- LOGIN
- MemberShip
- 2026-05-04 00:25:53
- Company
- Flu drug market enjoys rise for 6 consecutive quarters
- by Chon, Seung-Hyun May 21, 2024 05:56am
- The outpatient prescription market for flu drugs has recovered. Their prescription market, which had remained almost nonexistent for 3 years after the COVID-19 pandemic, has expanded significantly since then. With the flu epidemic lasting more than a year, the market for flu treatments has returned to pre-pandemic levels. According to the market research institution UBIST, the outpatient prescription market for influenza (flu) totaled KRW 6.8 billion in Q1, up 34.8% YoY. The prescription market for flu medications has grown more than 160 times in 2 years from just KRW 40 million in Q1 2022. The flu drug market has undergone significant changes throughout the COVID-19 pandemic and endemic. After recording KRW 8.2 billion in prescriptions in Q1 2020, the flu treatment market was virtually nonexistent until Q3 2022. Its prescription market was below KRW 100 million for 10 consecutive quarters since Q2 2020. This is due to the significant decline in other infectious disease outbreaks since the COVID-19 pandemic began in earnest, due to increased personal hygiene practices such as handwashing and mask-wearing. According to the Korea Disease Control and Prevention Agency, the number of suspected influenza patients per 1,000 outpatients never exceeded 5 until August 2022, since reaching 6.3 in the first week of March 2020, week 9. That means the flu had not even once spread and become an epidemic for 2 and a half years. The prescription market for flu treatments soared to KRW 10.4 billion in Q4 from just KRW 70 million in Q3 2022. On September 16, 2022, a flu epidemic warning was issued for the first time in 2 years and 6 months and then lasted for 2 years and 7 months until April this year. After the COVID-19 pandemic ended last year, the flu epidemic period has been lengthening due to fully lifted social distancing measures and an increase in outdoor activities. In Q4 last year, the amount of outpatient prescriptions for flu drugs exceeded KRW 20 billion. Number of suspected influenza patients per 1000 outpatients (Unit: people, Source: KDCA) In April, the number of suspected flu patients per 1,000 outpatients remained steady at around 10. The prescription market for flu medicines in Q1 of this year is similar to the KRW 6.7 billion in Q1 2019 before the pandemic. The prolonged flu pandemic has brought the market back to pre-pandemic levels. Oseltamivir accounts for most of the outpatient prescriptions in the flu treatment market. Oseltamivir is the main active ingredient in Tamiflu. The prescription market for oseltamivir was not able to surpass KRW 100 million until Q3 2022, after reaching KRW 8.2 billion in Q1 2020. However, it rebounded to KRW 10.4 billion in Q4 2022 and has continued to grow since then. In Q1 this year, oseltamivir-based prescriptions amounted to KRW 6.8 billion, up 34.8% year-on-year.
- Company
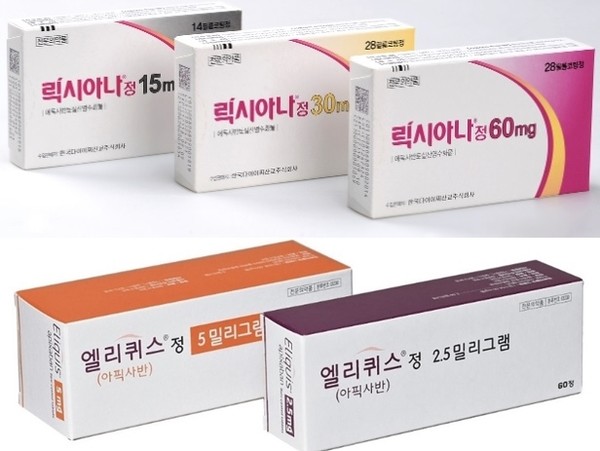
- Series of drug patent expiries change ₩250B DOAC mkt
- by Moon, sung-ho May 21, 2024 05:55am
- The direct oral anti-coagulant (DOAC) market is changing shape at an accelerated pace. This is due to the series of patent expiries of original drugs that had recorded high sales, mainly in the cardiology departments of university hospitals and clinic-level medical centers. #In particular, with the accelerated entry of generic drugs (generics), whether companies of original drugs will be restructuring their business units is also attracting attention. According to industry sources on the 18th, generic versions of original DOACs from major global pharmaceutical companies have recently been launched or announced one after another upon the originals’ patent expiry. DOACs that are currently prescribed in internal medicine hospitals and clinics and form the major market in Korea are Bayer’s Xarelto (rivaroxaban), Boehringer Ingelheim’s Pradaxa (dabigatran), BMS’s Eliquis (apixaban), and Daiichi Sankyo’s Lixiana (edoxaban). Among these, Xarelto’s decline in the prescription market has become more pronounced in recent years with its patent expiry in 2H 2022, and the bulk of domestic generics versions that followed. According to the drug research institution UBIST, Xarelto’s prescriptions plummeted 37% from KRW 49.4 billion in 2022 to KRW 31 billion in 2023. The downward trend has continued in Q1 this year, recording sales of KRW 7.6 billion. In addition, another blockbuster, BMS’s Eliquis, is also set to go off patent in 2H this year. As a result, generic versions of this drug are also expected to flood the market once the patent expires in September, which is expected to affect prescriptions. For reference, Eliquis has continued to dominate the clinical scene, with prescriptions totaling KRW 77.3 billion last year. In Q1 this year, its sales amounted to KRW 19.3 billion, but the general prospect is that this sales flow will change after the launch of its generics. In fact, major domestic companies are already preparing to launch generic versions of Eliquis. For example, Dongkook Pharmaceutical recently received approval from the Ministry of Food and Drug Safety for “Apigaban,” an apixaban generic. These changes are expected to solidify the sole lead of Daiichi Sankyo's Lixiana, which has been strengthening its market dominance in the field. Lixiana, which is marketed by Daewoong Pharmaceutical, has recently surpassed Eliquis in sales and solidified its dominance in the field. Last year, Lixiana's sales in Korea reached KRW 105.3 billion, and in Q1 this year, the drug posted 27.7 billion won in sales, continuing on its upward trend. "The sales of DOACs do not fluctuate much due to their need in clinical practice," said a professor of cardiology at A University