- LOGIN
- MemberShip
- 2026-04-24 08:22:21
- Opinion
- From pharmacy practice to new drug review
- by Jung, Heung-Jun Mar 25, 2026 07:20am
- Even after a new drug is developed and released to the market, there is one hurdle that must be cleared for patients to fully benefit from it: the National Health Insurance reimbursement listing.How this initial step is handled, in reviewing whether a drug should be covered by health insurance, determines both patients’ access to treatment and the balance of Korea’s insurance budget.The organization guarding that crucial gate is the Department of Drug Management of the Health Insurance Review and Assessment Service (HIRA). It is also one of the operational departments where pharmacists can create synergy based on their understanding of pharmaceuticals.Assistant Directors Jae-young Choi (left) and Jin-woo Song of the New Drug Listing Division, Department of Drug ManagementDailyPharm met with Jin-woo Song (38) and Jae-young Choi (34), assistant directors working in the Department of Drug Management, by drawing on their clinical experience in hospitals and pharmacies. We were able to hear their candid thoughts on what their new roles entail after hanging up their pharmacist’s coats, as well as the challenges and satisfactions they face.Both have been with the agency for 1-2 years. What has your career as pharmacists been like?Jin-woo Song (hereinafter Song): After graduating from pharmacy school in 2023, I worked at the Pharmacy Department of Severance Hospital for 2 years. I gained experience in the Inpatient Dispensing Unit, Special Dispensing Unit, Pharmacy Information Unit, and Outpatient Dispensing Unit before joining HIRA last June.Jae-young Choi (hereinafter Choi): I also have experience working as a night-shift pharmacist at a hospital and in a community pharmacy after graduation. I joined HIRA in December 2024, so it’s been just over a year now.Among various career options, why did you choose to join HIRA?Song: While working at the hospital, I realized that I am well-suited to a well-established system-based institution. I became curious about how reimbursement criteria and procedures for pharmaceuticals are set and decided to move to HIRA.Assistant Director Jin-woo SongChoi: After graduation, I became increasingly interested in social pharmacy. I applied because I became interested in new drug listing and management of already listed drugs to improve patient access to treatments. I found the idea of approaching pharmaceuticals from the public sector appealing.Did you prepare anything specifically in order to join HIRA?Song: Since they make the hiring process very convenient for pharmacists, there was nothing specific I needed to prepare. If you have at least 1 year of experience at a university, research institute, pharmaceutical company, hospital, or pharmacy, you only need to submit a cover letter. The written test is waived. As long as you meet the requirements, you can take the interview, and even upon joining, you are hired at Grade 4 (assistant director).What kind of work are you doing in the Department of Drug Management?Song: I have been working in the New Drug Listing Division since June last year. My work involves the overall process related to reimbursement listing for new drugs. I handle cost-effectiveness reviews related to expanded criteria for risk-sharing agreement drugs as well as assessments of the appropriateness of renewing RSAs for such drugs.Choi: I’m part of the Agenda Team within the New Drug Listing Division. I joined in December 2024, so it’s been just over a year. When I’m assigned cases involving new drugs or RSA drugs, such as applications for reimbursement decisions, requests to expand reimbursement criteria, or evaluations related to contract expiration, I review them based on criteria like clinical utility and cost-effectiveness. I then present those results to the Drug Reimbursement Evaluation Committee.How has your experience in hospitals and pharmacies been helpful?Song: I handled many anticancer drugs and medications for severe diseases at a tertiary hospital. Many of the drugs I encountered back then are now being submitted for reimbursement listing. The drug information, reimbursement criteria, and guidelines I referred to while handling information management tasks have been very helpful.Choi: I worked at a pharmacy until just before joining the agency, so there wasn’t much direct overlap with my current duties. However, having studied pharmacy for many years, my background knowledge helps when exploring new areas.Assistant Director Jae-young ChoiHas your perception of HIRA changed since joining?Song: At first, I worried that the rigid organizational culture might be difficult to adapt to. But after experiencing it firsthand, I found that there is a very horizontal and mutually respectful organizational culture.Choi: It wasn’t until I was about to graduate from pharmacy school that I learned there were pharmacists working at HIRA and that they handled drug management tasks. After joining, I realized that the drug-related work HIRA handles is more diverse than I had imagined.What is the most challenging aspect of your work?Song: Newly launched drugs are expensive, so using them as treatments without reimbursement places a heavy financial burden on patients. We receive many requests for reimbursement, but the challenge is that we cannot simply grant the requests of those who are just waiting for reimbursement.Choi: The fact that I have to continuously study new diseases is both the best part and the most challenging aspect of the job.When do you feel a sense of accomplishment?Song: As part of the New Drug Listing Division’s duties, we review the appropriateness of reimbursement at the DREC. I feel a sense of accomplishment when the reimbursement appropriateness of an item I reviewed is approved.Choi: It’s the moment a new drug I’ve reviewed gets actually listed. I still lack experience and have a lot to learn. Going forward, I want to think more deeply about the social impact as I do my work.Do you have any advice for pharmacy students or pharmacists interested in joining HIRA?Song: While you may be fulfilling various roles in hospitals or pharmacies, there are tasks that can only be performed at HIRA. You’ll likely find a sense of fulfillment and accomplishment here that’s distinct from what you’d experience elsewhere.Choi: Given the nature of our work, I feel we’re doing things that are hard to experience elsewhere. If you’re interested in the public sector beyond clinical practice, I encourage you to apply during our recruitment period.
