- LOGIN
- MemberShip
- 2026-05-08 13:04:47
- Company
- "Cibinqo secures solid data in atopic dermatitis"
- by Jung, Sae-Im Jul 13, 2023 05:35am
- 'Cibinqo (abrocitinib),' the third JAK inhibitor to be introduced for atopic dermatitis in Korea, will be reimbursed starting this month. Healthcare professionals have expressed high expectations for Cibinqo, as the drug has accumulated robust data in treating severe atopic dermatitis. On the 11th, the Korean pharmaceutical company Hanmi Pharmaceutical held a press conference to celebrate the reimbursement of Cibinqo, an oral atopic dermatitis treatment, at the Seoul Dragon City Hotel in Yongsan-gu, Seoul. Professor Yong-Hyun Jang of the Department of Dermatology at Kyungpook National University Hospital explained the background and significance of Cinbinqo's reimbursement for atopic dermatitis in Korea. Professor Yong-Hyun Jang of the Department of Dermatology at Kyungpook National University Hospital is presenting at the press conference celebrating the reimbursement and listing of Cibinqo Cibinqo is a Janus kinase 1 (JAK1) inhibitor that was approved by the Ministry of Food and Drug Safety in November 2021. It is also the 4th JAK inhibitor in Korea and 2nd JAK class treatment to be introduced by Pfizer. Unlike Xeljanz, which is used only for ulcerative colitis, Cibinqo can also be used for the treatment of severe atopic dermatitis. Cibinqo was granted reimbursement this month. The 50mg, 100mg, and 200mg doses of Cibinqo were listed at KRW 11,087, 17,739, and 25,942 respectively. The ceiling price is known to be set at 88% of the calculated average price of its substitutes. With the listing of Cibinqo, patients with severe atopic dermatitis now have a much wider range of new treatment options. In addition to the biological agent ‘Dupixent’ that was first introduced, 3 JAK inhibitors – the already-reimbursed Olumiant (baricitinib) and Rinvoq (upadacitinib), and Cibinqo – are now available for use in Korea. Among these treatments, Dupixent, Rinvoq, and Cibinqo can be used in adolescent patients aged 12 and above. Although being a latecomer, Cibinqo received recognition for having acquired solid data in atopic dermatitis from various clinical trials. 6 Phase III clinical trials, including JADE DARE, JADE COMPARE, and JADE TEEN, have been conducted. Cibinqo was the first drug that attempted to directly compare (head-to-head) its efficacy and safety with the biological agent Dupixent, and Cinbinqo was observed for the possibility of deterioration upon dose reduction or discontinuation of treatment after initial response, and evaluated on its ability to recover from the reaction. By confirming the drug’s effect on patients that showed no response to Dupixent, the company accumulated grounds for patients to switch to Cibinqo. The rapid improvement of itch, which was observed in various clinical trials of Cibinqo, is expected to significantly improve patient’s quality of life. Jang explained, "Itch is a very important symptom in atopic dermatitis. It causes patients to scratch their skin, weakens the skin barrier, and exacerbates inflammation as a secondary reaction. Cibinqo showed a rapid and significant improvement in itch from the day after initial administration, increasing patients' quality of life. Also, more patients reached the target response rate for the Eczema Area and Severity Index (EASI). We believe Cibinqo will benefit patients who are suffering from severe itch." Cibinqo has also demonstrated additional benefits in patients who did not respond to Dupixent. The JADE EXTEND study investigated the effect of switching to Cibinqo from Dupixent in patients who received Dupixent in the JADE COMPARE study. The patients were further divided into those that responded to Dupixent and those that didn’t respond to Dupixent. In the arm that did not respond to Dupixent, 80% achieved EASI-75 (75% improvement based on the Eczema Area and Severity Index). Also, 77% achieved an improvement of 4 points or more on the Peak Pruritus Numerical Rating Scale (PP-NRS). This is expected to serve as evidence when companies of JAK inhibitors apply for reimbursement extensions to cover patients switching from Dupixent. Jang stated, "Unlike other JAK inhibitors, Cibinqo holds significance as it conducted various studies solely in the field of atopic dermatitis, which is significant. With the basis for switching treatments established, we expect reimbursement extensions to follow in the near future. We are currently collecting various opinions in academia as well.”