Hospital, who requested anonymity. "But if generic versions are released, prescriptions will naturally be dispersed due to drug prices.” He added, "Due to rising interest in its utility after its reimbursement approval following transcatheter aortic valve implantation (TAVI) procedure, and due to the need for its continued use, pharmaceutical companies will essentially seek to own one product. It will be interesting to see how the global pharmaceutical companies that own the original products respond." The industry is watching BMS's moves following the patent expiration of Eliquis. BMS has recently undergone a major restructuring at its headquarters level. Last month, BMS reported Q1 earnings that beat expectations, with sales of multiple myeloma drugs ‘Revlimid’ and ‘Eliquis’ coming in higher than expected. However, one-time costs related to recently closed M&A deals turned the quarter into a loss, and the company said it would focus its resources on R&D programs that will provide the biggest return on investment going forward. In the process, the company announced that it would lay off 2,200 employees this year, discontinue some development programs, consolidate operations, and reduce management layers. In fact, after the BMS headquarters’ announcement, restructuring started in Japan and elsewhere. This is why there is a lot of industry speculation on whether the announcement will affect its Korean subsidiary as well. The series of generic Eliquis drugs that are set to be released upon the original drug’s expiry is also adding strength to the speculation. AstraZeneca also recently withdrew its diabetes drug Forxiga (dapagliflozin) from the Korean market after the launch of its generic versions, closed down the division, and offered voluntary retirement programs. In other words, some worry that global pharmaceutical companies will again launch early retirement programs following the patent expiry of their original drug and the launch of generics. However, BMS Korea denied any such plan. BMS Korea said, "We have no plans set following Eliquis’s patent expiry. We are alternating activities in the domestic market with Pfizer. Our plans at the headquarters level were implemented to strengthen our portfolio through recent M&A and intensive investment in R&D, and are independent of the Korean situation. At this time, we do not have any plans related to Eliquis’s patent expiry."
- Company
- Rolvedon's cumulative US sales surpasses ₩100B
- by Son, Hyung-Min May 21, 2024 05:55am
- Hanmi Pharmaceutical's neutropenia drug Rolvedon is cruising in the US market. Rolvedon has surpassed KRW 100 billion in cumulative sales in the US market. Hanmi Pharmaceutical's U.S. partner Assertio plans to further increase the drug’s market share by securing a same-day dosing indication. According to Assertio on the 18th, Rolvedon generated USD 14.5 million (about KRW 19.7 billion) in sales in Q1 this year, up 31.8% from the USD 11 million it generated in Q4 last year. Since its launch in the US in Q4 2022, Rolvedon has generated USD 80.2 million (KRW 110 billion) in cumulative sales. Rolvedon (Korean brand name: Rolontis) is a neutropenia treatment developed by Hanmi Pharmaceutical. In March 2021, it was approved as the 33rd homegrown new drug in Korea. Hanmi Pharmaceutical and its U.S. partner Spectrum (now Assertio) succeeded in obtaining U.S. Food and Drug Administration (FDA) approval for Rolvedon in September 2022. Quarterly Rolvedon Sales Hanmi Pharmaceutical licensed out Rolvedon to Spectrum Pharmaceuticals in 2012. Assertio acquired Spectrum in April last year and acquired the rights to market and develop Rolvedon and the lung cancer drug poziotinib. Assertio is a pharmaceutical company specializing in inflammation treatments like the non-steroidal anti-inflammatory drug Indocin and the orally disintegrating film Sympazan. After acquiring Rolvedon, Assertio braved the anticancer drug market. Since its release in October 2022, Rolvedon’s sales in the U.S. reached USD 10 million in Q4 sales. In December of the same year, Rolvedon was included in the National Comprehensive Cancer Network (NCCN) guidelines for prevention and treatment options for febrile neutropenia. Since then, Rolvedon's sales have continued to grow, to USD 15.6 million in Q1 and USD 21 million in Q2 last year. However, Rolvedon's growth has slowed since Assertio took over its sales. Rolvedon reported USD 8 million in revenue in Q3 last year, a 62.0% decrease from the previous quarter. Rolvedon has become reimbursable through the US’s public health insurance since April last year, but the terms are reportedly less favorable than the reimbursement system it had in place on its launch. Rolvedon’s sales rebounded in Q4 last year, recording KRW 10 million in revenue. Sales continued to grow in Q1 this year to KRW 14.5 million. Assertio attributed the rebound in sales to the new accounts it had signed using an updated commercialization strategy. Assertio has high hopes for its same-day dosing trial. Rolvedon is indicated for the treatment or prevention of neutropenia in cancer patients who receive myelosuppressive chemotherapy. As a granulocyte colony-stimulating factor (G-CSF) class that stimulates the granulocyte to increase neutrophil production, the drug has a similar mechanism of action with Amgen’s blockbuster drug ‘Neulasta (pegfilgrastim).’ However, neutropenia treatments like Neulasta cannot be administered until 24 hours after chemotherapy, which increases the number of hospitalization days for the patient hospitalized. Assertio is currently enrolling patients in a Phase I, same-day dosing trial with Rolvedon. Assertio CEO Heather Mason said, “Our goal is to have clinical data readout by the end of this year. We expect the new dosing regimen to serve as a differentiated strategy compared with competitors.”