- Policy
- Anticancer drugs account for record high share of pharma expenditures
- by Jung, Heung-Jun Mar 24, 2026 08:16am
- Claims for anticancer drugs are rising steeply within Korea’s national health insurance pharmaceutical expenditures. It exceeded KRW 3 trillion the year before last, surpassing anti-atherosclerosis drugs for the first time.While total drug expenditures rose by 5.6%, claims for anticancer drugs climbed by 15%, the largest increase by far among the 5 five therapeutic classes in terms of spending.Anticancer drug claims exceed KRW 3 trillionAccording to the 2024 Status of Expenditures on Reimbursed Drugs Report released by the National Health Insurance Service on the 23rd, health insurance drug expenditures totaled KRW 27.6625 trillion, a 5.6% increase from the previous year’s KRW 26.1966 trillion.Drug costs accounted for 23.8% of total healthcare expenditure of KRW 116.2375 trillion. As of 2023, the proportion of drug expenditures in Korea’s current health expenditure was 19.4%, which is 5 percentage points higher than the OECD average of 14.4%.Among major overseas countries (A8) that Korea uses for drug price references, Korea’s spending ratio was also higher than Japan (17.6%), Germany (13.7%), and the UK (9.7%).◆ Anticancer drug claims reach KRW 3.1432 trillion... share hits record high of 11.4%Claims and spending share for anticancer agents reached all-time highs. Claims totaled KRW 3.1432 trillion in the year before last, up 15% from KRW 2.7336 trillion the previous year.The top 5 therapeutic classes accounted for 40.4% of total drug expenditure, with anticancer drugs holding the largest share at 11.4%. Until now, anti-atherosclerotic drugs had previously held the highest claim amount and market share, but anticancer drugs rose to the top spot for the first time.Drug costs for cancer patients totaled KRW 4.2958 trillion, an 11.9% increase from the previous year. This marks the largest growth rate in the past 5 years.◆ By ingredient, “ezetimibe + rosuvastatin” ranked highest…up 16% to exceed KRW 700 billionBy ingredient, spending on ezetimibe + rosuvastatin rose 16.3% year on year, reaching KRW 704.6 billion.Choline alfoscerate came in second at KRW 557.6 billion, down 1% from the previous year. Atorvastatin also declined slightly, with claims of KRW 554.3 billion, down 0.8%. The antiplatelet agent clopidogrel reached KRW 441.8 billion, up 5.7%.◆ Decline in original drug claims...generic share rises to 44.4%Original drug claims showed a declining trend, while generic claims increased. In the year before last, spending on original drugs amounted to KRW 15.3434 trillion, accounting for 55.6% of total pharmaceutical expenditure, down from 59.1% the year before.In contrast, generic drug claims totaled KRW 12.2591 trillion. This accounted for 44.4% of total drug expenditure, an increase from 40.9% the previous year.Since 2021, the spending share of original drugs has continued to decline while that of generics has risen, narrowing the gap in reimbursement claims between the two categories.An NHIS official said, “In line with government policy direction, we will further refine implementation measures and actively support the execution of tasks for the benefit of the public and the development of the pharmaceutical industry, while making every effort to ease patients’ drug cost burdens and ensure the financial sustainability of the National Health Insurance system.”
- Policy
- Will Mifegyne gain momentum for Korean entry?
