- LOGIN
- MemberShip
- 2026-05-03 21:03:10
- Company
- Various treatment options introduced for multiple myeloma
- by Eo, Yun-Ho Jul 01, 2024 05:48am
- New treatment options for multiple myeloma treatment are emerging one after another. According to industry sources on the 28th, drugs such as Pfizer’s ‘Elrexfio (elranatamab)’ and GSK’s ‘Blenrep((belantamab mafodotin)’ are receiving attention as promising candidates in the field of multiple myeloma. In the case of Elrexfio, the drug was recently approved in Korea. Elrexfio has been designated as the 4th GIFT (Global Innovative products on Fast Track) drug by the Ministry of Food and Drug Safety. Elrexfio’s efficacy was demonstrated through the Phase II MagnetisMM-3 trial, an open-label, multicenter, non-randomized study that was conducted on 123 who had not received prior BCMA-directed therapy (ie, BCMA-naïve patients). The study evaluated the efficacy and safety of elranatamab monotherapy in patients with relapsed or refractory multiple myeloma (RRMM) who had previously received at least 1 proteasome inhibitor, 1 immunomodulatory drug, and 1 anti-CD38 antibody. Results showed that the overall response rate (ORR), the primary endpoint, of adult patients with RRMM who have received three or more lines of therapy, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 antibody but had not received prior BCMA-directed therapy was 61.0% for the Elrexfio arm, 56.1% of which showed very good partial response (VGPR). Elrexfio is expected to compete with Janssen's bispecific antibody Tecvayli (teclistamab). Like Elrexfio, Tecvayli targets both the BCMA and the CD3 receptor. GSK recently presented data from the DREAMM-8 study on Blenrep at the American Society of Clinical Oncology Annual Meeting (ASCO 2024). The DREAMM-8 study compared the existing treatment for multiple myeloma, pomalidomide plus bortezomib and dexamethasone (PVd), with Blenrep with pomalidomide and dexamethasone (BPd). The study enrolled 300 patients who had failed at least one first-line treatment for multiple myeloma. According to the presented results, 71% of the patients who received BPd survived without disease progression at 1 year. This is approximately 48% higher than that of the PVd combination arm (51%). This translates to a reduction in the risk of further disease progression or death at 1 year after treatment from 49% to 29%, which translates to a nearly half reduction. In 2022, GSK decided to withdraw approval of its BPd therapy determining that it did not improve outcomes for patients with multiple myeloma. However, GSK has since continued to test the efficacy of Blenrep in late-stage clinical studies. Meanwhile, Janssen's CAR-T drug Carvykti (ciltacabtagene autoleucel) also recently unveiled an additional study on multiple myeloma at ASCO. The study showed that Carvykti improved survival compared to the existing second-line standard of care of pomalidomide plus bortezomib plus dexamethasone (PVd) or daratumumab plus pomalidomide plus dexamethasone (DPd) in patients with functionally high-risk (FHR) multiple myeloma.