- Company
- Dong-A, Ildong join hands…R&D synergy and resources
- by Chon, Seung-Hyun May 21, 2024 05:55am
- Dong-A ST and Idience sign a strategic equity investment and joint development agreement. Officials, (from left) Ildong Pharmaceutical President Lee Chae-joon, Idience CEO Lee Won-sik, Dong-A ST R&D President Park Jae-hong, and Dong-A ST President Kim Min-youn, taking photos. Dong-A ST and Idience join hands to develop novel anticancer drugs. Dong-A ST invested KRW 25 billion in Ildong’s subsidiary, Idience, and will co-develop novel anticancer drugs. Consequently, Dong-A ST has secured a pipeline of potential novel anticancer drugs, and Idience successfully contracted investment for its novel drug development. Dong-A ST invested KRW 25 billion in Idience…strengthens its novel oncology pipeline According to industry sources on the 20th, Dong-A ST signed a strategic equity investment and joint development agreement with Idience, Ildong’s company specializing in new drug development. Idience is delivering a third-party allocation capital increase of KRW 25 billion by issuing 19,142,420 new shares to Dong-A ST. Prior to the capital increase, Idience had 11,492,538 common shares and 10,985,074 preferred shares outstanding. Dong-A ST is expected to become Idience's second-largest shareholder after the capital increase. Founded in May 2019 as a subsidiary of Ildong Holdings, Ildong Pharmaceutical's holding company, Idience is a biopharmaceutical company specializing in new drug development. Idience is a development-centric (NRDO, No Research Development Only) biotech venture, focusing solely on drug development rather than direct drug discovery. Idience is under development of ‘Venadaparib.‘ Venadaparib is a novel targeted anticancer candidate with a mechanism focused on selective inhibition of Poly ADP-ribose polymerase (PARP), which is involved in cancer generation. Ildong Pharmaceutical developed Venadaparib as an in-house product and then transferred the rights to Idience. It is under development as an oral, targeted anticancer therapy for the treatment of gastric cancer, breast cancer, and ovarian cancer. The analysis indicates that Dong-A ST has strengthened the competitiveness of its oncology pipeline through this investment, obtaining rights for the use of Venadaparib in combination therapies. Dong-A ST continues to build its oncology pipeline by making R&D investments. In November last year, immunotherapy DA-4505 was approved for phase 1/2a trials in South Korea. Furthermore, the company presented preclinical results of SHP1(Src homology phosphatase-1) inhibitor ‘DA-4511’ at the AACR annual meeting 2024, held in April, and proved the candidate product’s potential for immunotherapy for cancer. In December, Dong-A ST invested KRW 31.4 billion in acquiring AbTis, a company specializing in antibody-drug conjugates (ADC). AbTis developed a third-generation ADC linker technology called ‘AbClick,‘ which site-selectively conjugates drugs to antibodies without antibody modification. In the second half of the year, AbTis plans to file an IND for AT-211, a Claudin 18.2 ADC candidate for gastric cancer and pancreatic cancer, in the U.S. and Korea. ”We plan to seek various measures to expand in a global market through this strategic collaboration. We will develop innovative novel anticancer drugs by joining Dong-A ST and Idience technology and products,” Dong-A ST President Kim Min-young said. Idience, secures financial resources for novel drug development…secured KRW 90 billion since its inception Idience has secured an additional financial resource for its novel drug development through Dong-A ST’s investment. Since its inception, Idience has received a total of KRW 65 billion in investment. When Idience was established, Ildong Holdings initially invested KRW 500 million and later added another KRW 4.5 billion. In 2021, Idience successfully secured an investment of KRW 40 billion. It also received a total investment of KRW 40 billion from various firms, including Yuanta Investment, TS Investment, Mirae Asset Capital, and Seoul Investment Partners. In 2022, Idience raised additional investment through a third-party allocation capital increase of KRW 15 billion from Ildong Holdings and others. With the completion of Dong-A ST's capital increase, Idience is set to secure a total of KRW 90 billion in investment capital since its inception. Dong-A ST's cash and cash equivalents in the first quarter amounted to KRW 267.3 billion. With this ample funding, Dong-A ST aims to secure a new drug pipeline while Idience secures funding for drug development. Idience is currently working on the commercialization of Venadaparib, including clinical development for targeting various cancer types, such as gastric cancer, breast cancer, and PARP inhibitor-resistant cancer. In gastric cancer, Venadaparib received orphan drug designation from the U.S. FDA in 2022 and is currently undergoing clinical trials. Earlier this year, at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium, interim results from Phase 1 clinical trials were presented. These results demonstrated a broad range of applications and superior therapeutic effects compared to standard treatments, showcasing Venadaparib's competitive edge. In addition to their previous relationship in the Korean market, Dong-A ST and Ildong Pharmaceutical also built a collaborative partnership for developing novel drugs. Dong-A ST and Ildong Pharmaceutical are co-marketing a natural medicine Motilitone since 2019. Motilitone is a product utilizing natural substances extracted from the seeds of the morning glory flower and the stems of the fucus brown algae. Since 2019, Dong-A ST has been selling the antiulcer drug Gaster, produced by Ildong Pharmaceutical. Gaster is indicated for various conditions, such as gastric and duodenal ulcers, upper gastrointestinal bleeding, reflux esophagitis, Zollinger-Ellison syndrome, acute gastritis, and improvement of gastric mucosal lesions due to acute exacerbation of chronic gastritis. “It is significant to have partnered with Dong-A ST and successfully received investment. Idience’s R&D capacity and pipeline values are recognized through this deal,” Idience CEO Lee Won-sik said.