- by Lee, Jeong-Hwan Mar 24, 2026 08:16am
- The domestic introduction of Mifegyne, the abortion drug whose marketing approval by the Ministry of Food and Drug Safety has been delayed for a long time, is expected to gain momentum.This follows recent remarks by Yong-jin Park, vice chair of the Regulatory Reform Committee, who identified the delay in Mifegyne’s approval as a prime example of irrational administrative regulation and announced his intention to work toward a solution.Vice Chair Park is reportedly determined to ensure domestic approval through administrative action even before legislation is enacted, given that the introduction of Mifegyne is regarded as a matter of national policy.Vice Chair Yong-jin ParkVice Chair Park views the issue of Mifegyne’s introduction from a global standards perspective. He argues that the situation where only Korea is blocking a drug already permitted in over 100 countries worldwide, including OECD nations, solely due to administrative procedures, represents an irrational regulation unique to Korea.Vice Chair Park believes that the reality where women are forced to purchase the drug through underground channels despite its globally proven safety due to administrative barriers threatens women’s health rights and must not be ignored.The MFDS has maintained a cautious stance on approving Mifegyne on the grounds that follow-up legislation has not yet been made since the Constitutional Court’s 2019 decision declaring the abortion law unconstitutional.Vice Chair Park believes that if the MFDS takes a more forward-looking approach to the regulatory barriers it has tied itself to, a solution can be found.This effectively suggests the need to expedite the timing of domestic approval and introduction through proactive government action, even before the legislature completes the relevant process.Attention is now focused on whether the drug could be approved before the National Assembly passes a proposed revision to the Mother and Child Health Act, introduced by Democratic Party of Korea lawmaker In-soon Nam and others, that would allow medication-induced abortion.If the MFDS adopts administrative regulations or establishes a separate approval process, the official introduction of Mifegyne in Korea will gain momentum.An industry official stated, “If Vice Chairman Park’s plan to ease administrative regulations is effectively implemented, the MFDS will be able to exercise proactive administration even amid a legislative vacuum. It would become a representative example of the government putting forward a preemptive alternative at the administrative level.”
- Policy
- Afilivu’s price cut and lists PFS formulation to chase Eylea
- by Jung, Heung-Jun Mar 24, 2026 08:16am
- Samsung Bioepis is stepping up its pursuit of Eylea by lowering the price of its macular degeneration treatment Afilivu and expanding into the prefilled syringe (PFS) segment.Next month, Afilivu Prefilled Syringe 6.6 mg/0.165 mL (aflibercept) will be listed for reimbursement, and the price of the existing Afilivu injection will be voluntarily cut to KRW 198,000, matching the market’s lowest price.According to industry sources on the 23rd, Samsung Bioepis is simultaneously pursuing both a price reduction and the addition of a new formulation to strengthen Afilivu’s competitiveness.Samsung Biologics expands Aflilivu's competitivity by introducing a PFS version with reimbursement. AI-generated imageThe ceiling price of Afilivu 40 mg, previously KRW 298,000, will be voluntarily reduced by KRW 100,000 to KRW 198,000.This matches the lowest price set by Sam Chun Dang Pharm’s Eylea biosimilar ‘Vgenfli Inj.’The Afilivu PFS formulation entering reimbursement this time has also been listed at the same price of KRW 198,000. Not only Eylea, but also Celltrion’s Eydenzelt and Sam Chun Dang Pharm’s Vgenfli already have PFS formulations in their reimbursement lineups.Celltrion also expanded its prescription lineup by listing the PFS formulation last November. Although the Afilivu PFS formulation is a later entrant compared to these, it appears the company will launch a full-scale market offensive by lowering its price.Sales of Afilivu in Korea were suspended early last year due to a preliminary injunction filed by the original developer, Regeneron, seeking to halt sales. However, after the injunction was overturned in December of the same year, domestic distribution and sales resumed. Samil Pharmaceutical holds the exclusive domestic sales rights.It is said to have shown a rapid recovery in sales since resuming distribution. Samil is expected to continue expanding prescriptions through sales activities that now include the PFS formulation.The domestic Eylea market is estimated at about KRW 100 billion. Samsung Bioepis, Celltrion, and Sam Chun Dang Pharm are all closely chasing Eylea and competing to expand market share.In particular, fierce competition is expected between Samsung Bioepis and Sam Chun Dang Pharm, as they are clashing with a lowest-price strategy.
