- LOGIN
- MemberShip
- 2026-04-24 05:11:15
- Policy
- Industry calls for more proactive drug pricing incentives
- by Lee, Jeong-Hwan Mar 27, 2026 08:32am
- When asked what should be the top priority administrative action after the government fully implements the drug pricing reform plan in the second half of this year, the pharmaceutical industry immediately responded, “Establishing proper drug pricing incentive policies.”Given that the reform plan places significant emphasis on lowering generic drug prices, the industry is demanding proactive measures to design policies that either clearly favor generics, which have contributed to savings in the National Health Insurance budget, or ensure that no further price cuts are imposed.At the same time, the industry proposed conducting a proper post-evaluation of whether past policies, such as blanket price cuts for generics and differential pricing based on criteria, have actually achieved their intended goals of reducing NHI expenditure and addressing the excessive proliferation of generic products.Following the across-the-board price cuts in 2012, virtually all pharmaceutical companies decided to produce multiple generic products, triggering the first generic drug boom. The proposal calls for a thorough analysis of why a second boom, marked by a further increase in the number of generic drug items, occurred following the 2020 announcement of a tiered pricing system based on requirements for in-house bioequivalence testing and Drug Master File (DMF) registration, in order to set the direction for a new drug pricing system.On the 26th, the pharmaceutical industry criticized that government efforts to establish drug pricing incentives, which are closely tied to domestic companies’ investment in innovative drug R&D, remain significantly insufficient.It also criticized the government for repeatedly introducing price cuts without conducting follow-up administrative measures to evaluate policies in collaboration with the industry, despite the fact that a boom in the number of generics has occurred every time drug prices were revised.Domestic new drugs and generics lack sufficient incentives…“requires proactive administration”Domestic pharmaceutical companies argue that a multidimensional approach to pricing incentives is necessary to achieve the government’s stated goal of shifting toward a new-drug-centered innovative pharmaceutical industry structure.They criticize that the MOHW’s efforts are severely lacking, noting that corresponding preferential regulations are necessary to encourage pharmaceutical companies to develop globally competitive, first-in-class blockbuster drugs with no substitutes.Suggestions followed that it is urgent to design a drug pricing system that comprehensively recognizes the value of domestic pharmaceutical companies, including establishing exclusive preferential regulations for domestic new drugs, creating an exception track for post-listing price reductions for domestic new drugs, and reducing the price reduction rate for generics that have significantly replaced originals and contributed to savings in the national health insurance budget.In essence, they argue that the government must address the criticism that “there are plenty of policies that cut prices, but none that add value.”Criticism that the MOHW focuses solely on price reductions and neglects efforts to establish preferential pricing regulations for domestically developed new drugs persists even in this latest drug pricing reform plan.In the latest reform, the MOHW included provisions to delay or defer price cuts for already-listed generics for innovative and quasi-innovative pharmaceutical companies, and to apply a premium to their reduction rates. It also included preferential provisions for innovative and quasi-innovative companies regarding newly listed drugsNevertheless, the pharmaceutical industry remains steadfast in its position that there are no preferential policies that adequately compensate for the fair value of domestically developed new drugs.This is because, despite the MOHW’s stated intention to restructure the domestic pharmaceutical industry around innovative new drugs through the drug price reform, there are no preferential policies in place for new drugs first developed by Korean pharmaceutical companies.In fact, in response to requests from the National Assembly and the industry to introduce exclusive incentives for domestic new drugs, the MOHW has repeatedly stated that such measures must be carefully reviewed due to potential trade conflicts with countries such as the United States.In contrast, Japan applies innovation and utility premiums to domestically developed drugs with proven clinical value, and also provides a priority introduction premium for new drugs approved for the first time in Japan.The pharmaceutical industry is demanding that if preferential drug pricing (premiums) at the time of listing is realistically impossible, a track be created to grant them exceptional benefits when the post-listing price reduction mechanism is applied. In other words, they are asking that domestic new drugs be excluded from price-volume agreement negotiations, price reductions based on actual transaction prices, and price reductions due to expanded indications, or that the reduction rates be adjusted so that these drugs can derive tangible benefits.Furthermore, they argued that the domestic pharmaceutical industry’s innovation could be supported by recognizing the value of generics, which have a high substitution rate for expensive original drugs, and lowering their price reduction rates, in line with the goal of saving national health insurance finances, which is the very reason for the existence of generics.For generics with usage rates exceeding 80–90% in prescriptions, confirming clear contributions to NHI savings, the industry requested the government to explore proactive measures to lessen price reductions.The National Assembly is also demanding the establishment of a clear differential compensation mechanism for pharmaceutical companies that have demonstrated contributions to the development of the pharmaceutical industry and investments in the value of new drugs, in order to foster the pharmaceutical and biotech industries.Rep. In-soon Nam of the Democratic Party of Korea stated, “The current drug pricing system provides insufficient returns relative to R&D investment. A differentiated pricing system based on factors such as R&D investment ratio, successful new drug development, and global exports is needed, including expanded incentives at listing or exemptions and reductions in price cuts.Lack of post-evaluation in pricing reform also an issueThe pharmaceutical industry also urged the MOHW to reflect on the historical trajectory of Korea’s drug pricing system and jointly set the future direction.Korea established the framework of its pricing system with the introduction of the Positive List System (PLS) in 2006 and the price-volume agreement (PVA), and has since operated the system with incremental adjustments for nearly 20 years.In November 2011, the government reinstated the joint bioequivalence testing system and implemented a blanket price cut in April 2012, applying a generic pricing rate of 53.55%. In 2020, it introduced differential pricing based on direct bioequivalence testing and DMF requirements, along with the 1+3 joint bioequivalence rule.Then, in November last year, the MOHW presented a new reform plan, stating that distortions caused by the proliferation of generics under past policies had reached a point where they could no longer be ignored.The MOHW’s rationale is that the problem has become entrenched, with pharmaceutical companies uniformly launching dozens to hundreds of generics containing the