- Company
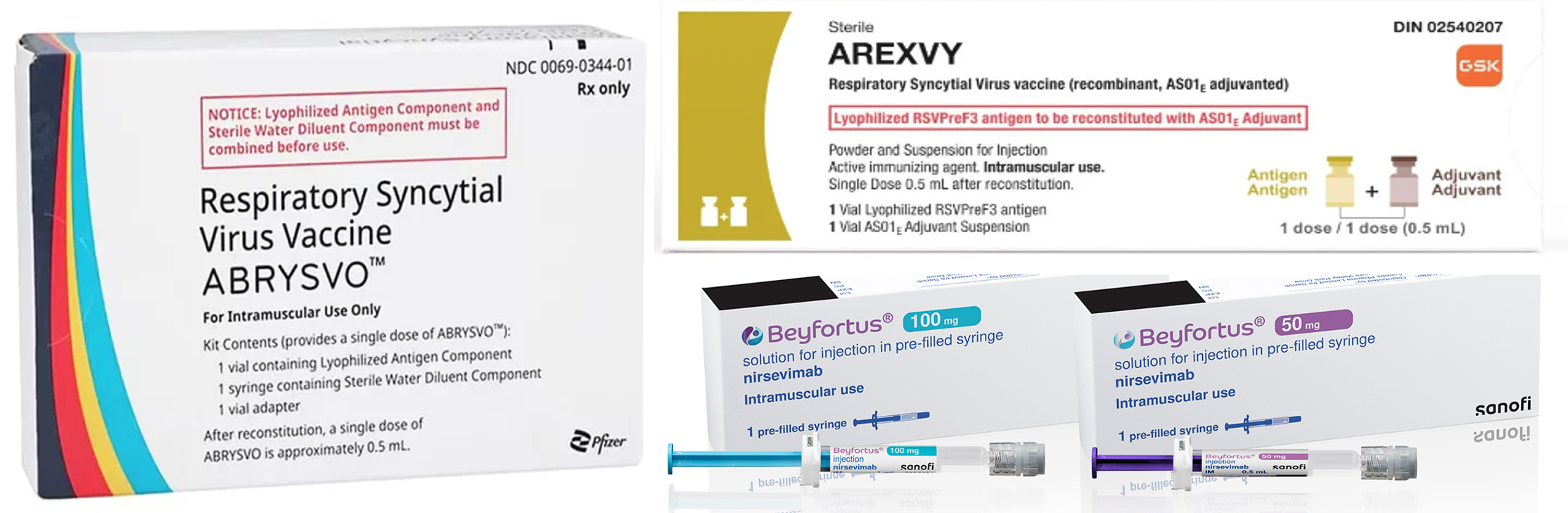
- Global RSV vaccines emerge…K-pharma enters clinical trials
- by Son, Hyung-Min May 20, 2024 05:42am
- The RSV vaccine Beyfortus, developed by Sanofi and AstraZeneca, GSK’s Arexvy, and Pfizer’s Abrysvo. Competition is expected to intensify as new drugs emerge in the respiratory syncytial virus (RSV) market. Sanofi received approval for its RSV vaccine and is the third company to acquire the approval, following GSK and Pfizer. Furthermore, Moderna’s messenger RNA (mRNA) vaccine awaits the FDA approval. In South Korea, EuBiologics has entered the Phase 1 trial. According to industry sources on the 20th, the RSV vaccine Beyfortus, developed by Sanofi and AstraZeneca, received approval in South Korea on April 30th. Consequently, Beyfortus has become the first preventative RSV vaccine targeting young children to obtain domestic approval from the regulatory institution. Last year, GSK’s Arexvy and Pfizer’s Abrysvo were launched overseas, but they have yet to start acquiring domestic approval. RSV is a virus that causes pneumonia and bronchiolitis. Although RSV can be infected at any age, young children are particularly prone to infection. It is the common cause of hospitalization for young children, with a high disease incidence, and 90% of young children worldwide are infected before 2 years. When infected, the symptoms are similar to the common cold. Certain young children experience worsening symptoms, having lower respiratory symptoms such as pneumonia and bronchiolitis. In South Korea, RSV Synagis Inj, an antibody directed to RSV, is available. However, it has the disadvantage of multiple injections due to short-lasting in the body. Moreover, Synagis can only be administered to patients with certain diseases. With the long-lasting Beyfortus, healthy children less than 24 months can be vaccinated. The efficacy and safety of Beyfortus were confirmed in MELODY clinical trials against medically attended Lower Respiratory Tract Infection (LRTI) caused by RSV through 150 days after the treatment. In clinical trials, Beyfortus reduced the RSV LRTI infection rate to 74.5% in full-term infants and late preterm infants. The Beyfortus effect was 70.1% in preterm infants. Furthermore, the NIRSE-Gal interim analysis result, which was the basis for the European real-world evidence (RWE), has shown that the Beyfortus administration reduced the hospitalization rate by 82% in infants less than 6 months compared to non-vaccinated infants. RSV circulates between October and March every year. Sanofi and AstraZeneca are producing Beyfortus before the RSV season and plan to distribute most vaccine stocks before October. Moderna completes clinical trials and waits for FDA approval...EuBiologics enters clinical trials in South Korea as the first and only Moderna’s RSV vaccine will receive the decision for the FDA approval within this month. Moderna’s mRNA-1345 vaccine is developed to prevent RSV in adults over 60. When approved, it will be the third RSV vaccine that targets adults, following Arexvy and Abrysvo. The ConquerRSV trials, which evaluated the safety and efficacy of Moderna’s RSV vaccine, involved about 37,000 adults 60 years or older in 22 countries, including the United States. They were conducted as randomized, double-blind, and placebo-controlled studies. The interim analysis was based on two definitions of RSV-LRTD defined as either two or more symptoms, or three or more symptoms of disease. According to the clinical results, mRNA-1345 demonstrated vaccine efficacy of 83.7% in RSV-LRTD defined as two or more symptoms, and all primary endpoints were met. Adverse reactions of mRNA-1345 were mild or moderate, and the most common adverse reactions reported in the mRNA-1345 group were injection site pain, fatigue, headache, myalgia, and arthralgia. Moderna expects approval this month and plans to launch its first RSV vaccine to the market by the second half of the year. Additionally, EuBiologics has entered full-scale clinical trials in South Korea. EuBiologics’ euRSV vaccine under development has recently started administration in three RSV patients in Korea University Guro Hospital. The clinical trials involve patients randomly assigned to a placebo group, EuRSV low-dose group, or high-dose group, and tolerability and the safety will be evaluated. EuRSV is a recombinant protein subunit vaccine that combines an RSV F protein antigen with immunostimulants. In 2017, EuBiologics acquired immune modulation technology (EuIMT) from KIST. EuBiologics emphasizes that the manufacturing process of EuIMT is simple compared to GSK’s immunostimulant technology, MPL(monophosphoryl lipid). Consequently, the company expects that vaccine mass manufacturing will be possible at a low cost. After starting a phase 1 trial in South Korea, EuBiologics aims to enter global phase 2/3 trials through its U.S. subsidiary EUPOP Life sciences, which has North America and Europe licenses.