- Policy
- Menarini withdraws approval for angina drug Ranexa
- by Lee, Tak-Sun Mar 24, 2026 08:16am
- ‘Ranexa (ranolazine), a new angina treatment with a novel mechanism of action that had garnered high expectations as the first of its kind in over 20 years, has given up on entering the Korean market five years after its local approval.Used as a second-line treatment for angina, the drug had remained approved but never been listed for reimbursement. As a result, it appears that the company decided to withdraw the product once the re-examination period expired.According to the Ministry of Food and Drug Safety, Menarini Korea voluntarily withdrew marketing authorization for three strengths of Ranexa extended-release tablets—375 mg, 500 mg, and 750 mg, as of the 23rd. Having obtained domestic approval in March 2020, the drug never made it to market and has now disappeared from the regulatory records after five years.Unlike existing beta-blockers or calcium channel blockers (CCBs), Ranexa garnered attention for its unique mechanism of action, which selectively inhibits sodium channels within cardiac muscle cells. Because it does not significantly affect blood pressure or heart rate, it has been considered an alternative for patients whose symptoms are difficult to control with existing medications.However, it never made it onto the national health insurance reimbursement list after approval. To make matters worse, the re-examination period granted at the time of approval reportedly expired on the 15th.Although three months remained before the application period for re-examination closed, the company appears to have chosen to withdraw the approval rather than pursue re-examination. Since no actual sales took place after approval, it is assumed that conducting a re-examination would also have been difficult.The drug was approved by the US FDA in January 2006. At the time, it was the first new angina treatment to gain approval in the United States in more than 20 years, drawing considerable attention.While it was approved in the US. as a first-line treatment for chronic angina, it was approved as a second-line treatment when it received approval in Europe. On July 9, 2008, the European Medicines Agency (EMA) approved it as an add-on therapy for first-line stable angina treatment.When it received domestic approval on March 16, 2020, the indications were also determined based on the European approval. Menarini Korea conducted bridging studies and other procedures to obtain domestic approval for this drug, ultimately receiving approval one year and eight months after filing.Low market potential is cited as the reason for withdrawing from the domestic market. With existing angina treatments already priced low, it is believed that Ranexa, as a second-line therapy, also faced an environment in which it would be difficult to secure a sufficiently high reimbursement price.Currently, there are no generics containing the same active ingredient available. Consequently, medications containing ranolazine are no longer available in Korea.An industry official said, “When a new drug gives up entering the Korean market because of marketability issues, it ultimately only lowers treatment accessibility for patients. When a new drug is withdrawn, it is necessary to closely examine whether there are any generics with the same ingredient or alternative medicines available.”
- Company
- Evrysdi reimb expansion will improve SMA treatment landscape
- by Son, Hyung Min Mar 24, 2026 08:16am
- The expansion of reimbursement criteria for the spinal muscular atrophy (SMA) treatment Evrysdi is expected to significantly improve treatment convenience for SMA patients in Korea.Experts note that the introduction of the tablet formulation, the extension of the prescription period, and the allowance for switching between therapies have reduced the burden of long-term therapy and increased the sustainability of treatment in daily life.On the 23rd, Roche Korea held a press conference at its headquarters in Gangnam-gu, Seoul, to commemorate the launch of the tablet formulation of its SMA treatment Evrysdi (risdiplam) and the expansion of the drug’s reimbursement criteria.Evrysdi is the only oral treatment among SMA therapies and is regarded as a drug that has brought about a shift in the treatment landscape, which was previously dominated by injectable formulations. This contrasts with Biogen’s Spinraza (nusinersen) and Novartis’s Zolgensma (onasemnogene abeparvovec), both of which are injectable therapies.Since its first approval in Korea in 2020 as a dry syrup formulation and subsequent reimbursement in 2023, a tablet formulation was later approved. Starting this March, the tablet formulation became eligible for reimbursement, and the existing reimbursement criteria were expanded.The key elements of the revised reimbursement criteria include: ▲ allowing one-time bidirectional substitution with injectable therapies ▲ extending the maximum prescription period to approximately 2 months ▲ and refining assessment tools to better reflect the patient’s condition.Previously, prescriptions for the dry syrup were limited to 3 bottles (a 36-day supply), effectively allowing only monthly prescriptions; however, following the revision, the reimbursement criteria have been expanded to a maximum of 5 bottles (a 64-day supply). Prescriptions for tablet formulations can now also be issued for up to 2 packs (56 days’ supply), helping to reduce the treatment burden on patients and their caregivers.Jong-Hee Chae, Professor of Pediatrics at Seoul National University HospitalIn addition, whereas previously only a one-time switch from Spinraza to Evrysdi was allowed, the revision now permits bidirectional switching, greatly expanding flexibility in treatment strategy.Jong-Hee Chae, Professor of Pediatrics at