- LOGIN
- MemberShip
- 2026-05-02 21:37:53
- Company
- Dong-A ST’s biosimilar first enters the U.S. market
- by Chon, Seung-Hyun Oct 14, 2024 05:49am
- Dong-A S.T. has succeeded in entering the US market 13 years after declaring its entry into the biosimilar business. It has received approval in the US for a biosimilar version of Stelara, which owns a global market worth KRW 14 trillion. It was the company’s second overseas market with a self-developed biosimilar. The biosimilar's entry into the U.S. market secured additional technology fee revenues and added a new item for the company. Pic of Dong-A ST headquarters According to industry sources, Dong-A ST's Stelara biosimilar ‘Imuldosa’ was approved by the US Food and Drug Administration (FDA) on the 12th. Stelara is a treatment for inflammatory diseases such as plaque psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis. It is a high-grossing product that generated more than KRW 14 trillion in global sales last year. Dong-A ST is the second Korean company to receive approval for a Stelara biosimilar in the US following Samsung Bioepis. Dong-A ST passed the US market gateway 11 years after it started developing its Stelara biosimilar in 2013. Dong-A Socio Holdings began codeveloping Imuldosa with Japan's Meiji Seika Pharma in 2013. In July 2020, the development and commercialization rights were transferred to Dong-A ST for efficient execution of the global project. From 2021 to the end of 2022, Dong-A ST conducted a global Phase III clinical trial of Imuldosa in 9 countries including the United States, Poland, and Estonia, on a total of 605 patients. Trial results confirmed that Imuldosa’s efficacy and safety was equivalent to is equivalent to Stelara for marketing authorization by the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) Imuldosa's FDA approval is expected to generate additional technology fees. In July 2021, Dong-A ST signed a technology export agreement with the multinational pharmaceutical company Intas Pharmaceutical to license out Imuldosa. Intas paid a non-refundable upfront payment of USD 10 million. The payments based on clinical, licensing, and sales milestones were set at up to USD 95 million. Royalties on product sales are guaranteed to be at least 10%. In addition, payments from Intas will be shared by Dong-A ST, Dong-A Socio Holdings, and Meiji Seika Pharma. Intas has secured exclusive rights to license and market the product globally, excluding South Korea, Japan, and select Asian countries. Intas is an Indian multinational pharmaceutical company with a global sales network in more than 85 countries. Global commercialization will be handled by Accord Healthcare, a subsidiary of Intas Pharmaceuticals. Accord Healthcare submitted a marketing authorization application for Imuldosa to the European Medicines Agency (EMA) in June last year and submitted an application to the FDA in October last year. By the end of the first half of last year, Dong-A ST had recognized KRW 41 billion as development costs for Imuldosa as an intangible asset. The Financial Supervisory Service has identified Phase III clinical trials as the stage at which new drug R&D costs can be capitalized. This means that KRW 41 billion was spent on global Phase III clinical trials of Imuldosa. Dong-A ST announced its entry into the biosimilar business in 2011. In 2011, Dong-A Socio Holdings established DM Bio with an investment of KRW 57 billion from Meiji Seika Pharma to build a biopharmaceutical plant. In March 2015, Dong-A Socio Holdings spun off DM Bio into a 100% subsidiary and transferred 49% of its shares to Meiji Seika Pharma. In 2021, Dong-A Socio Holdings acquired 1,117,200 shares of DM Bio from Meiji Seika Pharma for KRW 42.1 billion. The company's stake in DM Bio increased to 80.4% as it took over 60% of the 1.86 million DM Bio’s shares held by Meiji Seika Pharma. In 2022, DM Bio changed its name to STgen Bio. STgen Bio will be responsible for the production of Imuldosa. Imuldosa is the second biosimilar product that Dong-A ST has successfully commercialized in overseas markets. Dong-A ST sells Nesp's biosimilar version, ‘darbepoetin alfa’ in Japan. Dong-A ST conducted Phase I clinical trials of darbepoetin alfa and transferred the development and sales rights in Japan to Sanwa Kagaku Kenkyusho (SKK) in January 2014. Based on the results of a local Phase III clinical trial comparing the original Nesp with darbepoetin alfa, SKK received marketing authorization from the Japanese Ministry of Health, Labour and Welfare in September 2019, and launched the drug in late November of the same year. Dong-A ST will export the finished product manufactured on consignment by STgen Bio to SKK, and SKK is in charge of local sales. Last year, Dong-A ST recorded 20.6 billion won in exports with its dabepoetin alfa.