- Company
- ‘Much work remains to be done for new drug access in Korea'
- by Eo, Yun-Ho May 20, 2024 05:42am
- Kyung-Eun Bae (Kay Bae ), Chair of the Korean Research-based Pharmaceutical Industry Association Kyung-Eun Bae (Kay Bae, 53) has risen to become the 'center' of the pharmaceutical industry. After being appointed as the general manager to lead subsidiaries including Korea, New Zealand, and Australia of the Sanofi Group, Bae was also appointed Chair of the Korean Research-based Pharmaceutical Industry Association (KRPIA) Being appointed to head the association representing multinational pharmaceutical companies in Korea along with Sanofi Group’s major Asia-Pacific countries, Bae has become a key figure in communicating with the industry and regulatory authorities, including Sanofi headquarters and the Korean government. Bae’s appointment came at the busiest time. Bae has received an offer every time the KRPIA Chair position became open, but she has turned down the offer up to now. And in a situation where her scope of work became broader than ever before, she accepted the long-declined offer. Serving as KRPIA Chair at the busiest time of her career, there had been concerns amid rising expectations about whether Bae would be able to do her part and perform both roles well. As a control tower, KRPIA has been working to make new drugs more affordable than ever before. Making proposals to reform the drug pricing system and engaging in government communications for its support has become a key role of the association. In her two roles, Bae will be overseeing two of Korea’s regular drug pricing reference countries - New Zealand and Australia – at Sanofi, and be determining KRPIA’s key message and strategy on behalf of the industry. In other words, Bae may be best positioned to make the most appropriate policy recommendations. Dailpharm met with Bae to find out more about her new roles. - Your appointment as KRPIA Chair came as a surprise because I thought you were going to turn down the position again. I became the head of Sanofi Australia and New Zealand and the Chair of KRPIA at the same time. I was hesitant at first because I didn't think I had the capability yet. However, I have always felt that I should contribute to the association's development because KRPIA's mission aligns with the values that global pharmaceutical companies deem important. I thought this year was the right time to take on the role. It seems to be that time of life for me when I’m destined to work. So I plan to dedicate myself to the work at hand while taking care of my physical health. I apologize for the unpredictability (laughs). - This is a very important time for multinational pharmaceutical companies. Looking at the government's policy direction, although there are still some policies that add pressure, there are also positive signals, such as the recent plan to compensate for the value of innovative new drugs. The phrase 'new drug value recognition' appeared for the first time in the 'Second Comprehensive National Health Insurance Plan' released by the government this year. It's a good start and an encouraging achievement as it lays the groundwork for improving patient access, including recognizing the innovativeness and value of new drugs and strengthening coverage for seriously ill patients. However, it is important to come up with concrete measures that are effective in practice so that new drugs can be supplied to patients quickly and lead to a virtuous cycle of R&D. -What are the KRPIA's recommendations to the government to address the practical issues you mentioned? There are two main areas I would like to point out. First, Korea is still too late in introducing new drugs. Statistics show that the percentage of new drugs introduced in Korea within 1 year of initial global (US, EU, etc.) approval is