- LOGIN
- MemberShip
- 2026-04-25 23:56:21
- Policy
- First batch of drugs gain reimb through AEN pilot project
- by Jung, Heung-Jun Nov 17, 2025 06:11am
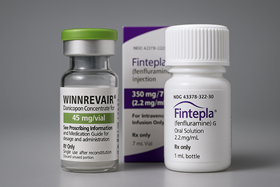
- With the first drugs under the Approval-Evaluation-Negotiation pilot program all securing reimbursement, the timing for the second batch of drugs currently under review to enter reimbursment listing is also approaching. Expectations are building for the possibility of their sequential insurance coverage as early as the first half of next year. With Bylvay Cap (odevixibat) gaining reimbursement coverage last month, all first-phase drugs under the Approval-Evaluation-Negotiation (AEN) project have now entered Korea’s reimbursement system, joining the previously covered Qarziba Inj (dinutuximab beta). The second batch of products under review are Winrevair (sotatercept) by MSD Korea for pulmonary arterial hypertension, Fintepla (fenfluramine) by UCB Korea for Dravet syndrome, and Limcato (anbal-cel) by the domestic company Curocell for large B-cell lymphoma. According to industry sources on the 16th, approximately 9 to 10 months have passed since the companies applied for reimbursement of these second-batch drugs. They are currently receiving cost-effectiveness reviews. Except for Winrevair, which received marketing authorization in July, the other two are also near approval. Among the three, Limcato was the first to apply for reimbursement in January, followed by Winrevair and Fintepla in February. Winrevair and Fintepla are being reviewed for cost-effectiveness, while Limcato is being reviewed for its clinical usefulness. Looking at the precedent set by the first batch of drugs that went through the reimbursement listing process, reimbursement for the second batch is expected early next year. As Qarziba was listed about 6 months after approval, if Winrevair, the only second batch drug approved as of now, follows a similar process, its reimbursement listing could occur as early as late this year or early next year. However, both Qarziba and Bylvay encountered hurdles at the Drug Reimbursement Evaluation Committee (DREC) stage, facing twists and turns before achieving reimbursement. Qarziba, which was approved by the Ministry of Food and Drug Safety (MFDS) in June last year, was initially deemed non-reimbursable at its first DREC review. It was subsequently granted for reimbursement in December last year after reapplying. It took approximately two years from Bylvay’s first reimbursement application in October 2023 to listing. Even at the point of its re-application last August, it had already been 14 months. This underscores the need to pass the DREC review without rejection and secure recognition of its reimbursement appropriateness, to achieve swift reimbursement listing. If a re-review process is required, reimbursement may be delayed beyond expectations. Meanwhile, the AEN linkage project is a pilot initiative aimed at enhancing access to high-priced treatments for severe diseases by shortening the period to listing of new drugs—which previously exceeded 300 days, including 120 days for MFDS (Ministry of Food and Drug Safety) approval, 150 days for HIRA (Health Insurance Review and Assessment Service) reimbursement evaluation, and 60 days for NHIS (National Health Insurance Service) drug price negotiations. During this year's National Assembly audit, the Ministry of Health and Welfare stated it would review plans to institutionalize the expedited listing system based on an analysis of the AEN pilot project's outcomes and feedback from the field.
- Company
- What's the outcome of the 2nd reimb attempt for 'Uplizna'?
- by Eo, Yun-Ho Nov 17, 2025 06:11am
- Attention has been drawn to whether 'Uplizna,' a new treatment for neuromyelitis optica spectrum disorders (NMOSD), will successfully gain insurance reimbursement listing at its second attempt. According to industry sources, Mitsubishi Tanabe Pharma Korea has accepted the conditional price that Health Insurance Review and Assessment Service (HIRA)'s Drug Reimbursement Evaluation Committee (DREC) suggested for Uplizna (inebilizumab), a treatment for adult patients with anti-aquaporin-4 (AQP-4) antibody-positive neuromyelitis optica spectrum disorder (NMOSD). The Ministry of Health and Welfare (MOHW) has ordered the National Health Insurance Service (NHIS) to initiate drug price negotiation. Therefore, the company is expected to begin discussions with the NHIS. The listing process for Uplizna was halted in October of last year during the drug price negotiation stage due to a supply issue. At that time, the company accepted a price below the amount evaluated by the HIRA's DREC and began drug price negotiations. However, a conclusion was not reached within the 60-day negotiation period. Although the National Health Insurance Service (NHIS) intended to enter into an extension negotiation, the renegotiation could not commence because the pharmaceutical company was unable to secure a domestic supply. NMOSD occurs when AQP4 autoantibodies, which are disease-specific markers produced by B cells, bind to the target antigen Aquaporin 4 (AQP4) on astrocytes in the central nervous system, thereby activating an immune response that leads to neurological damage. Uplizna is a CD-19-targeting humanized monoclonal antibody, developed using a novel mechanism, that prevents disease relapse by selectively binding to CD19, a B-cell-specific surface antigen, to deplete AQP4 antibody-producing B cells. Uplizna's safety and efficacy were demonstrated in the N-MOmentum clinical study, a monotherapy study conducted in 230 patients without concomitant immunosuppressants. The study results showed that 89% of patients who received Uplizna did not experience a relapse during the 197-day follow-up period, demonstrating a 77.3% reduction in relapse risk compared with the placebo group. The safety evaluation also showed a similar rate of adverse events to the placebo group.
