- LOGIN
- MemberShip
- 2026-05-04 00:25:51
- Policy
- 'BIO KOREA 2024' begins
- by Lee, Hye-Kyung May 10, 2024 05:47am
- 'BIO KOREA 2024' will be held on May 8-May10 for three days at Coex, Seoul. The conference is co-organized by the Korea Health Industry Development Institute (President Soon-do Cha, referred to as KHIDI) and Chungcheongbuk-do Province (Mayor Young Hwan Kim, referred to as Chungbuk), and sponsored by the Ministry of Health and Welfare (MOHW). Celebrating its 19th convention this year, BIO KOREA started back in 2006 with the participation of 300 companies from 20 countries. The number of participants has more than doubled, and by 2024, it reached 600 companies from 50 countries. BIO KOREA became the international bio-health convention representing South Korea. The theme for BIO KOREA 2024 is "The Future of Biotechnology Innovation and Global Collaboration." The convention aims to share the latest trends and prospects of innovative technologies in the bio-health industry and provide opportunities for business collaboration among global companies, institutions, researchers, and investors. Various programs will be organized for three days, including Business Partnering, Invest Fair, Exhibition, and Conference. This year, 640 companies from 55 countries are participating. At Business Partnering, Korean and foreign companies, including global pharmaceutical companies, participate in one-on-one business meetings to discover new partners with promising technologies, discuss technology collaboration and joint research, technology transfer, and investment. Of note, Global companies, including Eli Lilly, Takeda, Merck, MSD (Sharp & Dohme), Sanofi, Roche, Boehringer Ingelheim, and Johnson & Johnson, and companies representing the biopharma industry in South Korea, such as SK Bioscience, ST Pharm, GC Corp, Yuhan, Hanmi Pharmaceutical, Handok, and Dong Wha Pharm, are participating. At Invest Fair, equity firms utilizing K-BIO mega funds and global investors will share the prospects of the bio-health industry in South Korea and present the industry’s potential for growth. Experts from various companies will participate in presentations and panel sessions. The participants include Johnson & Johnson Development Corporation’s Melinda Richter, ARCH Venture Partners’s Ari Nowacek, Teralys Capital’s Cedric Bisson, Kurma Partners’ Daniel Parera, Yuanta Asia Investment’s ChiaYen Lo Sam. A session focusing on the successful attraction of foreign investment cases by domestic bio-innovation startups is also planned. This session aims to enhance understanding of investment value and global collaboration in the Korean biotech industry and explore strategies for commercializing innovative technologies. This year, participation from national delegations has expanded compared to the previous year, with 81 companies from 10 countries, including Australia, Sweden, Denmark, the United Kingdom, the United States, and Germany, set to showcase their promising technologies and engage in exchanges with interested companies in South Korea. Thirty-eight companies from fields such as CDMO, CRO, and microbiome are participating at the Australian Embassy in Seoul booth. Additionally, at the Nordic booth jointly operated by the Swedish Trade and Invest Council and the Danish Embassy in Korea, 13 companies specializing in CRO, vaccines, and drug development are participating. In addition, local companies will participate at the booths of the British Embassy in Seoul, BioCom California in the United States, and the state of Bavaria in Germany to promote their outstanding technologies and engage in business exchanges with domestic companies. At the conference, 71 speakers from 9 countries who are experts in the bio-health industry in South Korea and foreign countries. Speakers will participate in 11 sessions discussing 10 topics, including Drug Discovery Platform, Diabetes-Obesity Treatment, Cancer Vaccine, and Global Open Innovation. On May 9th, a special session called 'New Drug Modality Development Strategies' will be held. In this session, speakers will share the latest strategies for the development of new modality drugs, such as Antibody-Drug Conjugate (ADC), Targeted Protein Degradation (TPD), and Cell and Gene Therapy (CGT). They will also discuss cases of global cooperation. The speakers for the session include Zheru Zhang of WuXiBiologics, Mikio Kasahara of Roche, and Dr. Ulf bethke of Miltenyi Biotec. Following the session, some Korean companies, such as PINOTBIO and AbTis, will give pitch talks. They will showcase promising technologies developed by these Korean companies and discuss development strategies. KHIDI’s President Soon-do Cha said, "We hope that BIO KOREA will provide the opportunity for the domestic and foreign companies, institutions, and researchers in the bio-health industry to converge and discuss business strategies and collaboration.” He added, “We also hope that outstanding technology developed in South Korea will enter the global market through global partnership.” More convention details are available on the BIO KOREA 2024 website (www.biokorea.org). Additionally, people who have not pre-registered but wish to attend the convention can still register at the registration booth set up at Coex during the convention.