only 5%, which is very low compared to Japan and the US. Whether a new drug can actually be used by patients is most important. To address this issue, Korea has a Global Innovative Products on Fast Track (GIFT) system in place. However, we need to check whether the system is really shortening the review period and where the bottlenecks are. I think we first need to come up with a solution for drugs that are in urgent need of introduction. The second is in recognizing the value of new drugs. Companies are motivated when the value of new drugs is recognized, which in turn generates profits and creates a virtuous cycle of R&D. This ultimately requires a significant change in drug pricing policies, including a dramatic improvement in ICER values. When global pharmaceutical companies consider whether to invest in R&D in China, Japan, Southeast Asia, and Singapore, among other countries, they have to prioritize investing in countries with a strong R&D reputation. From an industrial perspective, this is why we need to move to reinforce Korea’s R&D competitiveness. Also, due to limited national health insurance finances, we need to take a look at the expenditure structure. In terms of the proportion of new drugs in drug expenditures, new drugs account for 60 to 70% of all drug expenditures abroad, but they account for less than 10% in Korea. We need to reorganize the inefficient systems that allow polypharmacy and clinic shopping. This way, we can budget for new drugs without putting a strain on insurance finances. -It was also surprising to see KRPIA voice the need to improve Korea’s expenditure structure. structure. Multinational pharmaceutical companies have been cautious in speaking out on the issue because the domestic market still has a strong tendency to look at new drugs as being from multinational companies, and generic drugs from domestic companies. Many were surprised that we released data on Korea’s drug expenditure structure (and the proportion of new drugs) last year. However, the government should be encouraging domestic companies to develop new drugs now. From the PE exemption system to ICER valuations, everything is connected, so KRPIA plans to communicate with the government on the issue whenever there is an opportunity. -It was also unexpected that KRPIA would request the government to disclose the discussions and results of committee reviews (such as those by the Pharmacoeconomic Evaluation Subcommittee) that new drugs undergo in the reimbursement process. Of course, securing the transparency of the reimbursement process may be a cautious matter for companies due to each company’s strategic secrets and so forth. But in the big picture of healthcare, patients are waiting. Information sharing is really important for decision-making. Transparency in that area is a win-win for the companies. I think this is why the government has been responding to many of the requests. -The government's efforts to strengthen post-listing control of drugs are also expected to be in full swing this year. Policies such as those that utilize real-world data (RWD) and switching the pharmacoeconomic evaluation exemption system to a temporary deferral of evaluation system may be of concern and dissatisfaction to KRPIA. I think post-listing control is necessary in part to manage the overall insurance finances. However, Korea’s post-management system is not integrated and is too fragmented, including the PVA (price-volume agreement), which often results in redundant drug price reduction mechanisms From the company's point of view, the system needs to be simplified to be predictable. Currently, the government operates a follow-up system that focuses only on drug prices, such as PVA, which creates a high administrative burden. I would like to see a system that takes into account the structural aspects, such as strengthening health insurance coverage for severe diseases over mild diseases. There seems to be too much focus on just reducing drug prices. Strengthening post-listing control without improving the system and policies for rapid new drug registration in Korea will eventually lead to another decline in access to new drugs. KRPIA is preparing a report on the problems of the post-listing control measures and suggestions for their improvement that we plan to propose to the government based on the views of professionals with extensive experience in the field.