- Policy
- Will the number of generics decrease?
- by Lee, Tak-Sun Jul 13, 2023 05:35am
- Analysts say that the government has begun strengthening the standard for cascading drug pricing in order to reduce the number of generics. A plan to reduce the existing list of 20 items, which is currently used as the standard for lowering the upper limit of new items, to 10 seems promising. However, the industry points out that there is a limit to reducing the number of large generics even if the tiered drug pricing standard is strengthened. Rather, it is predicted that it will only encourage development. The Ministry of Health and Welfare recently entrusted a research service to Kongju University professor Kim Dong-sook's team on the subject of 'preparation of measures to improve the drug price system for generic drugs'. In this study, the appropriateness of 20 products, which is the standard for the number of products to which differential price is applied, will be addressed. The 20 baselines were introduced in 2020 with the reorganization of the drug pricing system. If a generic enters the market with more than 20 identical products already listed, the upper limit will be set at 85% of the lower price between the lowest price of the same product and 38.69%. Unfortunately, the NHIS-affiliated Health Insurance Research Institute recently introduced the Japanese generic drug pricing system through a publication in the June 2023 issue and View, arguing that it needs to be reflected in the domestic system. In particular, when the number of generics is 10 or more, the generic price is calculated at 40% of the original price, and when there are 20 or more, the system is set at 90% of the lowest price. There was an atmosphere that the current baseline of 20 needs to be reduced to 10 like Japan. It is known that the Ministry of Health and Welfare, NHIS, and HIRA have also mentioned the plan to have a strict baseline for generic drug price cuts. Accordingly, the industry is of the opinion that it is not possible to further strengthen the cascading drug pricing system by creating a clear basis through research services. However, it is pointed out that there is a limit to reducing the number of large generics no matter how low the baseline for cascading drug pricing is. The cascading drug pricing system will be applied from the month after the first generic is listed. Therefore, in the first month, there is no penalty even if the number of entries exceeds 20. In the case of Forxiga's generic 10mg, which served as an opportunity for insurance authorities to pursue a plan to reduce the number of generics, 57 were listed in the first month of April alone. Most of the generic companies tried to register their products according to the patent expiry schedule to avoid the cascading drug pricing system. For the next two months, only one of Forxiga's generic 10mg was listed. Industry analysts say that generics with a large market size, such as Forxiga's generic, are highly likely to be listed on a large scale in the first month regardless of the cascading drug pricing baseline. The same is true for Januvia generics scheduled to be listed in September. Therefore, in Korea where generic competition is fierce, even if the price cut baseline is reduced from 20 to 10, the number of large generics will not change significantly. Rather, it is pointed out that generic companies can only waste social costs by trying to develop without asking regardless of expected performance in order not to be late for entering the insurance coverage. An official from the pharmaceutical industry explained, "There is a possibility that generic companies will flock to patent challenges at once to get reimbursed early." However, if the baseline is set from 20 to 10, the effect of not increasing the number of listed generics will be more significant. However, it is also pointed out that there is a limit to reducing the total number of generics. Another official in the pharmaceutical industry said, “Although Japan has generally reduced the price of generic drugs, it is different from Korea in that it is simultaneously promoting generic incentives, such as actively granting incentives to doctors and pharmacists for generic prescriptions.” He criticized, "Our insurance authorities are only concentrating on lowering the price of generic drugs and seem to have no interest in encouraging their use."