- Company
- ERT demonstrates effectiveness in treating Fabry disease
- by Whang, byung-woo Oct 11, 2024 06:25am
- "'It has been 20 years since ERT treatments became available for patients with Fabry disease. More treatment options are available to treat Fabry disease, but selecting treatment options and strategy requires precise attention based on research data." Analysis suggests that the treatment environment for rare diseases has improved due to the emergence of Enzyme Replacement Therapy (ERT) to treat lysosomal storage disease (LSD), such as Fabry disease. New treatment strategies and research for treatments are being conducted to complement the limitations of ERTs. For example, studies on treatments like Substrate Reduction Therapy (SRT) are being conducted. Antonio Pisani, a professor in the Department of Nephrology at the University of Naples Federico II. However, considering the nature of rare diseases, using available treatment resources to the fullest remains equally important. Antonio Pisani, a professor in the Department of Nephrology at the University of Naples Federico II, has emphasized the importance of monitoring when switching between medicines for treating Fabry disease during a meeting with Daily Pharm. A study comparing ERT→oral drug switching…disease management through monitoring is essential During the WORLD Symposium 2024, held in San Diego, USA, in February, a study result was presented that close monitoring for medication switching is essential. It is because after a patient switch from Fabrazyme to oral drugs, a long-term change in treatment outcomes has been reported. The study is expected to aid in selecting treatments as it compares the efficacy and safety of the ERT treatment, Fabrazyme, to an oral drug, migalastat, using registry data on patients with Fabry disease. "Migalastat has been approved in Europe in 2016. However, the use of the drug has been limited because there is no real-world evidence (RWE) besides pre-clinical or clinical results," Professor Pisani, who conducted the research, said, "The study analyzed data of patients who had been treated with Fabrazyme over a year and then switched to migalasta and continued treatment over six months. It confirmed various biomarkers for Fabry disease, changes to patient symptoms, whether a patient reached first-line treatment goal and clinical symptoms." Based on research results, migalastat treatment, compared to Fabrazyme, worsened an estimated glomerular filtration rate (GFR), a biomarker for Fabry disease, and GL-3 level, a glycolipid in cells. Additionally, a classical-type patient with Fabry disease who switched to migalastat had worsened UPCR and heart index, demonstrating that Fabrazyme is much more effective in treating patients with Fabry disease. "The study confirmed that migalastat has variability in efficacy in a clinical setting," Professor Pisani said, "We concluded that a close monitoring and patient follow-up is important when a patient has been switched to migalastat following Fabrazyme treatment." "A close monitoring of enzyme activation and changes to clinical symptoms is necessary. A change in enzyme activation is expected once a patient starts taking migalastat. Therefore, medication switching requires patient follow-up," Professor Pisani emphasized. Migalastat is approved in South Korea as a long-term treatment for patients over 12 years old diagnosed with Fabry disease with a gene variant. An oral drug can be reimbursed for second-line treatment when a patient has used ERT for over 12 months in a first-line treatment or cannot use ERT. If the study results were to apply to Korea, what could be the possible analysis? Professor Pisani advises that physicians must carefully consider medication switching based on treatment guidelines for Fabry disease. "There are studies implicating positive efficacy of migalastat. However, there are opposite results," Professor Pisani said, "When treating with migalastat, physicians must monitor patients by checking biomarkers, such as eGRF and proteinuria, to confirm the efficacy of the use." Treatment outcomes of migalastat may vary depending on the patient's condition. For example, a classical-type patient with Fabry disease may have a stable or worsened heart-related index upon switching medication to migalastat, but worsening renal functions may accelerate significantly. "In the end, a follow-up of patients is essential, and it is important to closely monitor symptoms and disease progression of patients with poor baseline indexes," Professor Pisani said. Still there are limitations to Fabry disease treatment…what are unmet needs? New Fabry disease treatments are available, and real-world data have been presented. Although these improvements have been made for patients with Fabry disease, there are still unmet needs. Professor Pisani highly values the benefits brought by ERT treatments to patients with Fabry disease. Yet, he emphasizes the need for new drug development. Studies being conducted related to new treatment strategies and drugs to complement ERT treatment limitations. For example, studies on substrate reduction therapy and gene therapy are being conducted. "We can consider possibilities, such as treatment development, including new ERT and chaperone, a combination therapy containing chaperone, or Fabrazyme in combination with gene therapy," Professor Pisani said. "We can approach Fabry disease with new treatments, such as SRT, as it is a lysosomal storage disorder." Yet, new treatment development takes time. Therefore, a strategic approach is necessary using known research results. "There are several possibilities for Fabry disease treatment. Physicians should now plan treatment option selection and strategy using research data," Professor Pisani said. "The dosage of treatment is an important factor for Fabry disease. A sufficient dosage administration can delay the disease progression." "I would like to emphasize the importance of early treatment through early diagnosis. Early treatment can delay the disease progression and worsening," Professor Pisani emphasizes.