- Policy
- NHIS completes pricing negotiations for Vemlidy, Brukinsa
- by Lee, Tak-Sun May 20, 2024 05:41am
- Negotiations between the National Health Insurance Service (NHIS) and pharmaceutical companies have been completed for 5 morning sickness drugs, hepatitis B drug Vemlidy Tab (tenofovir alafenamide hemifumarate), and Chinese company BeiGene Korea's new cancer drug Brukinsa Cap (zanubrutinib). The 5 morning sickness drugs underwent new drug pricing negotiations, Vemlidy underwent the price-volume agreement negotiations, and Brukinsa underwent a negotiation to expand its scope of use. According to the industry on the 17th, the NHIS recently updated its list of drugs completed negotiations and announced the completion of negotiations of the drugs listed above. The 5 morning sickness drugs that have completed negotiations are Hyundai Pharm’s ‘Diclectin Enteric Coated Tab,’ Boryung Biopharma's ‘Easy Morning Enteric Coated Tab,’ Dong Kook Pharmaceutical's ‘Mommyrectin Enteric Coated Tab,’ Shin Poong Pharm’s ‘Dinerzia Enteric Coated Tab,’ and Hanwha Pharma’s ‘Prelectin Enteric Coated Tab.’ These drugs are pyridoxine hydrochloride and doxylamine succinate combinations and are the only drugs indicated to treat nausea and vomiting during pregnancy in women who do not respond to conservative management in Korea. The government has been pushing for health insurance coverage of these items since last year as part of its policy to support pregnant women. The NHIS has been negotiating drug prices since mid-March and recently reached an agreement with the 5 pharmaceutical companies. Vemlidy has reportedly completed negotiations under ‘Type B’ of the Price-Volume Agreement system. The drug underwent the same type of negotiations last year, which resulted in a 4.7% reduction in its insurance cap price As the most-used drug in the hepatitis B treatment market, Vemlidy’s insurance claims continue to grow. Last year's prescription sales amounted to KRW 61.9 billion based on UBIST, up 26% YoY (from KRW 49.2 billion the previous year). PVA Type B is applied to drugs ▲whose insurance price had been adjusted under ‘Type A,’ or ▲that were listed for over 4 years without undergoing pricing adjustments under Type A and whose claims amount increased over 60% from the previous year; or ▲increased by 10% but the amount exceeds 5 billion. Vemlidy was found to have satisfied the conditions. Brukinsa was approved in May last year as monotherapy for the treatment of adult patients with Waldenström’s macroglobulinemia (WM) who have received at least one prior therapy. In March, the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee recognized the adequacy of expanding reimbursement for Brukinsa as treatment of mantle cell lymphoma (MCL) and chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) and has been in negotiations with the agency ever since. The items that successfully completed negotiations will the NHIS will reported to the Health Insurance Policy Deliberation Committee of the Ministry of Health and Welfare next week for reimbursement listing.
- Company
- Welireg can be prescribed in general hospitals in Korea
- by Eo, Yun-Ho May 20, 2024 05:41am
- Welireg, a rare anticancer drug for a small number of patients, can now be prescribed in general hospitals in Korea. According to industry sources on the 17th, MSD Korea’s oral hypoxia-inducible factor-2 alpha (HIF-2α) inhibitor, ‘Welireg (belzutifan) passed the drug committees (DCs) of various medical centers in Korea including the National Cancer Center, Samsung Medical Center, and Asan Medical Center. Welireg was designated an orphan drug in Korea for the treatment of Von Hippel-Lindau disease in January last year, then formally approved in May of the same year. Specifically, the drug is indicated for the treatment of adult patients with VHL disease who require therapy for associated renal cell carcinoma (RCC), central nervous system (CNS) hemangioblastomas, or pancreatic neuroendocrine tumors (pNET), not requiring immediate surgery. As a HIF-2α inhibitor, Welireg reduces transcription and expression of HIF-2α target genes associated with cellular proliferation, angiogenesis, and tumor growth. The drug’s efficacy was demonstrated in the open-label Study 004 trial, which investigated 61 patients with VHL-associated RCC who were diagnosed with at least one measurable solid tumor localized to the kidney. Patients enrolled in the trial had other VHL-associated tumors including CNS hemangioblastomas and pNET. The major efficacy endpoint of the clinical trial was the overall response rate (ORR) in patients with VHL-associated RCC as measured by radiology assessment using RECIST v1.1 as assessed by an independent review committee (IRC). Additional efficacy endpoints included duration of response (DoR) and time to response (TTR). In the study, Welireg showed an ORR of 49% in patients with VHL-associated RCC. All responses were partial responses. The median DoR had not yet been reached, and the DoR among responders who were still responding after at least 12 months was 56%. Median TTR was 8 months. Also, in patients with VHL-associated CNS hemangioblastomas, Welireg showed an ORR of 63%, with a complete response rate of 4% and a partial response rate of 58%. Meanwhile, Welireg was additionally approved for a kidney cancer indication in the US. Its efficacy for the indication was confirmed in LITESPARK-005, a trial conducted on patients with unresectable locally advanced or metastatic clear cell renal cell carcinoma (RCC) that had progressed following both a PD-1 or PD-L1 checkpoint inhibitor and a VEGF-TKI. Results showed that Welireg improved progression-free survival (PFS) and reduced the risk of disease progression or death by 25% compared to everolimus in patients with advanced renal cell carcinoma who received a PD-1 or PD-L1 immune checkpoint inhibitor and a VEGF receptor-targeted therapy, either sequentially or in combination.