- Company
- Schizophrenia drug Invega Hafyera lands in general hospitals
- by Eo, Yun-Ho Jul 13, 2023 05:35am
- The long-acting formulation of the schizophrenia drug ‘Invega’ has landed in general hospitals in Korea. According to industry sources, Invega Hafyera 1092mg/1560mg (paliperidone palmitate), Janssen Korea’s extended-release injectable suspension for patients with schizophrenia that is injected every 6 months, has passed the drug committees of medical institutions in Korea, including Seoul Asan Medical Center. Janssen has been continuously working to create a prescription environment since it was listed for reimbursement in May. Invega Hafyera was approved by the Ministry of Food and Drug Safety in September 2022. The 1-month extended-release injectable formulation of the same drug, Invega Sustenna, had been approved in July 2010 and granted reimbursement in November 2015. With reimbursement, the drug is covered for all patients with schizophrenia in Korea, including first-episode patients. The 3-month extended-release injectable formulation, Invega Trinza, was approved in June 2016 and is being reimbursed from September 2016. Invega Hafyera is indicated for use in patients who have been adequately treated with: Invega Sustenna (once-a-month extended-release injectable suspension) for at least 4 months; or Invega Trinza (every-three-month extended-release injectable suspension) for at least one three-month cycle. Patients who wish to switch to Invega Hafyera from Invega Sustenna 156mg receive Invega Hafyera 1092mg, and those switching from Invega Sustenna 234mg receive Invega Hafyera 1560mg as an initial dose. In the case of Invega Trinza 546mg and 819mg, patients may switch to Invega HafyeraTM 1092mg, and 1560mg, respectively. The safety and tolerance of the every-six-month injectable Invega Hafyera were confirmed through the PSY3015 study. Se Hyun Kim, Professor of Psychiatry at Seoul National University Hospital, said, “Invega Hafyera can offer a broader range of benefits to patients who have seen a stable effect with Invega Sustenna or Invega Trinza. The new formulation offers an opportunity to lower the barrier to injectable treatments for schizophrenia patients in Korea.” Kim added, “The long-acting injectable formulation allows continuous treatment with its stable medication adherence and improved convenience, and can provide benefits such as returning to society and regaining confidence for patients with schizophrenia.” In schizophrenia, the risk of worsening or relapsing symptoms is high with low medication adherence. The long-acting injectable formulations offer a benefit in that aspect as it improves medication adherence over oral formulations, reduce medical cost and relapse or rehospitalization rates, improve symptoms and social function of the patients (when switching to a long-acting injectable formulation), and offer convent administration. The treatment guidelines for schizophrenia in Korea allow long-acting injectable formulations to be administered to patients in all patients, from early to chronic stages.
- Policy
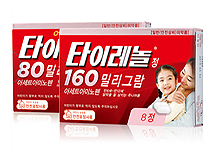
- 2 Tylenol products stopped production will be decided in 2Q
- by Lee, Jeong-Hwan Jul 13, 2023 05:35am
- In the second half of this year, the Ministry of Health and Welfare plans to discuss the cancellation of the safety household medicine designation and the designation of additional alternative medicines for two Tylenol items, which have stopped production due to the sale of Janssen Korea's Hyangnam plant. On the 11th, the Ministry of Health and Welfare Ministry of Health and Welfare's Pharmaceutical Affairs Policy Division announced through a press briefing on media reports that the response to Tylenol, a safe and household medicine that has been suspended for more than a year, was insufficient. Some items, such as Tylenol 80mg for children and Tylenol 160mg, were withdrawn in March 2022 due to the relocation of the Tylenol manufacturer's factory overseas. A media company reported a critical article claiming that the government was neglecting the two items even though they had not been supplied for a year and four months due to a halt in production. In response, the Ministry of Health and Welfare explained that there is a considerable amount of inventory that has already been produced and that there are items that are trying to re-permit after relocating the factory, so they are examining the situation in consideration of the use of inventory and the possibility of re-permission. In particular, it also announced that it would make a decision within the second half of the year on the cancellation of the designation of safety and emergency medicines for two items that have been discontinued and the designation of additional alternative medicines. Regarding the suspension of supply, the Pharmaceutical Affairs Policy Division said, “It is not that the supply to convenience stores has stopped because the stock that has already been produced is being distributed.” The Pharmacy Policy Division continued, "Tylenol tablets for children 80mg and Tylenol tablets 160mg are alternative items such as children's Ibuprofen syrup and children's Tylenol suspension." "We will quickly discuss and decide on the need to cancel the designation of safe household medicines for the two items that have been discontinued and the need to additionally designate alternative drugs. According to the safety and household medicine system, the designation decision is made in consideration of the ingredients, side effects, content, dosage form, awareness, and convenience of purchase of the item," the Pharmaceutical Affairs Policy Division said.