Seoul National University Hospital (Chair, Korean Child Neurology Society), said, “In the past, once a treatment was selected, it was difficult to change, but now we can apply more flexible strategies based on the patient’s age, environment, and treatment response.”She added, “With the assessment tools now more finely subdivided, it has also become possible to evaluate treatment effects more precisely in a way that reflects each patient’s actual condition.”SMA is a rare genetic disorder characterized by the gradual loss of motor neurons due to a deficiency of the survival motor neuron (SMN) protein, affecting respiration, swallowing, and overall motor function. It is classified into Types 1 through 4 based on the age of onset and functional level, with Type 1 being a particularly severe form with high mortality if untreated.Professor Chae said, “Because SMA is a disease that requires long-term treatment, it is crucial to ensure the sustainability of treatment in daily life. The introduction of tablets that can be stored at room temperature and the extension of the prescription period represent meaningful changes in improving the quality of life for patients and their families.”Long-term effects confirmed through real-world dataEvrysdi has also shown sustained effectiveness in real-world data (RWD).This drug is a small molecule that modulates SMN2 gene splicing, and it works by crossing the blood-brain barrier and increasing production of the SMN protein throughout the body, including the central nervous system.Hyungjun Park, Professor of Neurology at Gangnam Severance HospitalConsistent efficacy has been confirmed across 4 global clinical trials, ranging from infants before symptom onset to patients with prior treatment experience. Notably, in Types 1 and 2/3 patients, long-term data spanning over 5 years demonstrated the maintenance of motor function.Similar results were also confirmed in real-world data (RWD) recently published in Europe. In a study of patients in the Czech Republic and Slovakia, early improvements in motor function were observed even in severely affected patients, along with the maintenance of respiratory and motor function for up to 3 years.In another Croatian study, patients who switched from an existing injectable therapy to Evrysdi showed motor function improvement over 12 months comparable to prior treatment, confirming non-inferiority.Hyungjun Park, Professor of Neurology at Gangnam Severance Hospital, said, “Recent real-world data from Europe show that Evrysdi exhibits a pattern of significant functional improvement within the first 6 months of treatment, followed by stable maintenance over the long term. In particular, improvements and maintenance of motor function were confirmed in patients with SMA Types 2 and 3, and results showing stable maintenance without functional decline were also confirmed in Type 1 patients.”He further emphasized, “Given the natural course of SMA, which is characterized by continued functional deterioration, this represents a meaningful change. Because consistent efficacy and long-term durability have been confirmed across a wide range of patient groups, the drug has strong practical value in real-world clinical practice.”
- Policy
- CKD and Samjin win approval for 3mg donepezil 3 mg
- by Lee, Tak-Sun Mar 23, 2026 08:42am
- Leading generic manufacturers of the long-standing best-selling dementia treatment “Donepezil” are strengthening their product lineups by introducing a new 3mg low-dose formulation.Attention is focused on whether this will change a market structure that has so far centered on 5 mg and 10 mg products.According to the Ministry of Food and Drug Safety, following the approval of Chong Kun Dang’s “Neuropezil Tab 3mg” on the 18th, Samjin Pharmaceutical’s “Neutoin Tab 3mg” received marketing authorization two days later on the 20th.Neuropezil and Neutoin are leading donepezil generics that have recorded outpatient prescription sales of KRW 12.9 billion and KRW 5.2 billion, respectively, last year, based on Ubest data.The company that first opened the 3 mg donepezil market was Hyundai Pharm. On June 28, 2023, Hyundai became the first in Korea to win approval for ‘Hipezil 3 mg’, taking the lead in the low-dose market. Later, on February 27 last year, Myungin Pharmaceutical obtained the first generic approval with ‘Silvercept 3 mg.’ Silvercept also recorded KRW 5.3 billion in outpatient prescriptions last year, ranking relatively high among generics.At present, even Aricept, the original donepezil brand, does not have a 3 mg lineup in Korea. However, the fact that 3 mg products are already marketed and widely used in clinical practice in neighboring Japan became a key driver for Korean companies’ development efforts.The reason the pharmaceutical industry is focusing on the 3mg strength is safety. While donepezil is highly effective in alleviating dementia symptoms, a considerable number of patients report gastrointestinal side effects such as nausea, vomiting, and diarrhea during the initial stages of treatment.Korean companies, including Hyundai Pharm, developed 3 mg products so that the initial dose could be set at 3 mg for patients at high risk of gastrointestinal side effects or for elderly patients. During the approval process, two studies published in SCI-level academic journals were submitted as clinical evidence, and Japanese marketing authorization information was also used as reference material.An industry official stated, “Donepezil 3mg will serve as a useful alternative for high-risk patients who had to discontinue drug therapy due to side effects. With major pharmaceutical companies such as Chong Kun Dang and Samjin Pharmaceutical joining the field, the proportion of low-dose prescriptions in the dementia treatment market is expected to gradually increase in the future.”