- Company
- Samsung Bioepis targets Stelara market
- by Hwang, Byung-woo Jul 01, 2024 05:47am
- Samsung Bioepis is seeking to target the Stelara (Ustekinumab) market by directly marketing its Stelara biosimilar Epyztek in Korea. The company is adding Epyztek to the list of autoimmune disease treatments it has been selling directly since March, a strategy that is expected to strengthen its marketing capabilities while increasing profitability. A view of the Samsung Bioepis building in Songdo.Samsung Bioepis will launch Epyztek, an autoimmune disease treatment, in the Korean market in July. Epyztek is a biosimilar of Stelara, Janssen’s treatment for autoimmune diseases such as plaque psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis. Epyztek was approved by the Ministry of Food and Drug Safety in April. Stelara inhibits interleukin (IL)-12/23 activity, a class of inflammatory cytokines (signal transmitters). As of 2023, its annual global product was around KRW 1.4 trillion, and domestic market sales were around KRW 41.6 billion. According to the ‘Drug Reimbursement List and Reimbursement Ceiling Table' announced by the Health Insurance Review and Assessment Service as of July 1, Epyztek is priced at KRW 1,298,290 per 45mg/0.5ml prefilled syringe. This is about 40% lower than the price of the existing original drug. Although Stelara’s price will also be reduced following the launch of the biosimilar, Epyztek still has a price advantage. Professor A, Department of Gastroenterology at a tertiary hospital in Seoul, Korea, said, "There seems to be little doubt about the effectiveness of biosimilars as prescriptions have accumulated. The financial burden of Stelara’s initial intravenous infusion may serve to lower the threshold for the introduction of its biosimilars." The biggest characteristic of biosimilars is in their lower cost compared to the original. However, in Korea, the price difference is not large due to the low patient copayment rate applied through Korea’s insurance reimbursement system, but Stelara’s characteristics may still provide competitivity for the biosimilars. Another characteristic of Epyztek is that the company sells the product directly. Since March, Samsung Bioepis has switched to direct sales of 3 autoimmune disease drugs that it had been selling in partnership with Yuhan Corp. With the exception of anticancer and ophthalmic drugs, the company’s decision to switch to direct sales is interpreted as the company’s attempt to increase the profitability of the drugs. Currently, Samsung Bioepis has about 20 domestic sales personnel for direct sales, and in March, it secured a distribution network in the domestic market by signing a pharmaceutical third-party logistics contract (3PL) with Geo-Young. In the field, the prospect is that Epyztek’ssales will depend on Samsung Bioepis' marketing capabilities and how quickly it can land in the market. An industry official said, "It is true that the marketing capabilities cannot be ignored due to the nature of biosimilars. However, given how Samsung Bioepis already has experience in selling biosimilars for autoimmune diseases, we expect sales to be directly related to how quickly the drug can land in each hospital." A Samsung Bioepis official said, “We are able to offer various treatment opportunities for patients with autoimmune diseases with the launch of Epyztek. Based on the reasonable drug price, we expect the drug to address the unmet demand in the field and contribute to saving national health insurance finances."
- Policy
- Expenses for the major four severe diseases tops KRW 7T
- by Lee, Tak-Sun Jul 01, 2024 05:47am
- Pharmaceutical expenses for the four major severe diseases exceeded KRW 7 trillion last year. The four major severe diseases refer to cancer diseases, cerebrovascular disease, and rare·severe incurable diseases. Pharmaceutical expenses for this category have risen due to high-cost pharamceuticals. Consequently, an analysis suggests that managing pharmaceuticals in this category is crucial for continuing the National Health Insurance. The Health Insurance Review and Assessment Service (HIRA) published the '2023 Report on Reimbursement Claims for Pharmaceuticals Expenses' on June 27th. It indicates that the claim amount for the four major severe diseases last year was KRW 7.348 trillion, a 20% increase from KRW 5.8495 trillion in 2022. Pharmaceutical expenses for the four major severe diseases (unit: 1,000 cases, KRW 1 million). Notably, pharmaceutical expenses for cancer disease and rare·severe incurable diseases are increasing rapidly. Anticancer drug expenses have reached KRW 4 trillion. Last year’s expense was KRW 3.8506 trillion, up 23.5% Year-over-Year (YOY). Pharmaceutical expenses for rare·severe incurable diseases topped KRW 3 trillion. It was KRW 3.337 trillion for 2023, up 16.2% YoY. Pharmaceutical expenses for cerebrovascular and cardiac diseases were KRW 803.5 billion and KRW 700.5 billion, respectively, and they have maintained an increasing trend. In contrast, the percentage of pharmaceutical expenses out of the total medical fees for the National Health Insurance has remained at 23% for the past two years. Last year’s figure was 23.86%, an increase from 23.34% in 2022 but still at around 23%. From 2019 to 2021, pharmaceutical expense was at around 24%. However, last year’s rate of increase in pharmaceutical expenses showed the highest figure in the past five years. The rate of increase for pharmaceutical expenses was 12%, recording a double-digit figure. This seems to be relevant to the rapid increase in pharmaceutical expenses for the four major severe diseases. Last year’s total pharmaceutical expense was KRW 25.6446 trillion. The percentage of pharmaceutical expense for the major four severe diseases out of the total expense amounted to 27.4%. The government is in the process of improving the post-management system and re-assessment for pharmaceutical expense management. Specifically, it plans to establish a system to post-manage high-cost pharmaceuticals after reimbursement and re-assess the pricing, such as lowering the pricing after comparing to foreign drugs or patent-expired drug costs.