same active ingredient and focusing their efforts on sales and rebate competition for generics rather than innovative drugs, leaving the MOHW with no choice but to intervene.While the domestic industry partially agrees with this assessment, it points out that the root cause of the current multi-product generic structure lies in past government policies on approvals and pricing.In fact, when joint bioequivalence testing and across-the-board drug price cuts were implemented simultaneously in 2012, pharmaceutical companies sought to overcome declining profit margins by rushing into multi-product generic production. This triggered the first generic drug boom, resulting in a significant increase in the number of products despite short-term cost-saving effects.Every time the government implemented reforms to the approval and drug pricing systems, the number of domestic generic drug products increased significantly. (Korea Pharmaceutical and Bio-Pharma Manufacturers Association)According to the MFDS, newly approved generics nearly doubled from 727 in 2012 to 1,283 in 2013, and further increased to 1,684 in 2014 and 1,914 in 2015. The number of reimbursed generics also doubled from 1,094 in 2012 to 1,717 in 2013 and 2,359 in 2015.The 2020 reforms, including differential pricing based on criteria and restrictions on joint bioequivalence (1+3), triggered a second generic boom.At the time of the blanket price cut, the MOHW promoted the policy as a way to elevate Korea’s pharmaceutical industry to a global level. However, in reality, it resulted in the unintended consequence of an explosive increase in the number of generic products.ccordingly, academia and the pharmaceutical industry are calling for post-evaluation and reassessment of the MOHW’s past pricing policies aimed at reducing drug expenditure, curbing generic proliferation, fostering industry development, and promoting innovation.Professor Jong-hyuk Lee of Chung-Ang University College of Pharmacy stated, “It has been 20 years since the introduction of the positive list system. The government’s direction on managing new drug reimbursement and generic pricing is not wrong, but while the goal of addressing excessive multi-product listings is valid, we need to analyze whether the current reform will actually lead to increased R&D investment or reduced NHI expenditure.”Jae-hyun Lee, head of the Pharmaceutical Research Center at Sungkyunkwan University, added, “Both the 2012 blanket price cut and the current reform plan link generics and new drugs. While it is true that the government mobilizes various systems to achieve policy goals, it is necessary to evaluate and reflect on whether these systems were designed and implemented in accordance with their original intentions, and to make revisions accordingly.”Director Lee added, “It is undesirable for policymakers to design a system in a way that expects secondary effects on new drugs through generic drug pricing or aims to achieve other outcomes, such as eliminating rebates as a side effect. If generic drug prices are to be adjusted, the system must remain faithful to its original intent by limiting reductions and surcharges to generics alone. It is contradictory to adopt a half-hearted approach that allows innovative pharmaceutical companies to have their generic drug prices cut by a smaller margin.”
- Policy
- Can cutting generic prices really create new drugs?
- by Lee, Jeong-Hwan Mar 26, 2026 09:29am
- "It is unclear whether the policy objective of this drug price reform plan is to foster the domestic pharmaceutical industry or to rapidly introduce global new drugs from abroad. Will savings generated by lowering generic drug prices actually be used for domestic new drugs and robust domestic pharmaceutical companies, or end up flowing into the high-priced new drugs of multinational pharmaceutical companies? Can the government’s policy, which aims to create domestic pharmaceutical companies capable of producing blockbuster drugs by adjusting generic drug prices, truly be realized?"Although the Ministry of Health and Welfare has proposed a drug pricing reform aimed at shifting the domestic pharmaceutical industry toward innovation-driven new drug development by lowering generic prices and differentiating pricing for newly listed drugs by company, academia is questioning both its credibility and effectiveness.The concern is that if the government repeatedly implements across-the-board price cuts while merely touting the promotion of the pharmaceutical industry and the creation of domestic new drugs without presenting a detailed blueprint for where, how much, and how to allocate limited health insurance funds, it may ultimately result in savings from generics being used to reimburse high-cost global innovative drugs.At the same time, critics argue that the policy paradigm itself is flawed, as it attempts to generate innovative drugs that are rooted in basic and advanced science through partial adjustment of pricing policies for generics, which is a manufacturing-based industry.It is suggested that the MOHW must embark on a shift in its administrative paradigm, considering the need to design a two-track system that separates and distinguishes generic and new drug policies, in order to move closer to a rational policy that minimizes areas of conflict.On the 24th, the public health community criticized the ongoing backlash from the domestic pharmaceutical industry regarding the drug pricing reform plan announced by the MOHW for implementation this year, calling it “the result of an administration that has abandoned predictability.”While the MOHW cited “pharmaceutical industry structural innovation” as the rationale and justification for its drug pricing reform plan, which includes a drastic reduction in the pricing calculation rate for pre-listed generic drugs from 53.55% to the 40% range, the prevailing view among academics is that it is difficult to predict whether this will succeed.“The government’s concept of ‘industry promotion’ lacks clarity in both philosophy and target”Health economists and pharmaceutical pricing experts are questioning whether the MOHW has a clear philosophy for the rational management of drug expenditures within the national health insurance budget, and what the specific direction of its policy goal to foster the pharmaceutical industry actually is.They argue that it is unclear whether the “development of the pharmaceutical industry” mentioned by the MOHW refers to fostering the domestic pharmaceutical industry or to improving patient access through the rapid inclusion of original new drugs, many of which are produced by multinational pharmaceutical companies, for reimbursement in the national health insurance system.They also point out that the MOHW has not clearly defined how much pharmaceutical spending should be allowed within the insurance budget, how that spending should be allocated between prelisted generics and innovative new drugs, or how to address growing demands for coverage of ultra-high-priced drugs.Many scholars argue that tinkering with the drug pricing system in this manner without clarity makes it difficult to set precise policy goals, inevitably leading to the vague and ambiguous slogans of “fostering the pharmaceutical industry” and “reducing health insurance costs.”This is why concerns are being raised that the drug pricing reform plan recently proposed by the MOHW is disproportionately disadvantageous to domestic generic-focused pharmaceutical companies, while favoring multinational companies.Professor Jong-hyuk Lee of Chung-Ang University College of Pharmacy noted that the MOHW’s reform plan failed to align the levels of price reduction regulations imposed on generics and original drugs to a similar degree, despite its stated goal of addressing fairness between domestic and multinational companies.He argued that it is necessary to question whether the ministry is consistently lumping together generic drug regulations, which affect only domestic pharmaceutical companies, and patient access issues, which are directly linked to improved reimbursement rates for multinational companies’ new drugs, under the guise of “fostering the pharmaceutical industry.” The point is to demand transparency regarding the policy philosophy.