- Company
- Samsung Biologics, cumulative order amt exceeded 2 trillion
- by Hwang, Jin-joon Jul 11, 2023 05:41am
- Panoramic view of Samsung BiologicsSamsung Biologics announced on the 10th that it has signed a main contract for biopharmaceutical consignment manufacturing (CMO) worth 511.1 billion won from global pharmaceutical company Novartis. Through this contract, the cumulative amount of orders received this year increased to 2.3387 trillion won. This contract with Novartis was signed one year after the letter of intent (LOI) of 100 billion won in June last year. Samsung Biologics and Novartis signed a main contract with a size that is five times larger than the previous LOI. Samsung Biologics signed a CMO contract worth 1.2 trillion won with global pharmaceutical company Pfizer on the 4th before signing a large-scale contract with Novartis. The amount of orders received from Pfizer and Novartis alone amounts to 1.7 trillion won. This is similar to last year's total order amount of 1.7835 trillion won. Samsung Biologics is increasing large-scale, long-term contracts while securing major global big pharma customers. It has secured 13 of the top 20 global pharmas as customers. Samsung Biologics is strengthening its ability to win orders based on its competitiveness in the global No. 1 production capacity (CAPA), speed, and quality. Samsung Biologics started with the 30,000-liter plant 1 in 2011 and expanded the 154,000-liter plant 2 in 2013. In 2015, a third plant with a capacity of 180,000 liters was built. Starting in 2020, construction began on the 240,000-liter class 4 plant, the world's largest single plant, and began full operation last month. The total production capacity is 604,000 liters. Samsung Biologics started construction of its 5th plant in April to preemptively respond to the market's demand for biopharmaceutical CMO. Completion is targeted for April 2025. When completed, the total production capacity will be 784,000 liters. Through process innovation, Samsung Biologics has shortened the technology transfer period, which is essential for biopharmaceutical production, to three months, half of the industry average. Process optimization was carried out through a specialized technology transfer team. The deployment success rate of Samsung Biologics is over 98%. A batch refers to a biopharmaceutical production unit. The cumulative number of regulatory approvals was 231. Samsung Biologics is strengthening its capabilities to respond to the demand for next-generation drugs such as antibody-drug conjugates (ADCs). In April of this year, it invested in Araris Biotech, an ADC treatment technology development company, through the Life Science Fund. It also plans to have an ADC production facility by 2024. Samsung Biologics plans to actively respond to the blockbuster drug market, such as Alzheimer's treatment. Based on the world's largest production capacity, it plans to expand orders by focusing on products that require mass production and products that newly expand indications. It plans to win orders for the 5th plant targeting Alzheimer's treatment, which has a lot of unmet demand. An official from Samsung Biologics said, "We have expanded our global bases, mainly in North America, to narrow the physical distance with our customers and provide prompt services." "We opened a sales office in New Jersey in March of this year following San Francisco in October 2020 to provide services to global customers," he said.