- Company
- Ilaris’s reimbursement imminent in Korea
- by Eo, Yun-Ho Jul 01, 2024 05:47am
- The ultra-rare disease treatment Ilaris has finally passed the final hurdle to its reimbursement in Korea. Novartis Korea recently completed drug price negotiations with the National Health Insurance Service for Ilaris (canakinumab), a treatment for hereditary periodic fever syndrome. The company was quick to accept the conditional benefit decision from the National Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee in April, and quickly finalized what was expected to be a difficult drug price negotiation process. Novartis' determination for reimbursement is notable. The company resubmitted the application and received a re-deliberation from DREC on the 4th after receiving a conditional reimbursement decision in February, but received the same results. However, the reduced scope of data requested by the government opened up the possibility of reimbursement. In February, Novartis was unable to accept the condition proposed by DREC. However, during this round of review, DREC likely requested less additional supplemental data with the conditional reimbursement approval decision, which Novartis was able to accept this time. It is also notable how the government swiftly responded to the first claim and reduced the scope of the evidence that was required for submission. Ilrais’s reimbursement agenda will be passed along to the Health Insurance Policy Deliberation Committee, after which the company anticipates that the drug will be reimbursed starting in August. Ilrais is indicated for the following diseases in Korea: ▲Periodic fever syndromes (PFS), cryopyrin-associated periodic syndromes (CAPS), tumor necrosis factor receptor associated periodic syndrome (TRAPS), hyperimmunoglobulin D syndrome (HIDS)/mevalonate kinase deficiency (MKD), and familial mediterranean fever (FMF) ▲Active systemic juvenile idiopathic arthritis (Systemic JIA). For CAPS, the indication can be further categorized into the following symptoms: ▲Familial cold autoinflammatory syndrome (FCAS)/ familial cold urticaria (FCU) ▲Muckle-Wells syndrome (MWS) ▲Neonatal onset multisystem inflammatory disease (NOMID)/chronic infantile neurological, cutaneous and articular syndrome (CINCA).
- Opinion
- [Reporter’s View] No innovation without failure
- by Son, Hyung-Min Jul 01, 2024 05:47am
- Korean biopharmaceutical companies are exchanging joy and sorrow over out-licensing agreements. Last month, three companies, including HK inno.N, have successfully signed agreements to out-licensing of their new drug candidates in addition to AprilBio and ISU Abxis. In contrast, there are instances where new drug candidates that have been successfully out-licensing are later returned. GC Cell announced that a collaborative R&D agreement with Artiva Biotherapeutic, its U.S. subsidiary, for three treatments for CAR-NK solid tumors in 2021 has terminated. Furthermore, Olix Pharmaceuticals and Curacle received notifications of contract terminations from France’s Thea Open Innovation last month. In April, Voronoi terminated the out-licensing agreement made in 2022 with METiS Therapeutics, a biotech company in the United States, for its solid tumor treatment VRN14. Yet, it is too early to be disappointed about the termination of out-licensing agreements. There are instances where companies can succeed by developing drugs for new indications. For instance, last year, Hanmi Pharmaceutical confirmed the potential of Poseltinib for the treatment for recurrent or diffuse large B-cell lymphoma (DLBCL). The licensing of Poseltinib, a BTK inhibitor under development by Hanmi Pharmaceutical, was returned from Lily in January 2019 because the efficacy of the drug was not confirmed in a phase 2 trial involving patients with rheumatoid arthritis. Additionally, Hanmi Pharmaceutical is showing a process in clinical trials for efpeglenatide, a new drug candidate