- Company
- ‘Switching between AD drugs’ discussed at NA audit
- by Whang, byung-woo Oct 11, 2024 05:54am
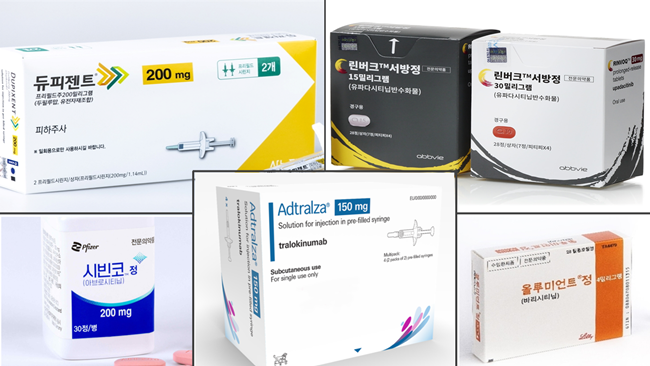
- The issue of switching between medications for severe atopic dermatitis, whose need had been voiced constantly in the clinical field, has been mentioned during the National Assembly Audit, attracting attention to whether the reimbursement environment will change. (Clockwise from top left) Pic of Dupixent, RInvoq, Olumiant, Adtralza, Civinqo During the National Audit of the National Assembly's Health and Welfare Committee on the 8th of this month, Democratic Party Rep Jin-Suk Jeon asked Minister of Health and Welfare Kyoo-Hong Cho about the issue of switching between medication for atopic dermatitis. Currently, the special calculation and reimbursement standards for switching between medications in severe atopic dermatitis stipulate that ‘starting treatment with one of the biologics or JAK inhibitors and then changing to another’ is not eligible for special calculation and reimbursement. However, experts have consistently expressed the opinion that atopic dermatitis is a disease that features various characteristics in each patient, which is why it is necessary to switch between treatments for personalized treatment. The Korean Atopic Dermatitis Association’s ‘2024 Korean Atopic Dermatitis Treatment Guidelines’ which was revised after 9 years, also made specific recommendations to address this issue of switching. The association’s position is that patients should be able to switch between medicines regardless of the line of therapy or class of drug, without having to decide which drug is more appropriate, as it is difficult to be sure of the decision. “Patients cannot switch from one drug to another even through other options are available,” said Jo-Eun Park, CEO of the Severe Atopic Dermatitis, Association who attended the meeting as a witness. “The government does not apply health insurance and special calculation cases when switching drugs, so patients have to pay for up to KRW 17 million on drugs per year if they switch to another drug.” “Even when a new drug is released, the only drug available to patients is the first drug they choose. If they want to change their medication due to severe side effects or ineffectiveness, they will have to stop treatment and worsen their condition in order to meet the criteria for reimbursement and special calculation of the other drug.” Park also pointed out that patients with psoriasis, a skin condition similar to severe atopic dermatitis, can switch to different medications when they are ineffective or have side effects, which shows the disparity in the treatment environment of the two. In this regard, Rep Jeon asked Minister Cho to review the need for switching between severe atopic dermatitis drugs and prepare measures. Minister Cho said, ‘I understand that switching between drugs for atopic dermatitis is an important issue, and I think the evidence is accumulating now. I was told that the Health Insurance Review and Assessment Service will actively review the issue.” Despite the positive response, the key to its outcome may depend on the budget budget, including Korea’s health insurance finances. In fact, the notification of the amendment related to the recognition of insurance reimbursement benefits for switching between JAK inhibitors in rheumatoid arthritis, which was scheduled to be implemented in October, has been put on hold. According to the industry, the delay is due to budgetary issues. The government is likely to face similar challenges to rheumatoid arthritis, as the core issue in atopy dermatitis is also focused on switching between biologics and JAK inhibitors. According to the drug market research institution UBIST, the outpatient prescription market for JAK inhibitors reached KRW 27.5 billion in the first half of last year. This is a 54% increase from the KRW 17.8 billion made in the first half of last year.