“The Ministry’s definition of the pharmaceutical industry is vague. Domestic and multinational sectors are being lumped together. In reality, this reform reduces the profits of domestic companies through generic price cuts, potentially reallocating those resources to reimburse multinational companies’ innovative new drugs.”He continued, “While it is not desirable to draw a line between domestic and multinational companies, this drug pricing reform plan contains virtually no provisions that would cause losses for off-patent original drugs, and instead tends to include numerous regulations that are disadvantageous to domestic companies, such as generic price cuts. Even regarding post-listing control, the timing is being consolidated and adjusted, which will likely lead to situations where original drug prices are cut less frequently. Consequently, multinational companies are more likely to reap the actual benefits than domestic firms.”“The government must clearly present its philosophy on how pharmaceutical spending will be allocated and what industry fostering and development concretely means. It’s unclear how much savings will be generated from generic price cuts, and whether those funds will be reinvested in domestic companies or used to expand access to innovative drugs. This is precisely why the industry is calling for joint research with the government, to clarify these uncertainties.”Linking generic drug prices to new drug development the right approach?… time to rethink the administrative paradigm”Some point out that the MOHW’s current administrative approach, which links generic drug pricing policies with the creation of innovative new drugs, is bound to lead to inevitable contradictions.The argument is that generics should be viewed from the perspective of protecting manufacturing, a traditional industry, and public health security, while new drugs should be supported through an environment that fully fosters basic and advanced science; however, linking the two often leads to conflicts.Rather than simply maintaining the current approach, where the MOHW adjusts generic drug prices to encourage more pharmaceutical companies to focus on innovative drug development, the government needs to consider a paradigm shift, such as adopting a two-track administrative system that separates policies for generics and innovative drugs.In essence, the purpose of generic drug pricing is to ensure a stable supply of cost-effective medications to the public within the constraints of limited national health insurance funds. Cost control through price reductions and management, along with the stabilization of quality and supply, are the core elements of the system.Conversely, fostering the development of new drugs hinges on pharmaceutical companies making bold investments in high-risk, high-return clinical trial R&D and strengthening the fundamental capabilities of basic life sciences, such as chemistry, biology, and genetics.Accordingly, the academic community urges the government to establish a nuanced drug pricing system that allows the generic industry to grow, based on its understanding of the value of generics, while simultaneously implementing a separate administrative framework that fully supports the creation of innovative new drugs, independent of the generic sector.Jae-hyun Lee, Director of the Pharmaceutical Regulatory Science Center at Sungkyunkwan University, stated, “The government needs to better understand the role and value of the generic industry. Generic pricing policy should focus on creating an environment where companies can sustainably produce high-quality, affordable generics. Trying to promote innovation through generic pricing is fundamentally contradictory.”Lee explained, “This means the drug pricing system should focus solely on creating an environment where pharmaceutical companies strive to manufacture generics properly. Instead of the current approach of uniformly lowering drug prices, we need a system that differentially adjusts prices by comparing the average price per therapeutic class or ingredient with international averages.He also criticized the reform for lacking predictability from the perspective of pharmaceutical companies.“Government policy must ensure transparency and predictability. Without predictability, companies will resort to workarounds—such as increasing prescription volume to offset price cuts. Japan gradually reduced prices to around 40% over a decade, allowing companies time to adjust. Korea should also adopt a similar phased approach and give companies time to prepare.”Industry voices the government’s lack of understanding on the value of genericsDomestic pharmaceutical companies also argue that the government significantly undervalues the role of generics.They complain that every pricing reform is built on the assumption of across-the-board generic price cuts, inevitably leading to repeated government and industry conflict.They further point out that generics play a critical role in healthcare security and should be actively supported by the government, rather than subject to one-sided cost-cutting policies that fail to adequately consider the difficulties faced by the pharmaceutical industry, including rising manufacturing costs, such as raw materials and labor expenses.An administration focused solely on generic drug regulations, such as price cuts, will ultimately encourage the use of cheap, low-quality raw materials and lead to problems such as the abandonment of production of essential drugs with low profitability, thereby deepening reliance on imported generics.Young-joo Kim, Chair of the Policy Planning Committee for the Emergency Task Force on Drug Price System Reform, stated, “Since generics account for about 50% of South Korea’s pharmaceutical and biotech industry, lowering drug prices will immediately trigger a massive decline in sales. This threatens the survival of pharmaceutical companies. leading to reduced R&D investment due to declining profitability, difficulties in retaining top-tier research talent, and workforce restructuring resulting from production cuts.”“Korea currently manufactures and supplies most generics domestically, which ensures quality control and supply stability while also funding R&D. In contrast, countries that aggressively cut generic prices now rely heavily on imports, leading to various issues such as frequent stockouts, quality control issues leading to fatalities, and supply instability.”A pricing official from a leading domestic pharmaceutical company added, “The government should move beyond abstract slogans like ‘industry advancement’ and properly recognize the value of domestic generics in its pricing policies. There needs to be clear criteria to reward companies that consistently produce and supply high-quality generics, along with a governance system that works in partnership with the industry,” he said.The official concluded, “The negative effects of blanket price cuts are already evident, such as the collapse of the domestic API manufacturing industry. If the government insists on linking generics and innovation, then companies that demonstrate true innovation should be fully exempt from price cuts. Without such changes, conflicts between the industry and the government will persist.”