for obesity. Efpeglenatide is a product that initially struck a licensing agreement with Sanofi in 2015 to treat diabetes and later returned to Hanmi Pharmaceutical. Then, Hanmi Pharmaceutical changed its development strategy for this drug to a Korean-focused obesity treatment. Based on recent clinical results, Efpeglenatide demonstrated weight loss effects. During the new drug development process, selecting and focusing are necessary steps. The company’s R&D strategy can be changed, and instances of new drug candidates that fail to demonstrate efficacy can occur. Out-licensing returns are common practice in the pharmaceutical industry. Fortunately, none of the four returned new drug candidates had significant issues regarding their efficacy. Companies with returned technologies plan to proceed with later phases of clinical trials without delays. Now is the time for companies to clearly announce their potential development directions to seek future investment opportunities. When clear directions are suggested, they could seek opportunities to out-license their new drug candidates again. There is no innovation without failure. If a company firmly believes in a pipeline item (new drug candidate), it may not worry about each case of returned licensing or the success of out-licensing. Only the companies that are not discouraged or give up are poised to survive.
- Company
- GC Cell-Artiva-MSD’s 'CAR-NK' co-R&D deal terminated
- by Kim, Jin-Gu Jun 28, 2024 04:33am
- Despite the recent termination of the collaborative R&D agreement with MSD, GC Cell and Artiva will continue their collaborative research on ‘CAR-NK solid tumor treatment.’ GC Cell and Artiva Biotherapeutic’s collaborative research and development (R&D) agreement with MSD for ‘CAR-NK solid tumor treatment’ has been terminated. MSD informed Artiva Biotherapeutic, GC Cell’s U.S. subsidiary, of the contract termination. As a result, Artiva Biotherapeutic and GC Cell’s research agreement has been automatically ended. On June 25th, GC Cell announced a collaborative R&D agreement with Artiva Biotherapeutic for three treatments for CAR-NK solid tumors has terminated. In January 2021, GC Cell signed a collaborative R&D agreement with Artiva for CAR-NK cell treatment targeting solid tumors. GC Cell received an upfront payment of US$15 million (approximately KRW 17 billion) and was to receive milestone payments of up to US$966.75 million (approximately KRW 1.34 trillion). At that time, MSD joined the agreement as well. Artiva was responsible for R&D, with MSD participating as the main sponsor. GC Cell was responsible for the research aspect of Artiva’s R&D. Then, GC Cell completed the research requested by Artiva and delivered the results. However, despite this effort, MSD recently informed Artiva of the contract termination due to an internal decision. As a result, the agreement between Artiva and GC Cell was also terminated. GC Cell stated that despite the termination of the collaborative R&D agreement, with MSD participating as the main sponsor, the CAR-NK platform research conducted by GC Cell and Artiva as a collaborative effort will continue. GC Cell is collaborating with Artiva to conduct research on three candidate products, including AB-101, AB-201, and AB-202. Specifically, AB-101 is in a phase 1/2a trial in the United States. AB-201 has received IND approval for its phase 1 trial in the United States. The company is preparing to enter the U.S. clinical trial for AB-202. “The 2021 agreement was primarily led by Artiva. After the contract between Artiva and MSD was terminated, the agreement between GC Cell and Artiva automatically ended,” GC Cell’s official said. “Consequently, we are not obligated to return the upfront payment of US$15 million.“ The GC Cell official explained, “This does not mean the end of collaborative R&D of the CAR-NK platform with Artiva. GC Cell will primarily continue the R&D of the CAR-NK platform.”