- Opinion
- [Reporter’s View] The dark side of the coveted MNC jobs
- by Eo, Yun-Ho May 10, 2024 05:47am
- Multinational pharmaceutical companies have become the wannabe companies for job seekers. With high salaries that are comparable to those of large companies and the flexible working arrangements that allow remote work, generous vacation days, generous in-house benefits, and a horizontal corporate culture, it seems anyone would jump at the chance to join these companies. However, there is a catch. For such a good foreign pharmaceutical company, labor conflicts are not uncommon. Layoffs that accompany various changes, such as divestitures, spin-offs, and reorganizations, are not a big deal in the industry anymore. Some people refer to labor unions in foreign pharmaceuticals as labor aristocracy and elite unionism, but there are definitely conflicts. Although the layoffs are coined differently in each company – including the Early Retirement Program (ERP), and Volunteer Separate Program (VSP) - these retrenchment programs all serve the same purpose and offer a substantial compensation compared to other Korean companies. Some joke that "you must have done well in your previous life to get ERP". For those nearing retirement age and those who can find another job, an ERP from a multinational can be an opportunity. The problem is that despite the compensation, many just feel despair. The medical representatives are affected the most. More recently, also the marketing staff, which were once the crown jewels of the pharmaceutical industry, are now the companies’ No.1 candidate for retirement. The irony is that the current era of new high-priced drugs is what is pressuring these business units. Contrary to how the drugs that the company sells have become better and more expensive, the departments that make the profits are subject to the layoffs. The reason is simple: the drugs that pharmaceutical companies are selling are no longer "competition-based.” Drugs these days are sold as long as they receive reimbursement, so are “reimbursement-based.” There are no longer 4 or 5 new drugs competing for the same mechanism of action for diabetes, hypertension, high cholesterol, etc. This is an era where a petition for reimbursement of a single drug gains 50,000 consents, an era of all-purpose drugs with characteristics as unique as their high price that are expanding indications like an octopus, an era where only 1-2 competitors exist for each condition, rendering the speed of reimbursement key. Market access (MA), Government Affairs (GA), and Patient Advocacy (PA) are the hottest positions right now. And the traditional commercial staff are feeling the pinch. Multinational companies are replacing the names of their sales personnel with terms such as "medical assistant," "expert information providers.” These vague and general terms were coined by the reporter to avoid referring to a specific company. on. They have drastically reduced their numbers and given them new tasks and motivations as literal “supporters,” but this new culture has not settled well. Global Big Pharmas are driven by profit. There's nothing wrong with that, but the workforce that has become irrelevant in the new era has done no wrong either. It remains regrettable that the wannabe companies don't seem to have the foresight to evolve and foster the unique talents of their employees.
- Company
- Dong-A ST will exclusively supply surgical robot Versius
- by Son, Hyung-Min May 10, 2024 05:47am
- Dong-A ST announced on the 8th that it has signed an agreement with CMR Surgical for the exclusive supply and distribution of the surgical robot ‘Versius’ in Korea. Versius was developed by CMR Surgical, a British company specializing in surgical robots. It has the advantage of having small, modular, and portable robot arms. It can be flexibly deployed according to the surgical method and operating room environment, being highly useful in operating rooms with space constraints. In particular, the company recently launched an indocyanine green (ICG) contrast imaging system that visualizes areas that cannot be seen with the naked eye with 3D HD technology, dramatically increasing the safety and precision of its operations. The market for Versius grew significantly in Europe. The robot has performed more than 20,000 surgeries across Latin America, Asia Pacific, and the Middle East. CMR Surgical has been continuously working to expand Versius’ geographic footprint. Driven by the success of Versius, the company has received investments from global companies such as SoftBank and Tencent since its inception in 2014. Dong-A ST plans to expedite the regulatory procedure for its globally verified Versius, and promptly introduce a new option to Korea’s robot-assisted laparoscopic surgery market. An official from Dong-A ST said, “Versius is a surgical robot that owns excellent technology, convenience, and proven safety. The product is expected to further enhance the efficiency and precision of surgeries for the medical staff.” Dong-A ST has been accelerating its medical device business expansion around the surgical sector. It expressed plans to create synergies by selling an endoscopic automatic suturing device this year and has secured new surgical products.