- Company
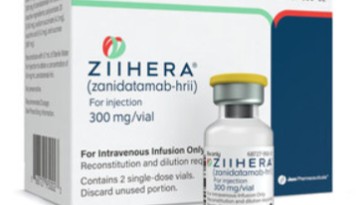
- Bispecific Ab for biliary tract cancer wins nod in KOR
- by Son, Hyung Min Mar 23, 2026 08:42am
- A targeted therapy with a new mechanism of action has emerged in the treatment landscape for biliary tract cancer. There is a strong trend moving away from the existing chemotherapy-centered approach toward biomarker-based precision medicine strategies.Biliary tract cancer treatment 'Ziihera'On the 19th, the Ministry of Food and Drug Safety (MFDS) approved BeOne Medicines' 'Ziihera (zanidatamab),' a treatment for human epidermal growth factor receptor 2 (HER2)-positive biliary tract cancer.Ziihera was approved as a monotherapy for adult patients with unresectable or metastatic HER2-positive (IHC 3+) biliary tract cancer who have previously received at least one systemic therapy.It is expected to become a new treatment option for a patient group that has had limited choices following previous therapies.Ziihera is a bispecific antibody that simultaneously binds to two distinct sites (ECD2, ECD4) of the HER2 receptor.The mechanistic characteristic of Ziihera is inducing tumor cell death by more potently inhibiting HER2 signaling while simultaneously activating various immune mechanisms such as antibody-dependent cellular cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), and antibody-dependent cellular phagocytosis (ADCP).Ziihera is a new drug developed by the Canadian biopharmaceutical company Zymeworks. Subsequently, the U.S. company Jazz Pharmaceuticals licensed the development and commercialization rights for the substance from Zymeworks, and, under the contract, BeOne Medicines holds the commercialization rights in Asian regions, including Korea and China, excluding Japan.This approval is highly significant, as it is the first bispecific antibody approved for biliary tract cancer. Analysis suggests that the treatment strategy targeting HER2 has become an option in clinical settings.The approval was based on the results of the global Phase 2b 'HERIZON-BTC-01' study. According to the clinical results, the confirmed objective response rate (cORR) based on independent central review (BICR) in the HER2-positive IHC 3+ patient group (62 patients) was 52%.The complete response (CR) was 3%, and the partial response (PR) was 48%.The median duration of response (DOR) was 14.9 months, and among responding patients, 59% maintained the response for 6 months or longer, while 44% maintained it for 12 months or longer. In the overall HER2-positive patient group, the ORR was 41%, and the median overall survival (OS) was 15.5 months.Regarding safety, the most commonly reported adverse events were diarrhea, infusion-related reactions, and anemia. Serious adverse events occurred in 47.7% of the subjects.Biliary tract cancer, low survival rates·treatment gaps... will the shift to targeted therapy accelerate?Biliary tract cancer occurs in the bile ducts through which bile is produced in the liver, and since early symptoms are not clear, a significant number of patients are diagnosed at an advanced stage. Because of this, it is considered a primary intractable cancer with a poor prognosis.According to the South Korea Central Cancer Registry, the number of patients with biliary tract cancer in Korea increased by approximately 40%, from 5,444 in 2011 to 7,617 in 2021. Although the patient population is relatively small, treatment outcomes remain limited due to the difficulty of early diagnosis, rapid metastasis to surrounding organs, and high recurrence rates characteristic of the disease.Targeted therapy for biliary tract cancer 'Tibsovo 'In fact, the 5-year relative survival rate (2017–2021) is only 28.9%, with more than 7 out of 10 patients reported to die.Particularly in patients with unresectable locally advanced or metastatic biliary tract cancer, the limited treatment options available after the failure of first-line therapy have been pointed out as the largest unmet medical need in clinical settings.Amid these treatment gaps, targeted therapy-based strategies have been rapidly expanding. Representative examples include Handok’s targeted therapies 'Pemazyre (pemigatinib)' for patients with FGFR2 fusion or rearrangement, and 'Tibsovo (ivosidenib)' for patients with IDH1 mutations.FGFR genetic abnormalities are known to be involved in tumor cell