- Company
- KOD will service platform for epilepsy patients next year
- by Lee, Tak-Sun Jun 28, 2024 04:33am
- A platform for epilepsy patients and healthcare professionals will soon be launched in Korea. Japan's Knock on the Door (KOD) announced in May that it has established its branch in Seoul and is preparing to launch the service next year. KOD was established in Tokyo, Japan, in 2018 with the goal of "Realizing a society where patients with intractable diseases and their families can shine and always stay true to themselves.” Hiroomi Hayashi, Representative Director of KOD, said, “To achieve this goal, we provide a platform where we work with patients with intractable diseases and their families to improve their medical care, life, and quality of life.” Specifically, KOD has built a platform for Japanese patients with intractable epilepsy. The platform includes "nanacara," an application that allows patients to record symptoms such as seizures and medication information, "nanacara for Dr.," which allows specialists to check the information recorded by patients and their families during medical visits, and "nana-medi," an online medical consultation and medication guidance service that allows patients to connect with specialists and pharmacists anywhere in the country. Director Hayashi explained, "By leveraging our accumulated data and network of patients, families, and doctors that use Nanacara, we provide solutions to pharmaceutical companies to help improve the quality of medical care for intractable diseases. We strive to create a society where high-quality medical care can be quickly accessed anywhere in Japan." Currently, there are approximately 1 million people with epilepsy in Japan. Approximately 300,000 are intractable epilepsy patients and approximately 100,000 are pediatric intractable epilepsy patients. There are about 900 epilepsy specialists in Japan, affiliated with 450 medical institutions. Currently, about 30,000 people in Japan have downloaded and used Nanacara, mainly pediatric intractable epilepsy patients and their families. This is around 30% of the affected patients in Japan. In addition, 265 of the approximately 450 medical institutions with epilepsy specialists have adopted Nanacara for Dr., which represents a 60% share. Director Hayashi said, “Through these platforms, we can now more easily and accurately measure the patients' seizure records and facilitate smoother communication with healthcare professionals in their institutions. Many patients and their families have also gained physical and mental relief with the use of our platform.” KOD began communicating with epilepsy patients and families in Korea last year. During the discussions, the company identified the need to document seizure records and rapid delivery of information amongst Korean patients as well. To address the need, KOD established a branch office in Seoul in May and is preparing to launch the service in 2025. In Korea, Nanacara will be rebranded as 'Borabora.’ Director Hayashi added, "We want to create an environment in Korea where more people can support people with epilepsy, and where people with epilepsy can live true to themselves. We hope that many people in Korea will use the platform and contribute to establishing an environment where patients can continue living a rewarding and valuable life.” KOD also participated in the International Epilepsy Congress that was held in Seoul from the 21st to the 22nd and held a booth presentation for Nanacara. During the exhibition, we received many opinions from Korean epileptologists. Building on the input, we will begin preparations to make Nanacara for Dr. available to Korean epileptologists as well. "We will also actively promote cooperation with related pharmaceutical companies that develop and provide anti-epileptic drugs in Korea. We hope our collective efforts will contribute to the development of medical care for epilepsy in Korea."