- Company
- 98% relapse-free Soliris is reimbursed for NMOSD in Korea
- by Son, Hyung-Min May 10, 2024 05:47am
- Ho Jin Kim, Head of Department of Neurology & Division of Clinical Research, National Cancer Center A new treatment option has been introduced for patients with neuromyelitis optica spectrum disorder (NMOSD), a disease that has over a 90% relapse rate. Soliris became fully reimbursable for NMOSD last month, recording a 98% relapse-free rate. However, there are concerns that the stringent reimbursement requirements for Soliris may reduce patient access to the treatment. On May 7, AstraZeneca Korea held a press conference at the Conrad Hotel in Seoul to celebrate the reimbursement of Soliris for NMOSD in Korea. Soliris is a C5 complement inhibitor developed by AstraZeneca. It binds to the C5 protein and prevents the activation of a complement terminal complex, which has been shown to be effective for several autoimmune diseases. Soliris is currently approved for paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), and neuromyelitis optica spectrum disorder (NMOSD). In Korea, its approval for the NMOSD indication was extended in 2021. AstraZeneca Korea succeeded in securing reimbursement for the indication in Korea approximately 3 years after expanding its indication. NMOSD is a rare autoimmune disease characterized by unpredictable, recurrent episodes of optic neuritis and myelitis. It can cause severe disability, including optic neuritis resulting in blindness and paraplegia. 7 out of 10 NMOSD patients are positive for anti-quantiporin-4 (AQP-4) antibodies, which can activate the complement cascade and cause necrosis of the optic nerve and spinal cord. 90% of the patients suffer relapses, and each relapse is characterized by worsening symptoms, increasing the likelihood of blindness, paralysis, and premature death. Soliris has gained attention for showing effects in patients with relapsing NMOSD. In the PREVENT study, at 48 weeks of treatment, 98% of patients in the Soliris treatment arm were relapse-free, compared to 63% in the placebo arm. These relapse prevention benefits were sustained over the 144-week treatment period. The PREVENT extension study confirmed Soliris’s relapse-free rate of 94.4% 197 weeks after treatment. The study also confirmed the drug’s long-term sustained relapse prevention effect. However, some have expressed concerns on how the reimbursement requirements may limit patient access. The drug is currently reimbursed for adult patients with NMOSD who are 18 years of age or older who are AQP-4 antibody-positive and have an Expanded Disability Status Scale (EDSS) scores of 7 or below at the time of Soliris administration and at least 2 symptomatic relapses within the past year or at least 3 symptomatic relapses within the past 2 years (including one within the past year). The issue is that patients have to have multiple relapses to qualify for Soliris’ use. Specifically, patients who are receiving rituximab, an existing treatment for NMOSD, off-label, are ineligible for Soliris’ reimbursement. Patients would have to experience worsening of their disease to receive reimbursement. Enspryng, which was approved for NMOSD prior to Soliris, also has strict reimbursement requirements that prevent many patients from receiving its benefits. Professor Ho-Jin Kim from the Department of Neurology at the National Cancer Center said, “Even a single relapse of NMOSD can cause permanent disability. Active relapse prevention treatment is essential in the acute phase to prevent permanent disability in patients.” He added, “New high-priced treatments that are highly effective in preventing relapses have emerged, but the current reimbursement conditions limit their use in Korea. The expensive drugs should be used when they are most beneficial, rather than in a predetermined order. There is still a lot of work to be done to ensure the most efficient use of these good drugs for our patients." Chul Woong Kim, Director of the Rare Disease Business Unit at AstraZeneca, said, “The reimbursement of Soliris will allow patients with NMOSD to receive treatment for rapid relapse prevention. We will work to improve patient access so that more patients can enjoy Soliris' benefits."