proliferation, survival, angiogenesis, and drug resistance, while IDH1 mutations are reported with relatively high frequency in intrahepatic cholangiocarcinoma.In fact, Tibsovo, which targets IDH1, improved progression-free survival (PFS) in the global Phase 3 ClarIDHy study, establishing itself as the first targeted therapy to succeed in a Phase 3 clinical trial in the biliary tract cancer field. However, unlike Pemazyre, there has been no news regarding reimbursement for Tibsovo since its domestic authorization in 2024.As treatments targeting specific genetic mutations are introduced one after another, the addition of a HER2-targeted treatment strategy is shifting biliary tract cancer treatment toward a biomarker-based, multi-layered approach.Furthermore, additional bispecific antibodies are awaiting commercialization. The bispecific antibody candidate 'tovesimig (HDB001A)', being developed by Compass Therapeutics in the U.S., recently achieved efficacy in the topline results of its global Phase 2/3 study.Tovesimig is a new biliary tract cancer drug candidate developed by the Korean company ABL Bio. Handok holds the domestic rights, and Compass holds the global rights. This drug candidate is a bispecific antibody that simultaneously targets delta-like ligand 4 (DLL4) and vascular endothelial growth factor (VEGF), both of which promote the formation of new blood vessels in the tumor microenvironment.According to the topline results, the ORR set as the primary endpoint was 17.1% for the tovesimig + Paclitaxel group, which was higher than the 5.3% of the Paclitaxel group. Additionally, progressive disease (PD) was 16.2% in patients co-administered tovesimig + Paclitaxel, whereas it was 42.1% in patients administered Paclitaxel alone.
- Company
- Diabetes drug 'Ozempic' prescriptions available at general hospitals
- by Eo, Yun-Ho Mar 23, 2026 08:42am
- 'Ozempic,' a diabetes treatment version of the obesity drug Wegovy, is entering the general hospital prescription areas.According to industry sources, Novo Nordisk Korea's GLP-1 receptor agonist (GLP-1 RA) Ozempic has passed the drug committees (DC) of 'Big 5' tertiary general hospitals, including Samsung Medical Center, Seoul National University Hospital, Seoul Asan Medical Center, and Sinchon Severance Hospital.Since it was added to the insurance reimbursement list in February, Ozempic has continued to expand prescription areas.Ozempic can be prescribed as part of a triple therapy including metformin and sulfonylurea (SU), or in combination with insulin. However, unlike existing GLP-1 analogs, the reimbursement criteria have been restricted to 'Type 2 diabetes patients whose glycated hemoglobin (HbA1C) levels remain at 7% or higher despite at least 2 to 4 months of combination drug therapy.'While analysis suggests that this is Ministry of Health and Welfare's measure to prevent misuse or abuse of Ozempic, some critics argue it limits patient access. It remains to be seen how Ozempic, with many limitations, will perform in the diabetes treatment market.Jang Won Son, a professor of the Department of Internal Medicine at Bucheon St. Mary's Hospital, stated, "Since Ozempic's domestic reimbursement criteria have some partial limitations, compared with the current guidelines, proactive discussions regarding flexible application that comprehensively consider potential complications are needed. Whether an initial measure that excessively considers misuses can be reassessed later is important."Meanwhile, Ozempic demonstrated superior blood sugar-lowering and weight loss effects compared to various diabetes medications, including DPP-4 inhibitor Januvia (sitagliptin), the exendin-4-based GLP-1 RA Byetta (exenatide), and the GLP-1 RA Trulicity (dulaglutide), through six Phase 3 studies (SUSTAIN 1–5 and 7). Notably, Ozempic showed superior blood sugar-lowering effects compared to insulin while maintaining a lower risk of hypoglycemia.The 'SUSTAIN 9' study confirmed additional blood sugar and weight reduction effects in Type 2 diabetes patients who did not achieve sufficient control following treatment with SGLT-2 inhibitors, a leading class of oral diabetes medications. In the 'SUSTAIN 6' Phase 3 trial, the drug reduced the risk of major adverse cardiovascular events (MACE) by 26% in adult patients with Type 2 diabetes and high cardiovascular risk.