- Policy
- MFDS Minister ‘will work with MOHW on INN prescriptions’
- by Lee, Hye-Kyung Oct 11, 2024 05:54am
- International non-proprietary names (INNs) and ingredient-based prescriptions should be introduced to prevent patients from circling pharmacies due to drug shortages, said a lawmaker. ‘Institutional inducements such as the introduction of INNs and ingredient-name prescriptions are needed to prevent drug supply disruptions and pharmacy bouncing issues from occurring again,’ said Representative Young-Seok Seo (Democratic Party of Korea) during the National Assembly Health and Welfare Committee's NA audit of the Ministry of Food and Drug Safety “According to a recent survey, the public's trust in generic quality is about 50%, but 80% responded that they will take generic versions if doctors prescribe them,” said Rep. Seo, adding, “There seems to be low awareness of the bioequivalence of generics drugs and original drugs, but hasn't the MFDS recommended making International Nonproprietary Names (INN) prescriptions?” Regarding the inquiry, Yu-Kyoung Oh, Minister of Food and Drug Safety responded, “The Ministry of Health and Welfare is the main ministry in charge of INN prescriptions, and the MFDS once said that we would cooperate if the Ministry of Health and Welfare takes initiative.” Oh added, “We can say that an effect of a drug is equivalent if we conduct bioequivalence tests. In the case of INN prescriptions, the MFDS will be able to cooperate if the MOHW initiates the discussion.” It was also pointed out that the public-private consultative body for the unstable supply and demand of drugs, which involves both the MOHW and the MFDS, is not working organically due to the disconnect between ministries. “I wonder if the disconnect between the ministries is aggravating the supply and demand instability. Why should there be a shortage of medicines when there is a consultative body ready?’ Seo asked, stressing that ’inter-ministerial discussions are needed to minimize the disconnect.’ In response, Oh said, “The MFDS receives reports of supply shortages from pharmaceutical companies, and the MOHW receives reports from pharmacies and hospitals, so the report process is different. So we create a system when we meet as part of the public-private consultative body, but as there are various causes of supply and demand disruptions, we plan to discuss them in depth at the consultative body’s meeting.”