- Opinion
- [Reporter's View] Advanced cancer patients face Tx gaps
- by Son, Hyung Min Mar 26, 2026 09:29am
- The cancer treatment landscape is shifting rapidly. However, the benefits of this trend are not equally applied to all patients. In particular, patients with advanced cancer repeatedly pushed to the back of the line regarding treatment access, even when viable treatment options exist.The structure is evident in the field of breast cancer. Driven by the expansion of national cancer screening programs, patients with early-stage breast cancer now account for approximately 70% of all cases, many of whom are managed as healthy survivors capable of long-term survival post-treatment. This represents undeniable progress in terms of early diagnosis and clinical outcomes.However, this progress does not translate consistently across all stages of the disease. The issue is not confined solely to metastatic (Stage IV) patients. Limitations in treatment access begin as early as the advanced stages, where the risk of recurrence is high.Disparity is observed in the adjuvant therapy stage. Recently, indications for several therapies were expanded to include adjuvant treatment for early-stage breast cancer, yet reimbursement has failed to keep pace. While regulatory approval has been granted, actual clinical use remains restricted.Adjuvant therapy is designed to suppress recurrence. The key is to preemptively eliminate micrometastases that may remain even after the visible lesion has been surgically removed. Although treatment at this stage can determine long-term prognosis, the current structure makes practical application difficult due to the heavy financial burden on patients.Ultimately, this gap inevitably leads to recurrence. Recurrence is not merely a progression of the disease. Recurrence destabilizes every aspect of a patient's life. As treatment resumes, the burden of medical expenses surges, and constraints on economic activity due to prolonged treatment become unavoidable. The burden of family caregiving also intensifies once again.Given that a significant portion of patients in certain cancer types are in their 40s and 50s, this is not just an individual issue but a cost borne by society as a whole.Despite this, the current system is closer to a model that allocates more resources to managing the aftermath of recurrence than to reducing it. Preventive treatments are restricted due to cost, while the treatment costs and social burdens incurred after recurrence are less considered.Industry experts point to a simple reason. The high volume of patients. As the patient population grows, the fiscal burden increases, ultimately raising the threshold for reimbursement.It does not mean that this issue cannot be disregarded. If the structure of restricting treatment access simply because of high patient numbers persists, the burden will eventually return to the patients and society at large.The problem is that this trend is not limited to a specific cancer type. Adjuvant therapy indications are continuously expanding, not only in breast cancer but also in major solid tumors such as gastric cancer. While treatment strategies to reduce recurrence are evolving rapidly, actual access is failing to keep pace.The issue is not about the availability of treatments. It lies in a structure where access is not granted in a timely manner when treatment is most critical. Therefore, it is time for a more realistic view that sees intervention at the stage of reducing recurrence as a structural necessity rather than a mere cost issue.