- Company
- ‘Leclaza will be free as first-line Tx until reimbursement'
- by Kim, Jin-Gu Jul 11, 2023 05:41am
- Wook-Je Cho, CEO of Yuhan Corp announced that it will be providing its anticancer drug ‘Leclaza (lasertinib)’ for free as a first-line treatment until its reimbursement is extended to cover first-line treatment. Cho announced the company’s launch of the Early Access Program (EAP) at a press conference it had held on the 10th at the Seoul Plaza Hotel to celebrate Leclaza’s approval as a first-line treatment in Korea. Leclaza is Korea's 31st homegrown novel drug that was approved for the treatment of NSCLC in January 2021. At the time of its approval, the drug was approved as a second-line treatment for patients with locally advanced or metastatic NSCLC who developed resistance after being previously treated with 1st generation or 2nd generation EGFR-TKIs. Last month, the company received additional approval for Leclaza as a ‘first-line treatment for non-small-cell lung cancer. Cho said, “The company will return a certain proportion of its profits to support the treatment of lung cancer. We will allow free access to Leclaza (as a first-line treatment) for patients suffering from lung cancer until the drug is applied reimbursement." In other words, the company will be providing Leclaza for free as a first-line treatment to all patients that wish to use the drug in the first line until the use is covered by reimbursement. Cho emphasized, “Regardless of the amount, all patients who need and want Leclaza will be able to benefit from our EAP.” Also, Cho dismissed the concerns that the EAP could violate fair competition with its competitors, saying "We feel confident because we have no alternative purpose." “Many patients with EGFR-positive NSCLC have been filing one petition after another through the National Assembly, the Ministry of Health and Welfare, and the President's Office. Many patients cannot afford to use Leclaza because the drug costs KRW 100 million a year without insurance reimbursement. After the drug was approved as a first-line treatment, we decided that it was right to provide its benefit faster to more patients.” “Carrying on the spirit of our founder, Dr.Ilhan New, we will be giving back our profits to society through the EAP. Regarding issues of compliance that have been voiced, we feel confident in providing EAP because we have no alternative purpose.”
- Company
- 'Leclaza effective as 1st-line treatment in Koreans'
- by Kim, Jin-Gu Jul 11, 2023 05:41am
- On the morning of the 10th, Professor Jin-Hyoung Kang of Medical Oncology at the Catholic University of Korea Seoul St. Mary's Hospital, said, "LASER 301,' the global Phase III clinical trial on Leclaza (lazertinib), is the only study that demonstrated the drug's efficacy as a first-line treatment for EGFR mutation-positive non-small cell lung cancer patients in Korea,” during a press conference held to celebrate Leclaza's approval as first-line treatment at Seoul Plaza Hotel. Leclaza is Korea's 31st homegrown novel drug that was approved for the treatment of NSCLC in January 2021. At the time of its approval, the drug was approved as a second-line treatment for patients with locally advanced or metastatic NSCLC who developed resistance after being previously treated with 1st generation or 2nd generation EGFR-TKIs. Last month, the company received additional approval for Leclaza as a ‘first-line treatment for non-small-cell lung cancer. During the conference, Professor Byoung-Chul Cho of Medical Oncology at the Yonsei Cancer Center explained the results of the LASER 301 Phase III clinical trial, which served as the basis for the approval of Leclaza as a first-line treatment. Among the total of 393 patients worldwide that were enrolled in the trial, 258 were Asians, and 172 of those were Korean patients. Cho emphasized that there were no significant differences between the overall results and the results in Asian patients. Professor Byoung-Chul Cho of Medical Oncology at the Yonsei Cancer CenterCho said, "The median progression-free survival (mPFS) in the Leclaza arm was 20.6 months, which is a statistically significant improvement compared with the 9.7 months in the gefitinib arm. The mPFS in Asian patients were consistent with those of the overall global patient population." “The subgroup analysis of patients with EGFR mutation subtypes also yielded noteworthy results. In patients with exon 19 deletion mutation, the mPFS in the Leclaza arm was 20.7 months, compared with the 10.9 months in the gefitinib arm. In patients with exon 21 L858R substitution mutation, the mPFS for the Leclaza arm was 17.8 months, compared with the 9.6 months in the gefitinib arm." "The results are encouraging. Leclaza demonstrated superior antitumor effect even in patients with an exon 21 L858R substitution mutation, who are known to have a relatively poorer prognosis than those with an ex19del mutation. Leclaza showed consistent results regardless of EGFR mutation subtypes.” Kang also mentioned the therapeutic benefits of Leclaza in Korean patients. He particularly highlighted the fact that although a higher proportion of patients with brain metastasis or the L858R mutation - factors known to have a poor prognosis - were present in Korean patients, the results were similar or even slightly better. Kang explained, "The LASER 301 trial included 172 Korean patients, and among them, one-third already had brain metastasis before the trial began. The investigator-assessed mPFS in the gefitinib arm was 9.6 months, compared with the 20.8 months in the Leclaza arm." Professor Jin-Hyoung Kang of Medical Oncology at the Catholic University of Korea Seoul St. Mary"These PFS results were consistently observed even in the subgroup of patients with brain metastasis. The safety data were also consistent with the previously reported clinical results." Kang noted, "Leclaza is the first treatment to demonstrate therapeutic benefits in a clinical trial that included a significant number of patients among the third-generation EGFR TKIs. The investigator-assessed mPFS of Korean patients with ex19del mutation in the Leclaza arm was approximately 2 years (23.3 months)." “The mPFS of Korean patients with exon 21 L858R substitution mutation who were treated with Leclaza was 17.8 months, compared with the 9.6 months in the gefitinib arm. Leclaza showed consistent effect regardless of type of EGFR mutation in all Korean patients, just as in the global patient population." Kang added that not enough data has been accumulated yet to determine overall survival (OS), which is another major endpoint. “Regarding the much-anticipated OS data, the follow-up period is still too short to obtain sufficient overall survival data. Only 29% of the data has been obtained so far.”