- Opinion
- [Reporter's view] Creating a virtuous cycle for K-bio growth
- by Son, Hyung Min Oct 11, 2024 05:54am
- The research and development (R&D) trend in the biopharmaceutical industry is focusing on new anticancer drugs. The field is gaining interest, especially following the clinical success of Yuhan Corp's Leclaza (ingredient: Lazertinib) and HLB's rivoceranib. In particular, most biotech companies in South Korea are confirming the clinical potential of drugs in combination with Keytruda. Keytruda is an immune checkpoint inhibitor developed by MSD and a successful blockbuster medicine with over 30 indications in South Korea alone. Keytruda's global sales this year are expected to amount to KRW 3.5 billion. Almost all biotech companies in South Korea aim to out-license their technologies to MSD through clinical trials of combination therapy with Keytruda. A combination therapy involving medicine with proven effects is highly likely to be commercialized. However, R&D in the Korean industry seems to be focusing only on anticancer drugs for the purpose of out-licensing them to a big global pharmaceutical company. To date, several biopharmaceutical companies in South Korea are reporting confirmation of the efficacy of their drugs in combination with Keytruda through preclinical trials or early-stage clinical trials. However, there have been no reports of entering late-stage clinical trials, thereby nearing commercialization, or of outstanding out-licensing. In addition to Keytruda, the next target of the biopharmaceutical industry in South Korea is Enhertu. A second-generation antibody-drug conjugate, Enhertu (ingredient: trastuzumab deruxtecan), has demonstrated efficacy and is effective for breast cancer, gastric cancer, non-small cell lung cancer, and colorectal cancer. The biopharmaceutical industry in South Korea is exploring the market for ADC anticancer drugs. Even the long-standing pharmaceutical companies and the biotech industry have shown interest in ADC development. Several companies have already begun clinical trials involving combination therapy with Enhertu. It is common for Korean companies to pursue out-licensing as it allows them to secure funds for investments in other pipelines through out-licensing and provides opportunity to discover new drugs. However, if the industry continues to chase after blockbuster new anticancer drugs, it will lead to a difficult system to develop new innovative drugs domestically. Consequently, companies must explore new fields besides anticancer drugs. Instead of pursuing a partner in a combination therapy involving a well-selling drug, they should explore a field with unmet needs to create various out-licensing opportunities. The R&D costs in the biopharmaceutical industry in South Korea are incomparably smaller than those of global pharmaceutical companies. A stable source of funds is essential to sustain a virtuous cycle. Running a company by continuously developing competitive new drug candidates and successfully out-licensing is almost impossible. As a result, it is important to have experience collaborating with other companies to secure Korean and Asian rights when discussing out-licensing agreements with a global pharmaceutical company. Pharmaceutical companies can only sustain stable earnings through selling drugs. The biopharmaceutical industry is one of the industries with a large gap between Korean and global companies. The biopharmaceutical industry cannot benefit from good deals because it cannot produce good products and sell them cheaply. Now, the biopharmaceutical industry in South Korea must consider challenging various fields and creating a system to profit from selling new drugs. Clinical trials involving combination therapy containing new drugs are already supersaturated.
- Company
- Will Tevimbra be reimbursed promptly in Korea?
- by Eo, Yun-Ho Oct 11, 2024 05:54am
- Whether an immuno-oncology treatment option will arise in the oesophageal cancer space is garnering attention. BeiGene Korea's immuno-oncology drug Tevimbra (tislelizumab) passed the Health Insurance Review and Assessment Service’s Cancer Disease Review Committee in August during its second attempt. Tevimbra is a PD-1 inhibiting immuno-oncology drug that has demonstrated clinical utility in second-line oesophageal squamous cell carcinoma and was approved in Korea in November last year. Currently, there are seven immuno-oncology drugs licensed and marketed in Korea, including ▲Keytruda, ▲Opdivo, ▲Tecentriq, ▲Imfinai, ▲Bavencio, ▲Jemperli, and ▲Tevimbra, which owns a total of 64 indications. However, only 21 indications (around 33%) are currently reimbursed. Also, none of these drugs are yet reimbursed for esophageal cancer. Currently, only platinum-based chemotherapy is reimbursed in Korea as both first-line and second-line treatment options for oesophageal squamous cell carcinoma. Reimbursement rates for immuno-oncology drugs by indication, including oesophageal cancer, are low due to high drug costs and finances. After being reimbursed for some cancers, such as lung cancer, overall claims for immuno-oncology drugs and their share of health insurance expenditures have increased significantly, leading to an increased burden on national health insurance finances. As of 2023, the total claims for anticancer drugs was around KRW 2.4 trillion, with immuno-oncology drugs accounting for about KRW 500 billion, or 20% of the total claims for anticancer drugs. This is why BeiGene plan to supply Tevimbra at a relatively low price is raising expectations. BeiGene’s corporate philosophy of providing ‘innovative new medicines at reasonable prices’ to eliminate underserved patients, has already been demonstrated through the reimbursement process it had shown with its blood cancer drug ‘Brukinsa (zanubrutinib).’ The company has also made Tevimbra available at no cost to select patients with oesophageal cancer through its Expanded Access Program (EAP). It remains to be seen if Tevimbra will make it onto the reimbursement list after passing discussions with the Pharmacoeconomic Evaluation Subcommittee, Risk Sharing Subcommittee, and Drug Reimbursement Evaluation Committee. In the global Phase III RATIONALE-302 study, Tevimbra prolonged median overall survival (OS) by 2.3 months (8.6 months vs. 6.3 months) compared to chemotherapy, resulting in a statistically significant 30% reduction in the risk of death. Compared to chemotherapy, Tevimbra resulted in more than twice as many patients responding to treatment (20% vs. 10%), and showed an improvement in the median duration of response of approximately 3 months, from 4.0 months to 7.1 months, with sustained responses and a reduction in tumor size, which is directly related to quality of life for esophageal cancer patients. As a result, the U.S. National Comprehensive Cancer Network (NCCN) revised its guidelines to recommend Tevimbra as a Category 1 preferred option for second-line treatment of esophageal squamous cell carcinoma.