- Company
- MSD seeks to expand NIP subjects for Gardasil
- by Hwang, Byung-woo Jun 28, 2024 04:33am
- With the aim of expanding coverage of its human papillomavirus (HPV) vaccine within the National Immunization Program (NIP), MSD Korea has been working to improve its vaccine awareness. Expanding NIP coverage of the HPV vaccine is one of the hot topics on the government's agenda. In fact, it was ranked the third-most important task in the 'Establishing a Mid- to Long-term Plan for the National Immunization Program' study. MSD KoreaHowever, the fact that the government has set limits on the cost and subject is acting as an obstacle. According to the study, the issues discussed expanding NIP coverage to 'vaccination of girls under 12 years of age with Gardasil 9' and 'vaccination of boys under 12 years of age'. In the study, expanding vaccination of Gardasil 9 to 12-year-old girls ranked third in the overall priority list. However, vaccinating 12-year-old boys with Gardasil 9 was set as the sixth priority. The researchers differentiated the priority for the same vaccine by subject. Against this backdrop, MSD Korea has been emphasizing the need for HPV vaccination in both boys and girls At a conference held in late May, MSD Korea mentioned that 33 out of the 38 Organization for Economic Co-operation and Development (OECD) countries (as of April 24, 2014) have introduced NIP for male HPV vaccinations. Sei-Young Lee, Professor of Otolaryngology and Head-and-Neck Surgery at Chung-Ang University Hospital, said, "In men, HPV-related cancers and diseases on the rise globally, and their effect in increasing disease burden and reducing the quality of life are being undermined. Korea's male HPV vaccination rate is in the single digits, which is far below that of Australia and the United Kingdom, which have been actively carrying out HPV prevention programs." In this situation, MSD Korea is trying to build awareness of the need through social media activities. In addition to the top-down approach of emphasizing the need for HPV vaccination through pharmaceutical companies and academic communities, MSD is also working on a bottom-up approach, increasing awareness of the need for Garadsil’s coverage through NIP amongst vaccine recipients. The company has been promoting the need for HPV vaccination through social media outlets that are occupied by young people during the back-to-school season, coming-of-age day, etc. The key message is that both men and women need to be vaccinated. Pic of Gardasil 9An industry official said, "The ultimate aim for most vaccines is to be included in the NIP, so MSD Korea is constantly knocking on the door for expanded NIP coverage for Gardasil 9. Awareness-raising activities can be cumbersome as even social media posts are subject to advertising review, but the company seems to have recognized the need to increase awareness." According to the drug research institution IQVIA, Gardasil 9’s sales grew to KRW 72.6 billion in 2021, and to KRW 117 billion in 2022, then fell to KRW 106.4 billion last year. If the NIP is only applied to HPV vaccines vaccinated to women, and the product is switched to Gardasil 9, its sales could further decrease due to lowered supply prices. In other words, apart from including male HPV vaccinations in the NIP, the company would need to build awareness of its needs. An industry insider said, "If Gardasil 9 is included in the NIP, the government’s supply price will have to be lower than the current market price. If the NIP vaccination covers boys, the reduced price will be offset by the increased coverage. Even if MSD's best-case scenario of expanding coverage to both genders does not become realized, improving awareness would be essential for sales."
- Company
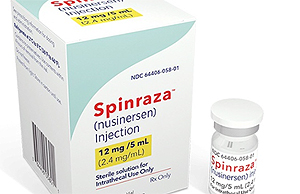
- Spinraza tops Q1 revenue in SMA mkt with KRW 17.7 billion
- by Nho, Byung Chul Jun 28, 2024 04:32am
- (From the left) Spinraxa Inj, Zolgensma Inj, and Evrysdi for spinal muscular atrophy Biogen Korea's Spinraza remains the No. 1 drug for spinal muscular atrophy (SMA) in the KRW 80 billion market. However, its performance has been somewhat sluggish due to the launch of competitors such as Roche Korea’s Evrysdi and Novartis’s Zolgensma. In terms of drug distribution performance, Spinraza's sales in Q1 of this year were KRW 17.7 billion, 9 to 14 times higher than that of Zolgensma (KRW 1.8 billion) and Evrysdi (KRW 1.2 billion). Sales of Spinraza (nusinersen sodium) were in the range of KRW 72.2 billion, KRW 61.2 billion, KRW 62.3 billion, and KRW 58.9 billion, in 2020, 2021, 2022, and 2023, respectively. Zolgensma (onasemnogene abeparvovec), which was approved in Korea in 2021 sold KRW 16.2 billion and KRW 14.4 billion in 2022 and 2023, respectively. Evrysdi (risdiplam), which was approved by the MFDS in 2020 generated KRW 600 million and KRW 1.123 billion during the same period. What is unusual is that Zolgensma exceeded last year's sales in just the first quarter of this year, while Evrysdi’s sales tend to be somewhat sluggish in terms of quarterly results alone. Spinraza (5m), Evrysdi (80ml), and Zolgensma (1 kit) are all ultra-high-priced orphan drugs and are priced at KRW 92.35 million, KRW 9.52 million, and KRW 1.98172 billion, respectively. . Spinraza can be administered directly to the central nervous system, where motor neurons are located, through intrathecal injection to deliver the treatment at the source of the disease. It can also be administered in multiple doses, making it easier to monitor clinical outcomes. Based on clinical study data and real-world evidence (RWE) accumulated over up to 8 years of treatment, the treatment has confirmed sustained efficacy and safety profile across all ages and types of patients. In addition, its reimbursement extension to patients with Type 3 SMA who have developed symptoms after the age of 3 is working in favor of the drug. As a one-shot treatment, Zolgensma works by inserting a functional replacement copy of the SMN1 gene into a carrier called a vector and delivering it to motor neuron cells in the body via intravenous infusion. The disadvantage is that it is difficult to administer multiple doses to patients who cannot be treated with a single dose.