- Company
- Pricing negotiations for Spinraza's reimb begin in KOR
- by Eo, Yun-Ho Jul 11, 2023 05:41am
- The spinal muscular atrophy treatment ‘Spinraza’ has finally entered the last stage of its reimbursement extension in Korea. According to Dailypharm coverage, Biogen is undergoing drug pricing negotiations with the National Health Insurance Service for its SMA treatment Spinraza (nusinersen). Also, another SMA treatment, Roche Korea’s ‘Eveysdi (risdiplam)’ has passed the NHIS’s Drug Reimbursement Evaluation Committee review and has started pricing negotiations almost at the same time with Spinraza. Discussions on the reimbursement and reimbursement extension of SMA treatments had seemingly made rapid progress due to rising demand for the abolition of Spinraza’s discontinuation standards, however, the discussions became prolonged as the requested range of reimbursement was broader than what the government and pharmaceutical companies had expected. Spinraza is currently reimbursed for the treatment of 5q Spinal Muscular Atrophy in patients that satisfy all of the following conditions: ▲received genetic diagnosis of 5q SMN-1 deficiency or mutation; ▲had onset of SMA-related clinical symptoms and signs at the age of 3 or less; and ▲ are not using permanent ventilators. In other words, only patients whose symptoms have been found before the age of 3 were allowed reimbursement until now. The reimbursement extensions currently under discussion aim to expand that age limit to ‘the age of 18 or less.’ At the time, the government had planned to proceed with discussions on the reimbursement listing of Evrysdi after agreeing on the scope of extended reimbursement for Spinraza, and both drugs entered into final negotiations. Whether the SMA drugs that have now entered the last hurdle to their reimbursement in Korea after various twists and turns, will be able to reach the finish line remains to be seen. The SMA treatments currently available in Korea with reimbursement are as follows: Spinraza, and Novartis Korea’s one-shot treatment ‘Zolgensma (onasemnogene abeparvovec-xioi).’ Spinraza is injected using a syringe directly into the cerebrospinal fluid (CSF), and Zolgensma is injected as an intravenous infusion. Evrysdi, which is attempting to be newly reimbursed, is an oral formulation that can reduce the socioeconomic burden borne by SMA patients and their caregivers such as school/work interruption, transportation costs, and nursing care that accompany the required injections. Also, Evrysdi can reduce drug costs for infant patients through customized prescriptions by age and weight.