- Company
- SK bioscience invests KRW 4.1B stake in a U.S. biotech
- by Kim, Jin-Gu Oct 10, 2024 05:51am
- SK bioscience announced on October 8th that the company has signed an agreement to acquire a stake in U.S. biotech Fina Biosolutions by investing USD 3 million (approximately KRW 4.1 billion). SK bioscience became the first and only strategic investor of Fina Biosolutions. According to the contract between both companies, the specific stake acquisition size remains undisclosed. According to SK bioscience, Fina Biosolutions, established in 2006, has the core technology of conjugate vaccines for pneumoniae, meningococcus and typhoid. Fina Biosolutions is known to manufacture 'CRM197 (cross-reacting material),' a protein carrier important for the conjugate vaccine development, and produce it in high yield. CRM197 conjugates to antigens that induce infectious disease prevention and enhance immune responses. Fina Biosolutions has developed 'EcoCRM' to enhance productivity, which is higher than that for CRM197, using its in-house expression system and filtration technology. Additionally, the company is researching to increase immunogenicity and productivity using the next-generation CRM197 technology, which targets an antigen-binding site. Fina Biosolutions provides various carrier proteins, including CRM197, in collaboration with various biotech companies and agencies, including the U.S. Inventprise and companies in China and India. Furthermore, the company is expanding its business by providing antigen·dextran (carbohydrate-derived polysaccharides) conjugate service using its specialty in conjugate technology. SK bioscience, which owns conjugate vaccines for pneumoniae·typhoid, plans to utilize Fina Biosolutions' CRM197 technology. The company aims to increase profitability by securing the high preventative benefits of conjugate vaccines and high-yield production. "We appreciate the confidence shown by SK bioscience," Fina Biosolutions CEO Andrew Lees said. "With the investment agreement, we anticipate acceleration of global commercialization and conjugate vaccine development." "We are pleased to continuously pursue the partnership opportunities with global companies that have next-generation vaccine technologies," Jaeyong Ahn, CEO of SK bioscience. "Thorugh long-term collaboration with Fina Biosolutions, we will enhance the quality of vaccines in development and enhance our competitiveness for global entry." SK bioscience has recently made active investments to create a synergy with global companies with technologies. The company also signed an agreement to acquire the business rights of a German company, 'IDT Biologica,' which ranked among the top 10 global vaccine CMOs in June and wrapped up the acquisition process earlier this month. In June SK bioscience conditionally acquired a stake in a U.S. biotech company, 'Sunflower Therapeutics.' Sunflower Therapeutics has its own 'yeast-based culturing system' required for antigen·antibody development. "We plan to continue securing competitiveness by making investments and M&As in companies with superior technologies to establish steppingstones as a global company," SK bioscience personnel said.
- Company
- Who will distribute Mounjaro in Korea?