- Company
- Korean AI-driven drug discovery speeds up
- by Nho, Byung Chul May 10, 2024 05:46am
- Ministry of Health and Welfare (MOHW) and the Ministry of Science and ICT will allocate KRW 34.8 billion in AI-driven new drug discovery. AI-driven new drug discovery through data collaboration among the biopharmaceutical industry, medical institutions, research institutions, universities, and public institutions has accelerated and gained significant attention. Previously, individual biopharmaceutical companies in South Korea conducted AI-driven new drug discovery through their in-house protocol, and the process was kept confidential. Starting with the recently organized business group called K-MELLODDY (Machine Learning Orchestration for Drug Discovery), a new system is expected to be implemented within the first half of the year to build upon a system to utilize sources and information related to various clinical and substance owned by 20 institutions. An individual company’s AI-driven new drug discovery could predict a new drug candidate’s ADMET (Absorption, Distribution, Metabolism, Excretion, Toxicity) value via in-vitro studies. However, these test results were insufficient to ensure the drug would pass in-vivo (pre-clinical) or clinical trials. Furthermore, ADMET prediction for clinical trial outcomes requires learning from clinical trial (in-human) data, but not enough clinical or in vivo data makes implementation extremely difficult. Furthermore, beyond predicting ADMET for a single drug molecule, solutions were needed that could expand to include interactions with specific targets, drug-drug interactions, responses based on user types, and various toxicity predictions. Therefore, optimized models based on federated learning have been considered for AI-driven drug development. K-MELLODDY aims to utilize new drug development data without leaking internal data to the outside world. In other words, federated learning and blockchain, which securely maintain companies' intellectual property data while leveraging AI, are evaluated as the core technologies of this project. The project’s characteristics and advantage are differentiated by 'Obtaining reliability of AI-driven new drug discovery,' 'High expandability of platform,' 'Securing ownership of data utilizing technology,' and 'Reducing new drug development cost.' Upon completion of the platform, utilizing AI predictions for the candidate product's metabolism and toxicity experiment results could reduce the number of experiments by half, leading to more than 50% cost savings. “K-MELLODDY project aims to build a federated learning-based AI drug development platform by utilizing the power of data held by Korea's academia, industry, and research institutes related to drug development. It aims to derive successful AI case studies in the drug discovery stage,” Hong Sungeun, Senior Researcher at the Convergence AI Institute for Drug Discovery (CAIID), stated. He added, “A budget of KRW 34.8 billion will be allocated over the next five years to complete the development of a Korean federated learning-based AI new drug discovery model.” Meanwhile, K-MELLODDY is described as a Korean AI new drug platform project that has been improved to suit the needs of the Korean situation while benchmarking the EU-MELLODDY project. EU-MELLODDY is an initiative for data collaboration among 10 pharmaceutical companies, first implemented in the EU. The initial business goal was to prove that data-based cooperation is possible between competing pharmaceutical companies and spread technological safety. Based on the results of EU-MELLODDY, which was conducted in 2019 for three years with KRW 25.6 billion put into, it was observed that the machine learning-based cooperative ADME/Tox prediction model was learned without leaking pharmaceutical companies’ confidential information. In addition, it outperformed the single model formulated by individual institutions without data leakage.
- Company
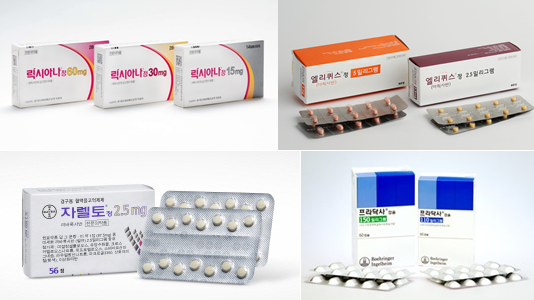
- 'Lixiana' dominates the DOAC market in KOR
- by Kim, Jin-Gu May 09, 2024 05:53am
- (clockwise from top-left) Lixiana, Eliquis, Pradaxa, and Xarelto. Daiichi Sankyo Korea’s 'Lixiana (edoxaban)' continues to dominate the market for Direct Oral Anti-Coagulant (DOAC). BMS’s 'Eliquis (apixaban)' sales have recently experienced a slowdown, and Bayer’s 'Xarelto (rivaroxaban)' prescription performance significantly dropped after the generic launch. The sales of Xarelto generic nearly chased the original product three years after its launch. The pharmaceutical industry eyes on the substance expiry of Eliquis this September. Eliquis generics that withdrew from the market after the Supreme Court’s ruling last 2021 may re-enter the market around the time of patent expiry, and a significant shift in the market is expected. Lixiana, 9% up in one year… Eliquis growth has been stalled According to medical market research firm UBIST, the market of DOAC treatment outpatient prescriptions in Q1 in South Korea amounted to KRW 62.9 billion, up 6% over a year compared to KRW 59.3 billion in Q1 last year. The mechanism of action of DOAC treatment is based on directly