- Company
- Yuhan provides Leclaza with 6 million won per month for free
- by Kim, Jin-Gu Jul 11, 2023 05:40am
- Leclaza Regarding Yuhan's providing free medicines to patients until the first treatment of Leclaza is covered by insurance, Yuhan said, "It is not an act of inducing patients and there is no problem under the Fair Trade Act." On the 10th, Yuhan held a press conference for permission for the first treatment of Leclaza at the Plaza Hotel in Seoul. On this day, CEO Cho Wook-je announced the free supply of Leclaza through the Expanded Access Program. Yuhan Corporation launches large-scale 'EAP' for the first time as a domestic pharmaceutical company EAP is a humanitarian system that provides treatment opportunities by supplying new drugs free of charge to patients who do not have adequate treatment. Usually, instead of patients participating in clinical trials, new drugs are administered more rapidly. According to Article 34 of the Pharmaceutical Affairs Act, 'drugs for clinical trials shall not be used for purposes other than clinical trials. However, if the approval of the Minister of the Ministry of Food and Drug Safety falls under each of the following subparagraphs, the relevant drug may be used for purposes other than clinical trials.' At this time, the exceptions are limited to 'patients with life-threatening serious diseases such as terminal cancer or acquired immunodeficiency syndrome' or 'emergency patients prescribed by Prime Minister Ordinance, such as life is in jeopardy or there is no alternative treatment method'. Based on this clause, Yuhan Corporation's plan is to supply Leclaza's first-line clinical trial drug to patients. If the clinical trial for the first treatment of Leclaza is conducted in the form of a researcher's clinical trial, Leclaza will be supplied to the participating patients. Yuhan had already operated EAP in the same way until Leclaza was approved as a second-line treatment and reimbursement was applied. In the pharmaceutical industry, Leclaza's EAP operation is seen as the first attempt among domestic pharmaceutical companies. This is because it is the first drug developed by a domestic company for an expensive and life-threatening treatment such as cancer. Cho Wook-je, CEO of Yuhan Corporation, said, "Because the drug is so expensive, there are many people who find it difficult to use Leclaza due to economic conditions. We will provide it free of charge through EAP until Leclaza is officially covered by reimbursement." The scale will be determined upon request from medical staff treating lung cancer on site.” "Tagrisso and Leclaza both use EAP as non-reimbursed drugs, and there is no problem with fair competition" Some criticize Yuhan's plan to use Leclaza for sympathy, saying that it violates fair competition. It is criticized that, as in the case of Novartis Glivec in the past, support for drug prices through EAP can be seen as an act of luring patients. Regarding this, Yuhan Corporation said, “It is different from the Glivec case,” and drew a line, saying, “We operated the same program until the benefit was applied after the second treatment was approved, but there was no problem.” Gleevec In 2013, Novartis stopped subsidizing the cost of Glivec, a leukemia treatment. Until just before that, Novartis subsidized 5% of the drug cost for patients taking Glivec. Since 95% of the cost of leukemia treatment was supported by health insurance finances, patients took Glivec virtually free. In June 2013, when the release of a large number of generic products was announced ahead of the expiration of the Glivec patent, Novartis decided to stop supporting the drug price. It is based on the judgment that supporting the price of Glivec even though a generic with the same ingredients has been released can be considered an act of attracting patients and can constitute an unfair trade act under the Fair Trade Act. If the competitive drug Tagrisso is covered, it is likely to be seen as an act of attracting patients, but since both drugs are not covered by insurance as the first-line treatment, there is no problem under the Fair Trade Act. An official from Yuhan Corporation said, "We understand that supporting the cost of medicines with the benefit applied is against fair competition. The case of Gleevec falls under this category." Once the reimbursement is decided, the free provision ends immediately. At this time, the patient has to pay all of his or her own expenses.” The official said, "The monthly cost of Leclaza alone is about 6 million won. According to the PFS results, if the drug is administered continuously for 20 months, the amount that the patient will have to bear will exceed 120 million won." It is purely a decision for the patient. From the company or doctor's point of view, there is nothing to take 10 won." "There will be more participants than in the second treatment... Lexraza is provided without size restrictions" Yuhan expects that the number of patients participating in EAP related to the first treatment will increase compared to the second treatment. According to Yuhan Corporation, around 10 patients participated in EAP after Leclaza's second-line treatment was approved and until insurance coverage was applied. It is explained that the number of participants was not large because the process from approval to reimbursement was fast and the procedure for participating in EAP was complicated. The key is when the primary treatment benefit is applied. The longer it takes for coverage of Leclaza to expand as a first-line treatment, the more patients are expected to participate in the compassionate use program. Yuhan plans to provide Leclaza in any size, no matter how long it takes to get paid. An official from Yuhan Corporation said, "It is impossible to estimate how many patients will participate in the program. Therefore, it is currently unknown how much the related budget will be. However, we expect more patients to participate than in the second treatment." AstraZeneca, which has a competitive drug, seems to refrain from the official response. However, AstraZeneca Korea explained that it is not considering a separate legal response against Yuhan Corporation. An official from AstraZeneca Korea said, “We are doing our best to increase the accessibility of patients who use Tagrisso in Korea,” and “We are reviewing ways to work for patients.”