- by Whang, byung-woo Oct 10, 2024 05:50am
- As Novo Nordisk's obesity drug Wegovy (semaglutide) is set to be released, the industry’s interest is on which domestic distributor Lilly will choose for its Mounjaro (tirzepatide). 마운자로 제품사진. According to industry sources on the 8th, Boryung Pharmaceutical and Chong Kun Dang are in the running to become domestic distributors of Wegovy’s rival obesity drug Mounjaro. Zuellig Pharma Korea, which handled Novo Nordisk's existing obesity drug Saxenda, was announced as the domestic distributor of Wegovy. The supply price is set at KRW 370,000 for 4 weeks’ worth. The reason why the competition amongst domestic distributors for Mounjaro is so interesting is the sales potential of obesity drugs. Due to its global craze, its sales are likely to have a positive impact on the sales of domestic distributors as well. In 2023, global sales of Wegovy reached USD 4.5 billion (about KRW 6 trillion), while Zepbound (the U.S. brand name for the Mounjaro’s obesity indication), which was launched in Q4 last year, generated sales of UDS 176 million (KRW 240 billion). In June last year, Mounjaro received approval as an adjunct for chronic weight management, about a year after it was approved for type 2 diabetes. In the SURMOUNT-1 and SURMOUNT-2 trials which became the basis of approval, all doses showed statistically significant weight loss results compared to placebo, and the rate of achieving weight loss of 5% or more was also higher in the Mounjaro arm than in the placebo arm. Lilly’s decision to add the obesity indication to its existing product rather than file a new drug application is also expected to accelerate its launch. The industry is currently expecting a May launch next year. The first pharmaceutical company being considered as a possible distributor for Mounjaro is Boryung. It has a distribution agreement for Lilly's diabetes drug Trulicity and therefore is expected to have an advantage in Mounjar, which is a similar drug. In addition, Boryung has acquired the domestic rights to the patent-expired cancer drugs Gemzar (gemcitabine) and Alimta (pemetrexed) and the schizophrenia drug Zyprexa (olanzapine) from Eli Lilly in 2020-2022. ‘We have been in charge of Trulicity’s distribution since 2016 after signing a sales agreement with Lilly. However, signing a new drug partnership is a completely different matter, therefore it is difficult to confirm any facts,’ said a Boryung official. Chong Kun Dang, which has experience in distributing obesity drugs, is also in the running. Currently, Chong Kun Dang is the co-promoter and distributor of Alvogen's obesity drug Qsymia. Qsymia was approved in the US in 2012 for the treatment of obesity in adults. It is a combination of phentermine, a short-term appetite suppressant, and topiramate, a neurotherapeutic agent. It was introduced in South Korea in late 2019. “From Lilly's point of view, experience in distributing obesity drugs will be important when choosing a domestic distributor. I think we are being considered because we have experience in distributing the obesity drug Qsymia,” said a Chong Kung Dang official.
- Policy
- "Preferential drug pricing excludes K-made new drug benefits
- by Lee, Jeong-Hwan Oct 10, 2024 05:50am
- The government's revised plan for the drug pricing system has been criticized for reverse discrimination against Korean pharmaceutical companies because it was primarily designed to favor multinational pharmaceutical companies. The criticism concerns the revised drug pricing system plan, announced by the Health Insurance Review and Assessment Service (HIRA) in August, that the plan excludes the core measure in favor of Korean pharmaceutical companies. Baek Jong-heon, a member of the People Power Party, questioned this during the National Assembly's audit of the Ministry of Health and Welfare (MOHW) on October 8th. In December 2023, the MOHW reported to the Health Insurance Policy Review Committee regarding potential improvements to the drug pricing system to reflect the new drug's innovative value and the nation's healthcare security. In February 2024, the MOHW announced the expansion of preferential drug pricing criteria for pharmaceutical companies as part of the 2nd comprehensive National Health Insurance Plan 2024. The MOHW's plan was reviewed by the Drug Reimbursement Evaluation Committee (DREC), and the MHOW announced in August a revision to the specific evaluation criteria for pharmaceuticals during the negotiations process. However, Baek pointed out that the plan excludes 'Preferential drug pricing for new drugs developed by pharmaceutical companies with high R&D contributions' and 'Improved measures to calculate drug pricing to support domestically produced new drug exports.' Because of the announcement of a revised plan for preferential drug pricing, excluding measures to provide benefits to Korea-made new drugs, Jeil Pharm's new drug developed by Onconic Therapeutics, listed in September, suffered reduced export pricing. Document prepared by Baek Jong-heon, a member of the People Power Party. If the plan had been revised according to the Health Insurance Policy Review Committee report, the drug could have been listed under 'essential reimbursement pricing.' However, due to delays in drug pricing system improvement, the company did not receive the benefit. Baek questioned Cho Kyoo-hong, Minister of Health and Welfare, about the rationale for excluding a measure that provides practical benefits to Korean pharmaceutical companies from the revised plan, despite Korean pharmaceutical companies requesting improvements to the current policy related to new drug development and exports. "As part of the 120th national agenda, the Yoon Suk Yeol government has decided to foster bio-health business as the core export business, aiming to leap as a leading global country in the bio·digital health field," Baek said. "The MOHW announced the revised plan without taking into account practical supporting measures." "MOHW's role is to lead the growth and development of the Korean biopharmaceutical industry and enhance international competitiveness, thereby contributing to the nation's health and welfare improvement," Beak emphasized.