- Policy
- Erwinase price increases… Imfinzi renews RSA
- by Lee, Tak-Sun May 20, 2024 05:41am
- Immunotherapy drug for cancer, Imfinzi inj The price of Erwinase Inj (L-asparaginase), used to treat patients with acute lymphoblastic leukemia (ALL), is expected to increase. Additionally, Imfinzi, an immunotherapy drug for cancer, also succeeded in renegotiating risk-sharing agreements (RSA). Rhopressa, a treatment for open-angle glaucoma and intraocular pressure, has entered negotiations with the National Health Insurance Service (NHIS). According to industry sources on the 16th, the NHIS posted these negotiation results and schedules. Clinigen’s Erwinase inj was first listed in August 2019. The listing took 12 years and 5 months to be granted after the approval in South Korea. It can be covered by reimbursement when used in combination with other chemotherapy agents in patients with acute lymphoblastic leukemia (ALL) hypersensitive to Escherichia coli-derived asparaginase. At the time of reimbursement listing, the price of Erwinase was negotiated for KRW 520,000 per bottle, which was lower than the lowest price among A7 countries (KRW 702,564 (UK)). Erwinase Inj has been in negotiations with the NHIS regarding drug price increases through the adjustment application, and it recently met in agreement. The price increase has not been disclosed. In March, Erwinase Inj was granted for the changes of administration requirements from Grade 3 to Grade 2 from the Cancer Disease Review Committee of the Health Insurance Review and Assessment Service (HIRA). AstraZeneca’s Imfinzi (Durvalumab) had an RSA agreement when it was reimbursement listed in April 2020. The contract ended in March. Despite of the contract ending, it continued negotiations with the NHIS and succeeded recently in negotiations. Imfinzi is covered by reimbursement for the treatment of patients with unresectable non-small cell lung cancer (NSCLC) who have not progressed after platinum-based concurrent chemoradiotherapy (CCRT). It is pursuing reimbursement expansion for bile duct cancer. The NHIS also disclosed pharmaceuticals under negotiations. As reported, Novartis' Ilaris injection (canakinumab) initiated negotiations in May. Ilaris passed the DREC review of the HIRA through real-world evidence (RWE) submission as a pharmacoeconomic evaluation exemption drug, following the principle of post-listing assessment. It is subject to a total cap on the type of reimbursement with RSA. Santen's glaucoma treatment, Rhopressa Ophthalmic Soln, has also entered negotiations. In April, it received conditional approval from DREC for reimbursement if priced below the evaluated amount. Subsequently, the pharmaceutical company has accepted the evaluated amount set by the DREC. Given that most pharmaceutical companies tend to take the pricing set during this period as the basis for price negotiation exemption, it is anticipated that negotiations with the HIRA will focus on expected reimbursement amounts.
- Company
- 'Imfinzi' shown to be effective for liver cancer
- by Son, Hyung-Min May 17, 2024 05:47am
- Hong Jae Chon, Professor of the Department of Hemato-Oncology at CHA Bundang Medical Center Imfinzi, an immunotherapy treatment for cancer, demonstrated effectiveness for liver cancer in addition to bile duct cancer. Consequently, it is expanding its uses in gastrointestinal cancer. AstraZeneca confirmed the effectiveness of Imfinzi in combination therapy with Imjudo, which targets CTL-A4, as a first-line treatment for liver cancer. The medical experts analyzed that this combination therapy has given hope for extended survival in patients with liver cancer. AstraZeneca hosted a press conference at Grand InterContinental Seoul Parnas on May 14th to commemorate the launch of a combination therapy, Imfinzi plus Imjudo. AstraZeneca’s Imfinzi is an immunotherapy for cancer that targets PD-1. It has been launched as a combination therapy with Imjudo, which targets CTL-A4, for the treatment of liver cancer. In June of last year, Imjudo was approved as a combination therapy with Imfinzi for the first-line treatment of liver cancer. Through this launch, Imfinzi has expanded its treatment area in gastrointestinal cancer, including bile duct cancer and liver cancer. Imfinzi was approved as a first-line treatment for bile duct cancer last month. The HIMALAYA Phase 3 trial demonstrated the effectiveness of Imfinzi plus Imjudo combination therapy. The trial compared the efficacy and the safety profile of Imfinzi plus Imjudo combination therapy to Bayer’s Nexavar, enrolling 1,171 patients with unresectable liver cancer who are 18 and over and have no prior treatment history. As part of the treatment regimen, Imfinzi plus Imjudo was administered on day 1 of week 1, followed by Imfinzi monotherapy only for the next 4 weeks. Nexabar was administered twice per day. Clinical results showed that Imfinzi plus Imjudo combination therapy reduced the risk of death by 22% compared to Nexabar monotherapy. Imfinzi plus Imjudo combination therapy’s overall survival (OS) was 16.4 months, whereas that of Nexabar monotherapy was 13.8 months. The difference in OS between Imfinzi plus Imjudo combination therapy and Nexabar monotherapy expanded over time. At week 18 of treatment, the OS of Imfinzi plus Imjudo was 48.7%, and that of Nexabar was 41.5%. The difference in OS was maintained up to 3 years of treatment. For the safety profile, the rate of Grade 3 or Grade 4 treatment-related adverse drug reactions in Imfinzi plus Imjudo combination therapy was 25.8%, and that of monotherapy was 36.9%. The Imfinzi plus Imjudo combination therapy is expected to compete with Roche’s combination therapy of Tecentriq, an immunotherapy for cancer, and Avastin, a targeted anticancer agent. Previously, Tecentriq and Avastin also proved to improve OS compared to Nexabar. Hong Jae Chon, Professor of the Department of Hemato-Oncology at CHA Bundang Medical Center, said, “In addition to Imfinzi, we now have more treatment options for liver cancer with the emergence of Tecentriq combination therapy. The advantage of Imfinzi plus Imjudo combination therapy is reduced risk of bleeding. The risk of bleeding in patients with liver cancer is high due to decreased liver function. Because Imfinzi combination therapy has reduced risk of bleeding, endoscopic surgery is possible right away.” “Patients with liver cancer who have accompanying liver function reduction have a high mortality rate. However, Imfinzi does not worsen the liver function. Based on my experience, treatment outcome is better than expected,” Chon added. “If combination therapy reimbursement is approved, its use will increase. Currently, it will be used without coverage for patients requiring the therapy.”