- Company
- Will the non-reimbursed Gavreto vanish in Korea?
- by Jung, Sae-Im Jul 10, 2023 05:21am
- The possibility that the new RET-targeted anticancer therapy ‘Gavreto (pralsetinib)’ will vanish without ever being released in Korea has been rising. With Roche giving up sales rights for Gavreto, the prospects of its reimbursement listing in Korea had also gone up in smoke. As Blueprint Medicines, the original developer of Gavreto, does not have a branch in Korea, the company would need to find a new partner to market the drug in Korea, which is more easily said than done. According to the Health Insurance Review and Assessment Service, the Drug Reimbursement Evaluation Committee determined Gavreto inadequate for reimbursement and made a ‘non-reimbursement’ at its 7th 2023 DREC meeting that was held on the 6th. Drugs that receive a non-reimbursement decision, not a conditional pass or re-discussion decision, must reapply for reimbursement with supplementary materials. As such drugs need to undergo all the steps for reimbursement again, the time to their reimbursement listing becomes delayed indefinitely. Also, it is unclear which pharmaceutical company will reapply for Gavreto’s reimbursement in the future. Before, Roche Korea was in charge of its approval and distribution in Korea. Roche had paid $775 million upfront for Gavreto’s global sales right in all countries excluding China. However, in March, only 2 years and 7 months after signing the agreement, the company returned the rights for Gavreto to Blueprint due to lower sales expectations. With the termination of the collaboration, Roche will be supplying Gavreto until February next year. To continue supply, the original developer Blueprint Medicines will need to directly supply the drug or find a new partner. Without its own Korean subsidiary, the realistic alternative for Blueprints would be to resume supply through a new partner. So far, there has been no word on Gavreto’s new partner. Roche Korea submitted an application for the reimbursement of Gavreto in November last year. However, its discussions fell through after the global termination of the collaboration was announced ahead of its CDDC review. Roche Korea was unable to continue the drug pricing negotiations for the drug in a situation where it was unclear which company would continue marketing the drug. As a result, Gavreto’s reimbursement standards were not set at the CDDC meeting, resulting in its non-reimbursement by DREC. The market conditions are also working against Gavreto. Its competitor, Lilly's 'Retevmo (selpercatinib)', passed DREC review in May and is set to be reimbursed soon. The reimbursement listing timing for Retevmo and Gavreto, both of which received domestic approval in the same month, widened significantly in the reimbursement listing process. Unlike Retevmo, which passed the Drug Committees (DCs) of major general hospitals in January of this year, Gavreto was unable to even be registered for prescriptions. At this rate, it is highly likely that Retevmo will dominate the domestic RET mutation-targeting therapy market. In addition, Gavreto recently voluntarily withdrew its thyroid cancer indication in the U.S. With Retevmo already owning the indication, Gavreto lost the opportunity to extend its indication to the area. This signifies the lower profit expected for Gavreto in Korea. Previously, sales of Retevmo surpassed Gabretto in global sales, reducing the sales expectations for Gavreto. It seems that it will not be easy to find a pharmaceutical company that will sell Gavreto in this situation, as the patient group is small, sales expectations are low, and reapplications need to be made for its reimbursement. If Gavreto goes through the withdrawal process at this pace, Retevmo will become the only targeted anti-cancer drug that can treat RET mutation-positive cancer in Korea. RET is one of the major emerging biomarkers that induce cancer. It causes malignant tumors through fusion mutations or point mutations. RET mutations are partially found in several cancer types, including lung cancer, breast cancer, and colorectal cancer. The rate of RET mutations in non-small cell lung cancer is about 2 to 6%, and RET fusion mutations have been reported in up to 40% of thyroid cancers.