerns that consumers may mistake them for medicines.Some products reportedly mimic pharmaceutical packaging colors and design layouts, further increasing the risk of confusion.The KPA mentioned that it had previously raised the issue during a National Assembly Health and Welfare Committee audit last year.Rep. Bo-yoon Choi pointed out that distinctions among pharmaceuticals, quasi-drugs, and health functional foods are not clearly conveyed to consumers. She cited examples such as Ursa–Ursaji, Jeil Cool Pap–Jeil Pap Cool, and Madecassol–Madecassol Care, where similar names and packaging may blur distinctions.At the time, Rep. Choi stated, “Even if the legal distinction is clear, if identical display and similar packaging persist, consumers inevitably perceive them as the same drug,” pointing out the resulting risks of delayed treatment and side effects.Eun-gyo Kim, Director of Health Functional Foods at the Korean Pharmaceutical Association, said, “Pharmaceuticals are therapeutic products whose safety and efficacy are verified through clinical trials and regulatory approval by the Ministry of Food and Drug Safety, whereas health functional foods and general foods are supplementary products that cannot replace medicines. Selling products with similar names or that exploit the recognition and social interest in treatments for specific diseases poses a high risk of misleading consumers.Meanwhile, the KPA urged consumers to always verify the distinction between pharmaceuticals, health functional foods, and general foods, along with their labeling information, when purchasing products. It also stated that it will continue monitoring similar cases and actively pursue policy improvements to protect public health.Furthermore, it stressed the need for: ▲ Establishing prior review and restriction criteria for products using names closely resembling medicines ▲ Creating new regulatory standards for packaging and designs that evoke pharmaceuticals ▲ Strengthening mandatory distinction labels and warning statements to prevent confusion ▲ Enhancing inspection and management of advertising and online promotional practices that imply disease treatment effects.
- Policy
- MOHW to newly establish 'Bureau of Regional·Essential·Public Healthcare'
- by Lee, Jeong-Hwan Mar 03, 2026 09:21am
- 고형우 국장The Ministry of Health and Welfare (MOHW) plans to complete the establishment of the 'Bureau of Regional·Essential·Public Healthcare' within the first half of this year. The Office of the President agrees with the necessity of strengthening 'Bureau of Regional·Essential·Public Healthcare,' and the Ministry plans to create a new bureau following consultations with the Ministry of the Interior and Safety.On the 2nd, Ko Hyung-woo, Director of the Division of Essential Healthcare at the MOHW, shared these plans during a briefing with the Korea Special Press Association.Director Ko explained that the new division is still in its early stages and requires further coordination with the Ministry of the Interior and Safety.However, he added that discussions are in full swing following the passage of the Regional Essential Healthcare Act in the National Assembly, and that the Office of the President is responding positively.Consequently, the Ministry intends to establish the new Division-level organization during the first half of the year to take full charge of the new healthcare policies.Ko stated, "While the 'Bureau of Regional·Essential·Public Healthcare' will include three distinct bureaus (Regional Healthcare, Essential Healthcare, and Public Healthcare), we will not be creating all new divisions," adding, "Some will be reorganized and recombined from existing structures."Ko added, "Bureaus that require new establishment, such as those related to national medical schools, will be created. I hope that the workforce will increase under this new Bureau-level structure. The healthcare sector is short-staffed. If sufficient personnel are deployed, the quality of our policies will be significantly higher than it is now."Regarding the allocation of KRW 1.13 trillion annually for the Regional and Essential Healthcare Special Account, Director Ko said, "If we design our projects well, we can pull budget from other general finances if necessary. Therefore, the KRW 1.13 trillion is not set but it could increase or decrease."Ko emphasized, "The important part is our decision to proceed with KRW 800 billion for new projects. Typically, projects exceeding KRW 50 billion over five years must undergo a preliminary feasibility study, which takes considerable time and would prevent immediate implementation next year," adding, "Since KRW 800 billion over five years totals KRW 4 trillion, we must apply for an exemption from the preliminary feasibility study for this KRW 4 trillion plan. We are putting efforts into developing the content for this."Ko further explained, "According to our surveys of local governments, the majority believe that support is essential for childbirth, pediatrics, emergency, and cardiovascular care," adding, "While medical personnel represent the most critical portion of the KRW 800 billion budget, regional doctors will not emerge for another 10 years. Therefore, we are currently considering how to address non-physician medical personnel."