- Policy
- Criticism over MFDS’s delayed crackdown on oral albumin
- by Lee, Tak-Sun Mar 26, 2026 09:29am
- Although the Ministry of Food and Drug Safety announced a planned inspection targeting false and exaggerated advertising of ‘oral albumin’ products, the market is criticizing the move as a ‘belated response’ that came only after consumer harm had already occurred.At the time the issue was first raised earlier this year by media outlets and civic groups, sales had already peaked, and illegal advertising intensified further during the Lunar New Year holiday season. Critics argue that the MFDS should have officially announced the inspection earlier through media channels to curb illegal market practices.AI-generated imageMFDS’s “Emergency Response Team for Unfair Food Practices,” which officially launched on the 24th, announced a focused inspection of unfair advertising for albumin-containing foods as its first initiative. Although albumin derived from eggs or milk is broken down into amino acids upon consumption and does not directly increase blood albumin levels, false advertisements have been rampant on home shopping channels and social media, claiming it has the same effects as the pharmaceutical ‘serum albumin.’In particular, as of December last year, albumin products ranked first in the number of home shopping health product broadcasts, and some products sold out more than 10 times, showing explosive sales. Despite being a high-priced product costing around KRW 100,000 per month, it successfully targeted consumers eager to recover from fatigue and boost their immunity.In January, Dailypharm had already published an in-depth report on the harms of oral albumin supplements, followed by similar media coverage.As the MFDS response remained insufficient despite media reports, civic groups and the medical community called for stronger action. On the 20th, the consumer advocacy group Consumers Together issued a statement calling it “a nationwide fraud that packages ordinary food without proven medical efficacy as if it had therapeutic effects. We ask the MFDS to conduct a full investigation into false advertising and impose strict penalties under a zero-tolerance principle.”Earlier, the Korean Medical Association also raised concerns, stating that “there is no clinical evidence supporting oral albumin.” It further indicated that it is considering referring so-called ‘show doctors’ who appeared in misleading advertisements by exploiting their professional authority, to the Ethics Committee and recommending disciplinary action.“Only acting after the peak?”… industry criticism growsIt was only after the KMA and consumer groups issued strong demands that the MFDS officially announced its first planned inspection of oral albumin products through its Emergency Response Team for Unfair Food Practices. However, the pharmaceutical sector and related industries are questioning the practicality of this planned inspection, as large-scale sales had already taken place during peak seasons such as the Lunar New Year.According to the distribution industry, many companies have already generated substantial profits and entered a discount phase to clear remaining inventory following the crackdown announcement. A pharmacist who requested anonymity criticized, “By the time the MFDS declared enforcement, companies had already sold most of their stock,” calling it “a textbook case of a belated response.” An industry official also pointed out that “At the very least, targeted inspections should have been conducted before the Lunar New Year, when sales peak.”In response to criticism of delayed action, the MFDS stated, “We have continuously conducted crackdowns on misleading advertisements for oral albumin products. The launch of this emergency response team is aimed at tackling overall unfair food practices, and is not a targeted inspection specifically for oral albumin.”However, criticism has emerged that there have been no officially planned inspections regarding false advertising of edible albumin products to date, and that existing crackdowns have relied primarily on online post-hoc monitoring. This suggests that there are limitations in responding to real-time advertisements on home shopping channels or social media.While the MFDS aims to overcome these limitations through the new emergency response team, it is expected to face growing accountability issues over consumer damage that has already occurred.
- Opinion
- [Reporter's View] KIMES calls for policy making
- by Hwang, byoung woo Mar 26, 2026 09:29am
- 'The 41st Korea International Medical & Hospital Equipment Show (KIMES 2026)' unveiled the Korean medical device trend shift.More companies are showcasing Artificial Intelligence (AI), which is now regarded as essential, and the technology is shifting to 'how AI is utilized.'A prominent feature of this year’s exhibition is the presentation of specific visions for the stage after adopting technology. Rather than simply emphasizing AI's accuracy, major companies provided detailed explanations of interoperability with hospital systems (PACS/EMR), the repurposing of accumulated data, and revenue structures based on Software as a Service (SaaS) models.This means that the medical AI industry has moved past the initial stage of debating 'whether to adopt' and has entered a maturity phase, focusing on clinical utility and sustainable business models. However, a brutal reality confirmed at the show is that this technological advancement is not immediately translating into market expansion.While there is a consensus that new technology is necessary in the field, the pace of implementation often falls short of expectations due to cost burdens and institutional limitations.This trend in the industrial field aligns with the direction of the recently launched "Second Stage of the Korea Interagency Medical Device R&D Project."While the first stage focused on establishing a technological foundation and building a pipeline for domestic medical devices, the second stage clearly defines a 'market entry-centered' support structure that covers clinical trials, licensing, and commercialization to ensure these technologies take root in actual medical settings.It is encouraging that government policy and industrial trends are aligned with the same goal. The second stage serves as a 'verification stage' to confirm whether developed technologies can avoid being shelved and instead have their value recognized in the market. However, industry workers remain concerned about the disconnect between each stage.The reason for this concern is that the process leading from technological development to hospital adoption, reimbursement application, and market expansion often fails to connect organically, frequently stopping at individual stages.Ultimately, the success or failure of the domestic medical device industry will depend not on the perfection of the technology itself, but on how the 'connection structure' is designed to allow that technology to flow into the actual market.Industry experts suggest that leaving the integration of technology entirely to the market may take too long and face inherent limitations. They point out that more proactive consideration is needed, especially since the global market is currently competing on a similar level.In fact, the platform strategies and subscription models emphasized at KIMES will inevitably face constraints on expansion unless the reimbursement system and institutional support functions are provided.The industry goals identified at KIMES 2026 and the policy direction of the second interagency project are in alignment. However, a gap in speed and execution remains between these two trends. How effectively this policy-wise and field-wise gap can be bridged will likely be the key variable determining the global competitiveness and market growth rate of Korea's medical devices in the future.It is now time for policy-making that ensures R&D achievements do not remain confined to show booths but lead to outcomes in patient clinics.