affecting the blood coagulation factor, thereby preventing blood clots. It is expanding its usage as it replaces warfarin, which prevents vitamin K metabolism. In South Korea, Xarelto was approved in 2009, followed by Pradaxa and Eliquis in 2011. Pradaxa and Eliquis were also approved in 2015. When the product was launched, it was called New Oral Anti-Coagulant (NOAC). Ten years after its launch, the term was changed to Direct Oral Anti-Coagulant (DOAC) to indicate that the product directly affects the coagulation factor. Four original products have seen a shift in the market. The top-selling product, Lixiana, is strengthening its position as the market leader. Prescription sales of Lixiana in Q1 of last year were KRW 27.7 billion, up 5% over a year compared to KRW 25.5 billion in Q1 of last year. Although it was launched late, its prescription performance grew fast, reaching the top after 2019. Last year, Lixiana became the first DOAC product to reach annual prescription sales of KRW 100 billion. Quarterly DOAC prescription performance (unit: KRW 100 million, source: UBIST). The second-ranked Eliquis has seen a stall in the growth recently. Eliquis prescription sales have seen a 2% growth, increasing from KRW 19 billion in Q1 of last year to KRW 19.3 billion in Q1 of this year. After Q3 of 2022, Eliquis prescription sales remain around KRW 19 billion. As Lixiana sales grew fast while Eliquis sales stalled, the sales gap between the two products is widening. The difference in the prescription sales between these two products in Q1 of this year amounted to KRW 8.5 billion. It continues to widen every year, with a difference of KRW 4.4 billion in Q1 of 2022 and KRW 6.5 billion in Q1 of 2023. Low Xarelto generics sales… Riroxban>Riroxia>Rivoxaban Over the last two years, Xarelto has experienced a significant decrease in prescription sales among the major DOAC products. Xarelto’s prescription sales in Q1 this year amounted to KRW 7.6 billion, down 5% compared to KRW 8 billion in Q1 last year. Its prescription sales declined by almost half over the past two years compared to KRW 14.8 billion in Q1 of 2022. Xarelto’s prescription sales decline is likely due to its patent expiration and generic release that followed. Xarelto generic was launched first in Q2 of 2021. Xarelto’s prescription sales decline is likely due to its patent expiration and generic release that followed. Xarelto generic was launched first in Q2 of 2021. In Q3 of 2022, the stoppage of reducing drug prices for Xarelto due to an administrative suit was lifted. When the pricing of Xarelto decreased, prescription sales started to decline. Eventually, the presence of Xarelto generics increased while the sales of the original prescription gradually decreased. Xarelto generics have gradually increased their prescription sales, nearly catching up with those of the original products. As of Q1, the net prescription sales of generic amounted to KRW 5.9 billion, up 61% over a year compared to KRW 3.7 billion in Q1 of last year. According to Q1 prescription performance report, Hanmi Pharm’s 'Riroxban' generated KRW 1.6 billion, CKD Pharm’s 'Riroxia' generated KRW 1.2 billion, and Samjin Pharmaceutical’s 'Rivoxaban' generated KRW 1.2 billion. Trends in prescription performance of rivaroxaban-containing original product and major generics (unit: KRW 100 million, source: UBIST). Pradaxa, another original DOAC product, is also facing a significant sales slowdown. In Q1, Pradaxa prescription sales amounted to KRW 2.4 billion, down 25% year over year. Eliquis’s substance patent set to expire this September…generic re-entry predicted The future variable in this market is the expiration of Eliquis' patent. Eliquis' substance patent expires on September 9th of this year. Generic manufacturers are waiting for the expiry of the substance patent, with Eliquis' formulation patent set to expire in 2031 after being invalidated. Generic companies won both the first and second trials related to substance patents in 2018 and 2019, respectively. After June 2019, Eliquis generic products were subsequently launched and have since been steadily growing in prescription sales. However, the circumstances changed when the Supreme Court ruled on the reversal of the original decision in April 2021. Generic companies have retrieved their products that could potentially infringe on the patent. On the contrary, Eliquis successfully regained its dominant position in the market for DOAC treatments containing the apixaban ingredient. With the substance patent set to expire this September, a flood of Eliquis generics is possible. Major generic manufacturers have experience releasing their products and are expected to expand their influence in the market quickly. On the other hand, it is expected that Eliquis will experience a sharp decline in prescription performance following the release of generics. From BMS's perspective, there is no justification for delaying the government's price reduction decision following the generic reimbursement listing. Due to this, the pharmaceutical industry anticipates that Xarelto will further strengthen its dominant position in the DOAC market.