- Policy
- Tightened rebate rules pressure innovative pharma certifications
- by Lee, Jeong-Hwan Mar 23, 2026 08:42am
- As the Ministry of Health and Welfare designs a reform of the innovative pharmaceutical company certification criteria, the pharmaceutical industry is raising concerns that the revised framework must include a system capable of reflecting companies’ self-regulatory efforts in compliance management, such as the prohibition of illegal rebates.The argument is that the reform plan must explicitly include provisions allowing the certification review process to consider a corporation’s efforts to prohibit rebates when illegal rebates are provided or detected due to individual misconduct by sales representatives, despite thorough management and supervision by the company itself.The logic behind this is that incorporating such provisions into the reform plan would prevent cases where certification is unfairly revoked due to rebates in which the corporation was not involved, while also creating incentives for pharmaceutical companies to operate internal compliance systems and lead self-policing efforts against rebates.According to the pharmaceutical industry and the ministry on the 20th, an amendment to the administrative notice improving the certification review criteria for Innovative Pharmaceutical Companies will soon be released for public comment.The MOHW plans to incorporate a revised statute of limitations for rebates, which would exclude illegal rebates occurring “more than five years prior” to the date of certification or recertification following the implementation of the reform plan.This is intended to encourage pharmaceutical companies’ commitment to developing innovative new drugs by not linking the revocation of Innovative Pharmaceutical Company certification to illegal rebates that occurred too long ago.However, the pharmaceutical industry argues that, in addition to this five-year rebate exclusion rule, the reform should also contain a mechanism to address the unintended consequence where pharmaceutical companies that have made sufficient efforts to prevent illegal rebates still face unconditional certification revocation, following the exemption for rebates older than five years.In other words, even where a company has established systems for monitoring and managing rebate prohibitions and has worked diligently to maintain compliance, it should be guaranteed the right to present a defense during the certification revocation review process if a rebate occurred due to the misconduct of an individual sales representative.On this, the industry said, “We are not asking for all rebates stemming from individual sales representatives’ misconduct to be automatically excluded from grounds for certification revocation. Rather, we are asking that an administrative track be established to allow for due consideration of the corporation’s efforts toward compliance-oriented management.”The reason for this is that if this provision is not reflected in the reform plan, pharmaceutical companies’ efforts toward compliance management to prohibit rebates will not be reflected at all during certification revocation reviews. This would increase the need to contest the unfairness of certification revocation in court, potentially leading to a significant rise in unnecessary administrative revocation lawsuits between pharmaceutical companies and the MOHW.In particular, pharmaceutical companies point out that the current regulations under review by the MOHW, which hold corporations jointly liable for the personal illegal acts of their internal sales representatives, while specifying that administrative sanctions for illegal rebates by Contract Sales Organizations (CSOs) should be imposed solely on the CSOs without linking them to the corporations, raise concerns that this could encourage pharmaceutical companies to abandon their own sales organizations and shift to CSO-based sales operations.In addition, as the ministry’s current drug pricing reform proposal moves in the direction of strengthening preferential measures based on whether a company is certified as an innovative company, there is concern that the management damage to companies whose certification is unfairly revoked will become far greater than before if no provision is included to reflect compliance management efforts.An industry official said, “If companies that have internal rebate-prevention systems and have made genuine efforts to control illegal conduct are judged under the same certification revocation standard as companies that do not implement compliance management, then the incentive for firms to eliminate illegal practices on their own disappears. There is also concern about the adverse effect that, as a disparity in administrative penalties arises between a pharmaceutical company’s internal sales staff and external CSO sales staff, if pharmaceutical companies decide to reduce the risk of administrative penalties by relying on CSO sales, the risk of circumventing regulations through backdoor rebate practices will increase.”Meanwhile, it appears unlikely that this reform proposal will include a provision to convert the revocation standard for innovative company certification in rebate-related cases into a demerit-point system.