- Company
- Takeda’s ‘Takhzyro’ listed for reimbursement
- by Son, Hyung Min Mar 26, 2026 09:28am
- Hereditary angioedema (HAE) treatment strategies are showing signs of shifting from an emergency response–centered approach to long-term prevention. With Takeda’s kallikrein inhibitor ‘Takhzyro’ entering the reimbursement list, changes are expected in the existing treatment paradigm that relied on repeated management of acute attacks.On the 25th, Takeda Pharmaceuticals Korea held a press conference at the Glad Hotel in Yeouido, Seoul, announcing the reimbursement listing and domestic launch of the HAE treatment Takhzyro (lanadelumab).Reimbursement criteria include ▲Cases where, despite receiving the androgen-suppressing agent ‘Danazol’ for six months or more, attacks requiring an average of three or more monthly doses of pirazir (acetate) have occurred in the past six months, ▲ases where Danazol administration is contraindicated or cannot be administered due to side effects, provided that emergency treatment was required an average of three or more times per month during the six months prior to administration of this drug.Professor Kyung-min Ahn, Division of Allergy and Clinical Immunology, Ewha Womans University Seoul HospitalHAE is a rare genetic disorder characterized by recurrent severe swelling of the face, hands, feet, abdomen, and especially the airways, caused by a deficiency or dysfunction of C1 esterase. It is characterized by painful swelling without urticaria or itching, and laryngeal edema can lead to fatal asphyxiation.Takhzyro is a treatment used for the routine prevention of symptoms of hereditary angioedema. This medication works by selectively inhibiting the plasma kallikrein (pKal) enzyme, which triggers bradykinin production, thereby preventing angioedema.The efficacy and safety of Takhzyro were confirmed through Globee’s Phase III HELP study. This study enrolled 125 patients with Type 1 and 2 HAE who experienced an average of 3.7 acute swelling episodes per month.In the trial, the group receiving Takhzyro 300 mg every two weeks showed an 83% reduction in moderate-to-severe acute attacks and an 87% reduction in attacks requiring acute treatment compared to placebo.Additionally, in the HELP OLE extension study, which followed 212 patients for approximately 30 months, an average 87.4% reduction in acute attacks was maintained compared to baseline. No new safety concerns were reported in long-term follow-up.Professor Kyung-min Ahn of the Department of Allergy and Clinical Immunology at Ewha Womans University Seoul Hospital stated, “The reduction in acute angioedema and long-term maintenance of efficacy confirmed in clinical studies demonstrate that Takhzyro is a treatment capable of stably managing the disease.”He added, “In major countries such as the U.S. and Europe, prevention-focused treatments have already become the standard of care. In particular, both emergency and preventive treatments are reimbursed, enabling personalized care. The inclusion of Takzyro in the national health insurance reimbursement system aligns with global treatment strategies.”“Fundamental prevention of HAE is needed”Young Joo Cho, Professor of Allergy and Clinical Immunology, Ewha Womans University Mokdong HospitalHAE treatment is divided into preventive therapy and acute treatment. Danazol, an androgen therapy, has primarily been used for prevention.However, danazol has serious adverse effects in women, including hirsutism, acne, voice changes, and amenorrhea. As a result, regular blood tests (liver function, lipid profile) and ultrasound monitoring are essential.Firazyr is used for acute treatment. Approved in 2018, Firazyr is a subcutaneous injection that blocks the action of bradykinin, which causes vasodilation, and relieves acute swelling within two hours. It has the advantage of allowing patients to self-administer the medication.However, experts agree that in order for patients to maintain a normal life, treatment should shift toward prevention-based therapy.The 2021 guidelines from the World Allergy Organization (WAO) and the European Academy of Allergy and Clinical Immunology (EAACI) clearly define the treatment goal for HAE as “complete disease control and normalization of patients’ lives,” emphasizing the need for long-term preventive therapy.Professor Young Joo Cho of the Department of Allergy and Clinical Immunology at Ewha Womans University Mokdong Hospital explained, “Acute attacks significantly affect patients’ quality of life, including work, leisure, and travel. The reimbursement of Takhzyro represents an important step toward shifting Korea’s HAE treatment strategy from acute management to prevention.”She added, “D Although danazol is inexpensive, it causes side effects that make it virtually unusable for women. Danazol is rarely used overseas. It is regrettable that the criteria for domestic reimbursement for Taczyro require the use of danazol first.”
- Company
- Drug price pressure from US…"K-similars may need to join GENEROUS model"
- by Kim, Jin-Gu Mar 25, 2026 07:21am
- As the U.S. pharmaceutical policies under the Trump administration have been rapidly shifting, suggestions have emerged that South Korean biosimilar companies should consider participating in the CMS (Centers for Medicare & Medicaid Services) new drug pricing model, 'GENEROUS,' as a strategic breakthrough.Professor Dong-chul Seo, Professor Emeritus at Chung-Ang University College of Pharmacy, advised this on the 23rd during a seminar held at the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA) titled 'Changes to Pharmaceutical Policy in the Second Trump Administration and Response Strategies for Domestic Companies in South Korea.'Professor Seo analyzed that the GENEROUS model could offer Korean firms an opportunity amid the unpredictable tariff risks of Trump's second presidency and the pressure for Most Favored Nation (MFN)-based price reductions.The GENEROUS (Generating Cost Reductions for U.S. Medicaid) model is a pilot program designed by CMS to reduce costs for U.S. Medicaid. Scheduled to run for five years starting in 2026, this model's key is the application of MFN pricing based on companies' 'voluntary participation'.Participating companies must provide additional rebates to Medicaid referencing the second-lowest price among eight reference countries (UK, France, Germany, Italy, Canada, Japan, Denmark, and Switzerland). Unlike the Inflation Reduction Act (IRA), which mandates price reductions, this is closer to a negotiation-based model in which companies accept price cuts in exchange for securing status in the nation's public insurance programs.CMS introduced this model to lower patients' out-of-pocket costs by reducing drug prices. CMS calculates the Guaranteed Net Unit Price (GNUP) based on the MFN price. Pharmaceutical companies must meet this standard by adding extra rebates to existing ones. State governments accept the prices negotiated by CMS with manufacturers, and further price-reduction negotiations are restricted.Professor Seo stated, "This system is designed to provide additional rebates to the government if foreign drug prices are lower," adding, "Since it is a voluntary participation method with applications accepted until the 30th of next month, companies can review their participation."Analysis suggests that the GENEROUS model may favor biosimilars, as it secures standing in public insurance on the premise of price reductions.For original drugs, accepting MFN pricing is difficult due to the burden of maintaining global pricing tiers. Conversely, biosimilars, which have already been competing at lower price points, are differentiated by the possibility for additional reductions. Accordingly, this is seen as an opportunity to expand market share in the Medicaid market, where the influence of original drugs is diminishing.There is also the possibility of using this as a tool to counter tariff risks. The Trump administration previously proposed 'tariff exemptions for the next three years' to 16 global pharmaceutical companies that agreed to supply at MFN prices. Other mentioned benefits include the provision of FDA's National Priority Vouchers and the application of 'PreCheck' programs. As the GENEROUS model is also a program predicated on fiscal savings, similar policy incentives may be granted.However, suggestions have been made that precise profit-and-loss calculations are needed, given the inherent unpredictability of the Trump administration's pharmaceutical policies. This means a thorough review is required to see if the volume increase from market share expansion outweighs the price cuts and potential tariff burdens.Professor Seo stated, "It would be beneficial for Korean biosimilar manufacturers to participate in this model and experience the actual price application methods," and suggested, “Since generics and biosimilars are highly price-sensitive and have relatively limited global price-linkage effects, participation in the GENEROUS model can be reviewed."Professor Seo further stated, "Companies must comprehensively consider their Medicaid market dependency and sales proportions, the risk of exposing reference country price in MFN is applied, and the impact of price linkages in other nations," and concluded, "Participation may require systematic management of global price exposure and its subsequent effects.”