- Opinion
- [Reporter's View]Industry expectations rise with new changes
- by Eo, Yun-Ho Oct 10, 2024 05:50am
- The criteria for innovative new drugs that are eligible for the flexible application of the ICER threshold and the measures for the expansion of the risk-sharing agreement (RSA) have been revealed. As always, the industry expressed a mix of expectations and concerns. The disclosed ‘Detailed evaluation criteria for new drugs etc. subject to negotiations’ goes as follows. Firstly, the requirements for ‘innovativeness’ to qualify for the flexible application of the ICER threshold were established. A new drug is regarded as ‘innovative’ when it satisfies all of the following criteria: ▲ there is no substitutable or therapeutically equivalent product or treatment ▲ a significant clinical improvement, such as prolonged survival, is recognized in the final outcome, ▲ the new drug is approved by the MFDS under Article 35(4)(2) of the Pharmaceutical Affairs Act through expedited review or is recognized as equivalent by the committee. The criteria are not very different from what was proposed in the ‘Preferential treatment measures for innovative new drugs’ that was first released last year. The difference is that in that draft, the U.S. FDA Breakthrough Therapy Designation (BTD) and the European EMA Priority Review (PRIME) were required, but this time, only the MFDS’s GIFT (Global Innovative products on Fast Track) designation is required. This is certainly encouraging. Of course, most truly innovative drugs receive BTD and PRIME designations. However, the fact that only MFDS’s GIFT is required as a criterion is, in many ways, removing the hurdle for innovative new drug designations. The most notable changes were made in the RSA eligibility criteria. The amendment expands the scope of the second condition in the RSA criteria, which was ‘drugs eligible for special calculation or equivalent,’ to further specify the ‘or equivalent’ diseases. The ‘or equivalent’ part was specified to severe diseases that do not qualify for the special calculation but are difficult to cure, irreversible disability or organ damage due to the progression of the disease, and hold a significant disease burden. In addition, if the expected additional claim amount for refund-type RSA-type drugs is less than KRW 1.5 billion within the scope of the reimbursement standard expansion, the drug may omit Drug Reimbursement Evaluation Committee evaluations and conduct NHIS negotiations. However, if the drug falls under the second RSA criteria, a new condition was added, stipulating that the expenditure cap must be applied even if it is not a pharmacologic evaluation exemption drug. This raises the potential for a number of issues. Firstly, the removal of the ambiguous phrase ‘special cases or equivalent’ is welcome. The clinical criteria of irreversible disability and organ damage also seem to be specific enough. However, it is unclear how many drugs will be eligible for less than KRW 1.5 billion additional claims criteria. Simplifying the process of expanding the RSA reimbursement criteria has been long desired by the industry, the problem is that while there weren't many drugs that exceeded that 'total amount' in the past, things have changed as we've entered the era of high-priced drugs. Furthermore, the criteria that set all drugs that fall under the second condition to be applied the expenditure cap RSA is a concern. In fact, recently listed drugs are being contracted with combined type RSA that includes the expenditure cap type. The problem is that while there weren't many drugs that exceeded that 'total expenditure cap’ in the past, things have changed as we've entered the era of high-priced drugs. In this situation, it remains to be seen whether the unilateral application of the ‘expenditure cap’ for drugs falling under the second criteria will lead to smooth reimbursement listing.