- Policy
- Gov't releases generic drug price comparison study results
- by Lee, Tak-Sun Jun 27, 2024 05:47am
- The government's decision to fully disclose the 'Study on Measures to Improve the Generic Drug Pricing System,' which ended in April, ahead of the final meeting for the external reference pricing reevaluations, is being analyzed as a sign that the authorities intend to push ahead its plan to cut generic drug price through reevaluations. In particular, the industry believes the government is using media to publicize the study findings that the generic drug price is up to 10 times more expensive in Korea than in other countries, excluding the U.S., as a means to justify the reevaluation. According to industry sources on the 25th, the 10th meeting on the external reference pricing reevaluations is scheduled for the 27th. The government is reportedly planning to finalize the process of collecting opinions from the pharmaceutical industry within this month. Therefore, the 10th meeting is expected to be the final meeting between the government and the industry. In this situation, there are voices that the recently released study on how to improve the generic drug pricing system is just the government’s means to justify its plan for external reference pricing reevaluations. The study compared drug prices with the A7 countries (Canada, France, Germany, Italy, Japan, Switzerland, and the United Kingdom), excluding the United States, where generic prices are relatively high. As a result, as of 2022, the price of hyperlipidemia generics in most countries was 0.09 to 0.41 times cheaper than those in Korea. However, the study concluded that the comparison of Korean generic drug prices with those of A8 countries showed differences by efficacy group and API and that there is a limit to unilaterally applying price cuts to generic drugs. The pharmaceutical industry analyzes that the comparison of drug prices excluding the U.S. used in the study is similar to the recent government's proposed external reference pricing reevaluation, which suggested using an adjusted average price that excludes the highest and lowest prices among the A8 countries. This is because excluding the highest price removes the U.S. price, which has the highest generic drug prices, from the comparison. As shown in the study, excluding the U.S. price will lead to Korea having relatively high prices for hyperlipidemia drugs, and domestic pharmaceutical companies are concerned that the performance of related generic drugs will suffer from the price cuts that may follow. An industry official said, "Off-patent drugs can become cheaper abroad than in Korea over time. In foreign countries, generics occupy a higher market share than the original over time, so they can maintain sales even at the lowered price." On the other hand, the industry official explained that there is no difference between Korea and the A8 countries when comparing generics that newly entered the market upon patent expiry. "It is problematic to compare overall prices based on only one aspect when generic prices vary depending on the time of launch or disease. We should be giving incentives to increase the generic drugs’ market share and grow the market pie like foreign countries." The study also proposed the creation of a market structure that allows generic drugs to gain competitiveness. For example, in order to create a drug pricing structure where drug prices can be reduced over time after generics enter the market, it is necessary to strengthen not only the pricing policies used at the time of drug listing but also policies to encourage the use of low-priced products in the field. However, the government will likely first focus on lowering domestic generic prices through external reference pricing system.