- Policy
- Pharma companies may suffer KRW 600B annual loss shock
- by Lee, Jeong-Hwan Mar 25, 2026 07:21am
- The Ministry of Health and Welfare (MOHW)'s 'drug pricing system reform plan' to raise the low-price purchasing incentive rate for hospitals and pharmacies from 20% to 35%. Concerns about pharmaceutical industry shock.The government's 'drug pricing system reform plan' to raise the low-price purchasing incentive rate for hospitals and pharmacies from 20% to 35% is facing criticism. Critics pointed out that it is an administrative move that will increase pharmaceutical companies' losses.The pharmaceutical industry is already enduring approximately KRW 350 billion in annual revenue losses under the 20% incentive system. Concerns are rising that if the government transitions to a market-linked actual transaction price system by expanding the incentive rate to 35%, annual losses will exceed KRW 600 billion, inevitably causing market contraction.Given that the government already implements price cuts for existing generic drugs, analysis suggests that raising the low-price purchasing incentive rate will further worsen pharmaceutical companies' profitability, potentially threatening the production and stable supply of essential medicines.On the 24th, the Ministry of Health and Welfare (MOHW) announced a regulation raising the incentive rate paid to medical institutions and pharmacies for low-priced purchasing from 20% to 35% as part of the actual transaction price-based price reduction method. This reform aims to restructure the current system into a market-linked actual transaction price model.The low-price purchasing incentive is a system in which, if a medical institution or pharmacy purchases a drug from a pharmaceutical company at a price below the government-set insurance ceiling price, a portion of those savings is returned to the institution as an incentive.Under the current 20% rate, if a hospital purchases a drug at KRW 100 below the reference price (ceiling price), it receives a KRW 20 incentive.While systemically designed to improve purchasing efficiency and save the National Health Insurance budget, the system has been criticized for pressuring pharmaceutical companies to reduce prices.The pharmaceutical industry estimates that current losses amount to KRW 350 billion annually under the 20% rate. Based on this, pharmaceutical industry argues that a 35% incentive rate would result in annual drug revenue losses to KRW 600 billion or more.Furthermore, assessments suggest that if the price-reduction mechanism for actual transaction price investigations fails to operate properly despite the 35% rate, pharmaceutical companies could face actual losses exceeding these calculations.Concerns are rising that the overall profit base of the pharmaceutical industry could be destabilized if additional price pressures, such as the increased incentive rate, are added to the Ministry's general policy of cutting prices for listed drugs.The MOHW's proposal to raise the incentive rate (from 20% to 35%) is seen as a measure that strengthens the mechanism for transferring price-cutting pressure to manufacturers. Hospitals and pharmacies will have a greater incentive to purchase drugs at the lowest possible prices to secure larger rewards, which may put pressure on pharmaceutical companies to lower their supply prices.An official from a mid-sized Korean pharmaceutical company stated, "While the official insurance drug price may remain the same, the structure forces actual transaction prices to keep falling. If the incentive rate is raised, hospitals, which have more authority, will demand stronger price cuts. Pharmaceutical companies may face significant burdens."The official added, "The current actual transaction price reduction system and the 20% incentive rate must be maintained so that it can alleviate the excessive burden on the industry," and emphasized, "Raising the incentive rate exacerbates the issue of increased losses, especially when a pharmaceutical company sells more. This may ultimately force certain companies to abandon the stable supply of essential medicines."