- Policy
- Celltrion Pharm completes localizing 3 Edarbi products
- by Lee, Hye-Kyung May 09, 2024 05:52am
- Celltrion Pharm has completed the localization of Edarbi Tab (azilsartan medoxomil potassium), a treatment for essential hypertension. On the 8th, the Ministry of Food and Drug Safety (MFDS) simultaneously withdrew and approved the authorization of Celltrion Pharm's Edarbi Tab 20mg. In December 2020, Celltrion Pharm acquired the entire rights, including sales and patents, of Takeda Pharmaceuticals' 12 ETC and 6 OTC drugs in 9 countries in the Asia-Pacific region for USD 278.3 million (approximately KRW 307.4 billion). The Edarbi license that was withdrawn this time is the item Takeda Pharmaceuticals received approval for in May 2017 and then sold to Celltrion Pharm. Celltrion Pharm had previously received approval for its Edarbi 40 mg and 80 mg in December 2023 and then withdrew the licenses for the Edarbi Tab 40 and 80 mg that the company acquired from Takeda Pharmaceuticals in March 2024. With the simultaneous approval and withdrawal of Edarbi 20mg, the company completed preparations to localize the production of the hypertension treatment. Celltrion Pharm acquired all rights to Edarbi and has been preparing to self-produce and sell the drug to ensure stable product distribution. Edarbi is an angiotensin II receptor blocking (ARB) class drug used for the management and treatment of hypertension. The drug lowers blood pressure by blocking angiotensin II receptors and suppressing angiotensin II, a hormone that increases blood pressure. Adults may take 40 mg of Edarbi once daily, with or without food, and may increase the dose to a maximum of 80 mg per day if the patient’s blood pressure is not adequately controlled. Edarbi 20 mg is indicated as an initial dose for the very elderly (75 years or above) who are at risk for hypotension, patients with mild-to-moderate hepatic impairment, and patients with possible intravascular volume depletion. Following the launch of its alogliptin+metformin combination therapy for diabetes the past year, Celltrion Pharm has received approval for its Edarbi as ‘Celltrion Azilsartan Medoxomil Tablets’ for export and as ‘Edarbi Tab’ for domestic use.
- Policy
- Name of transferred and aquired drugs can be changed in KOR
- by Lee, Hye-Kyung May 09, 2024 05:52am
- Korea’s regulations now stipulate that brand names of a drug transferred to a different company can be changed. According to industry sources on the 7th, the Ministry of Food and Drug Safety recently announced the revision of the 'Casebook for Drug Product Naming Practices (Citizen's Guide)' and announced the allowance of brand name changes. The MFDS changed the regulation that only allowed the company name of a product to be changed upon transfer of a drug that was specified in 2016 to allow the product name to be changed as well. However, in response to inquiries from the pharmaceutical industry regarding the transferred drug’s product name, the MFDS has been responding that "the product name is determined after comprehensive consideration on its domestic use (including similar product names) at the time of completion of the marketing authorization (report).” The pharmaceutical industry had also inquired whether a pharmaceutical company that voluntarily withdraws its marketing authorization for the product and acquires a product with the same ingredient from a third party can apply for the grant to change the product name of the new item to the name previously held by the company. In general, in order to change the product name upon receiving approval for the change in manufactured and imported items (report) due to transfer or assignment, Article 10 (9) of ‘Regulation on Pharmaceuticals Approval, Notification, and Review’ allows for change of a product’s name if it meets the criteria of Article 10 (8) of the same regulation. If a company wishes to use the same product name as the withdrawn item, the company may change the product name to the other name if it is clear that the withdrawn item was not distributed in the market and lacks production and import performance within the period of use granted for the withdrawn item before the transfer. However, the authorities added that the situation at the time of marketing authorization would need to be considered. However, during the revision of the citizen’s guide, the MFDS has given a clear answer that "it is possible to use” the previous product’s name in the event of a product transfer (merger or acquisition, etc.). The MFDS may grant permission to a company to change the name of a product in accordance with Article 11, paragraph 2 of the 'Rules on the Safety of Medicines, etc.' if the product name the company seeks to use is in accordance with Article 11, paragraph 2 of the 'Rules on the Safety of Medicines, etc.’ Meanwhile, the MFDS also reviews the conformity of the product name change as part of the drug's labelling during marketing authorization reviews.