- Policy
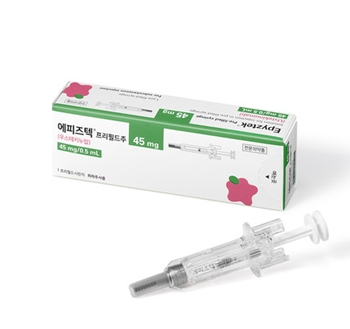
- Stelara biosimilar ‘Epyztek Pen’ listed for reimb next month
- by Jung, Heung-Jun Mar 25, 2026 07:21am
- The pen formulation of Samsung Bioepis’s autoimmune disease treatment Epyztek (ustekinumab) is scheduled to become the first reimbursed product of its ingredient class next month.Samsung Bioepis appears to be pursuing a differentiation strategy with a pen formulation that even the original product, Janssen Korea’s ‘Stelara,’ has neither received approval nor reimbursement for.According to industry sources on the 24th, Samsung Bioepis will expand its reimbursement lineup next month with a prefilled pen formulation of its Stelara biosimilar Epyztek.Like Stelara, Epyztek is an autoimmune treatment indicated for plaque psoriasis, psoriatic arthritis, Crohn’s disease, and ulcerative colitis. In South Korea, it competes in the market alongside Celltrion’s biosimilar ‘Steqeyma’ and others.The pen formulation, scheduled for listing next month, allows patients to administer the medication conveniently and accurately, giving it a competitive edge in self-administration settings.Stelara is currently approved only in intravenous, subcutaneous, and prefilled syringe formulations, making the prefilled pen the first of its kind among products with the same active ingredient. Samsung Bioepis secured its place on the reimbursement list just 3 months after receiving approval from the MFDS last January.This preemptive addition of a new formulation is expected to continue its offensive not only against biosimilar competitors but also in the battle for market share against the original product.Samsung Bioepis is targeting the market with a price lower than the original. The newly listed Epyztek Prefilled Pen 90 mg/1 mL is expected to receive an insurance ceiling price in the KRW 1.27 million range.The original Stelara Prefilled Syringe 45 mg/0.5 mL is priced at KRW 1,745,600, while Epyztek Prefilled Syringe at the same dose is about 30% cheaper at KRW 1,233,376. The pen formulation offers double the dosage but is priced about 27% lower.Leveraging its lower price and the addition of new formulations, the company is expected to begin a full-scale push to expand its market share starting in the second quarter of this year.Samsung Bioepis sells Epyztek (known as Pyzchiva in the U.S. and Europe) not only in Korea but also in Europe and the U.S. through its partner, Sandoz.
- Company
- Imfinzi approved as perioperative gastric cancer therapy in KOR
- by Son, Hyung Min Mar 25, 2026 07:21am
- Immuno-oncology drug ‘Imfinzi’AstraZeneca Korea (CEO Eldana Sauran) announced on the 23rd that Imfinzi (durvalumab) has received approval for perioperative treatment of gastric cancer in Korea.The approved indication is for the treatment of patients with resectable gastric or gastroesophageal junction adenocarcinoma, as perioperative therapy in combination with 5-fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) chemotherapy before and after surgery, followed by Imfinzi monotherapy as adjuvant treatment.Under the approved regimen, Imfinzi is administered in combination with FLOT (5-fluorouracil, leucovorin, oxaliplatin, and docetaxel) for two cycles before surgery, followed by surgery. After surgery, two cycles of Imfinzi plus chemotherapy are administered, and then maintenance treatment with Imfinzi monotherapy is initiated. With this approval, Imfinzi becomes the first immuno-oncology drug approved in Korea for perioperative treatment of gastric cancer.While surgery is the primary treatment for gastric cancer, recurrence rates have historically been high even after surgical intervention. In fact, about 40% to 60% of gastric cancer patients die from recurrence. It is also reported that among patients who experience recurrence after surgery, 50% recur within two years and 90% within five years.This approval of Imfinzi is viewed as a new therapeutic approach that can reduce the risk of recurrence and provide survival benefits as a perioperative therapy for gastric cancer.The approval was based on results from the global Phase III MATTERHORN study in patients with resectable gastric cancer and gastroesophageal junction adenocarcinoma. The MATTERHORN study evaluated the efficacy and safety profile of Imfinzi plus FLOT chemotherapy followed by Imfinzi monotherapy as perioperative treatment, compared with a control arm that received FLOT chemotherapy and surgery alone.Study results confirmed that Imfinzi demonstrated clinical benefits in terms of the primary endpoint, event-free survival (EFS), as well as the secondary endpoints of overall survival (OS) and pathological complete response (pCR) rates.Perioperative Imfinzi reduced the risk of disease progression, recurrence, or death from any cause by 29%, showing a significant improvement in EFS. At 24 months, overall survival was numerically higher in the Imfinzi perioperative treatment group at 75.7%, compared with 70.4% in the control group, although statistical significance was not established. The pCR rate was also approximately 2.7 times higher, at 19.2% in the Imfinzi group versus 7.2% in the control group.The safety profile of perioperative Imfinzi observed in the MATTERHORN study was consistent with the known safety profiles for each individual agent.Professor Keun-wook Lee of the Department of Hematology and Oncology at Seoul National University Bundang Hospital stated, “Gastric cancer is a type of cancer in which recurrence occurs in a substantial number of patients even when treated at a resectable stage. In particular, it has been reported that a significant number of patients with stage II–III gastric cancer experience recurrence despite receiving adjuvant therapy after surgery, rendering a new treatment strategy encompassing both pre- and post-operative settings urgently needed.”He added, “In the MATTERHORN study, clinical benefit in terms of event-free survival and overall survival versus placebo was confirmed, and the pathologic complete response rate was also 2.7 times higher than with standard treatment, making this a clinically significant finding.”Hyun-joo Lee, Executive Director of the Oncology Business Unit at AstraZeneca Korea, said, “Through the MATTERHORN study, we confirmed, for the first time as an immuno-oncology agent, clinical benefit in perioperative treatment for patients with resectable gastric cancer. Since Imfinzi has been approved for the treatment of gastric and gastroesophageal junction adenocarcinoma based on this study, we are deeply honored to be able to provide a meaningful new treatment option to Korean gastric cancer patients, for whom there has been a significant unmet need.”