- Policy
- AZ renews orphan drug Strensiq’s risk-sharing agreement
- by Lee, Tak-Sun May 09, 2024 05:52am
- The risk-sharing agreement (RSA) for Strensiq Inj (asfotase alfa, AZ), a treatment for hypophosphatasia, has been successfully renewed in Korea. The drug was last covered under the RSA scheme in June 2020, when it was listed for reimbursement in Korea under the Expenditure Cap Type and Refund Type RSA. According to the National Health Insurance Service on the 8th, AstraZeneca and NHIS recently completed negotiations to renew the risk-sharing arrangement for 5 Strensiq Inj items. Both sides reportedly settled on appropriate terms for the agreement. The 4-year RSA term for Strensiq Inj was set to expire on May 31, but with the renewal, the drug will continue to be reimbursed for 5 years thereafter. The drug’s prior authorization requirement was also removed recently. Previously, if a patient with perinatal/infantile- and juvenile-onset hypophosphatasia wished to use Strensiq as a long-term enzyme replacement therapy to treat bone symptoms, he or she had to be reviewed on an individual basis by the Heath Insurance Review and Assessment Service’s review panel. After 4 years of prior authorizations, Strensiq’s reimbursement status has been converted to post-approvals due to its high prior authorization rate and lack of fluctuation in the number of patients. When it was first introduced, Strensiq was distributed domestically by Handok but has been supplied by AstraZeneca Korea since last year. The exclusion of the prior authorization requirement and RSA renewal are expected to improve patient access to Strensiq in Korea.
- Company
- Novel targeted therapy for gastric cancer 'Vyloy' may launch
- by Eo, Yun-Ho May 09, 2024 05:52am
- 'Vyloy,' a targeted therapy for gastric cancer with a novel mechanism of action, is expected to be launched in South Korea. According to industry sources, Astellas Korea has submitted an application for approval of Vyloy (zolbetuximab), a treatment for claudin 18.2 (CLDN18.2) positive, HER2-negative, unresectable, advanced, or recurrent gastric cancer. In March, it acquired the final approval in Japan. Vyloy is a monoclonal antibody targeting CLDN18.2, which is expressed in the stomach. Its mechanism of action involves binding to CLDN18.2, a protein expressed on the cancer cell membrane of gastric epithelial cells. The efficacy of Vyloy was proven through the Phase 3 SPOTLIGHT and GLOW clinical trials. The SPOTLIGHT trial, which enrolled 557 adult patients with metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma, compared the efficacy and safety of zolbetuximab plus mFOLFOX6 combination therapy (fluorouracil, leucovorin, and oxaliplatin) to placebo plus mFOLFOX6. In the clinical trial, zolbetuximab combination therapy recorded progression-free survival (PFS), the primary endpoint, of 10.6 months, 8.7 months longer than the comparator. Additional clinical results showed that zolbetuximab combination therapy had an overall survival (OS) of 18.2 months, which was improved by 2.7 months compared to the comparator. In the GLOW study, zolbetuximab combination therapy improved PFS and OS. The study compared zolbetuximab plus CAPOX therapy (capecitabine and oxaliplatin) to placebo plus CAPOX therapy. The clinical results demonstrated that zolbetuximab combination therapy had a PFS of 8.2 months, 1.4 months longer than the comparator. The OS of zolbetuximab combination therapy was 14.4 months, compared to 12.2 months for the comparator. The common side effects related to safety were nausea and vomiting. Meanwhile, Vyloy is in progress for the discussion with the U.S. FDA, Europe, and China approvals. In the United States, its approval was rejected once over manufacturing issues.