- Product
- KMA protests in front of the NA opposing INN prescribing
- by Kang, Shin-Kook Nov 17, 2025 06:10am
- Representatives of doctors nationwide gathered in front of the National Assembly on the 16th to protest the government’s push to mandate international nonproprietary name (INN) prescriptions and allow Korean medicine practitioners’ use of X-ray devices. The Korean Medical Association (KMA, President Taek-woo Kim) held a ‘National Doctors’ Assembly to Protect Public Health and Oppose Anti-Medical Laws’ in front of the National Assembly in Yeouido, starting at 2 PM on the 16th. Following the rally, participants marched to the Democratic Party headquarters, voicing strong opposition to harmful medical legislation. Doctors KMA President Taek-woo Kim stated, “The issue of unstable drug supply is something the government must take responsibility for. Yet, instead of addressing that problem, the government is trying to use it as an excuse to enforce INN prescribing. The proposed bill even includes provisions for criminal punishment for physicians who do not comply — a completely unreasonable and excessive piece of legislation.” Kim added, “This is a clear anti-medical law that shatters trust between doctors and patients, threatens patient safety, and collapses the accountability structure. If drugs are substituted without a doctor's judgment simply because they contain the same ingredients, who will be responsible for the resulting erosion of doctors' prescribing authority and patient safety?” “Since the separation of medical and pharmaceutical services in 2000, we have steadfastly maintained the boundary between prescription and dispensing. However, the INN prescribing now being forced through by the National Assembly and the government is an act that clearly undermines the principle of separation of medical and pharmaceutical services upheld for over 20 years. Forcing ingredient-name prescriptions is tantamount to declaring the abolition of the separation of medical and pharmaceutical services.” He further pointed out, “The National Assembly is now pushing an absurd bill to include Korean medicine practitioners as responsible persons for the safety management of radiation-emitting devices. The idea of entrusting Korean medicine practitioners with the safety management responsibility for X-rays is a clear encroachment on the academic domain of medicine and erases the boundaries between professional licenses.” Kim declared, "The three major anti-medical initiatives — the revision of laboratory test regulations, the enforcement of generic prescriptions, and the allowance of X-ray use by Korean medicine practitioners — are not separate issues.. They represent the reckless policy rush of the National Assembly and government, which disregards public health and safety and brutally tramples on expert voices. We, as representatives of doctors nationwide, have gathered here to unite the anger and resolve of all 140,000 physician members and launch a comprehensive, powerful, all-out struggle to save healthcare in the Republic of Korea." Doctors Kyo-yoong Kim, Chair of the KMA Council of Delegates, also appealed, “The forced implementation of INN prescriptions is a harmful law that threatens patient safety and seriously infringes on doctors' prescribing rights. It must be immediately and permanently abolished.. Stop this irresponsible experiment right now, which could cause medication errors by using patient safety as collateral.” “If lawmakers insist on pushing through such an unjust bill, those who proposed and support it should be the first to apply it to themselves. If the true aim is public benefit and cost reduction, patients should be allowed to choose freely, and the healthcare system should transition to a one-stop in-hospital dispensing system rather than going through pharmacies.” Doctors The KMA adopted a resolution pledging to wage an all-out fight should the government and parliament proceed with enforcing INN prescribing or X-ray use by Korean medicine practitioners.
- Company
- Generic companies win 1st trial on unlisted Jardiance patent
- by Kim, Jin-Gu Nov 17, 2025 06:10am
- Generic drugmakers have scored the first victory in Korea’s ongoing disputes over unlisted patents related to the SGLT-2 inhibitor class diabetes treatment Jardiance (empagliflozin). Industry observers expect this decision to set the tone for similar disputes surrounding Jardiance's unregistered patents that are pending. According to industry sources on the 15th, the Intellectual Property Trial and Appeal Board (IPTAB) ruled in favor of four Korean generics companies, including Chong Kun Dang, Genuone Science, Korea Prime Pharm, and Huons, in an invalidation trial they filed against Boehringer Ingelheim’s unlisted use patent for Jardiance. This is effectively the first conclusion made regarding the disputes surrounding Jardiance's non-listed patents. Since January last year, Chong Kun Dang and the other companies have filed successive invalidation trials against the patent for ‘Therapeutic Uses of Empagliflozin (Patent No. 10-2318207)’. This patent pertains to the treatment and prevention of cardiovascular diseases in diabetic patients using empagliflozin. It expires in April 2034. A total of 7 unregistered patent disputes related to Jardiance are currently ongoing. Generic companies have filed successive trials against unlisted Jardiance-related patents since August 2023. All related patents pertain to the uses of empagliflozin or empagliflozin+linagliptin. Specifically: ▲1 patent expiring November 2027 (10-1463724) ▲1 patent expiring August 2028 (10-1491554) ▲1 patent expiring in February 2030 (10-1694136) ▲1 patent expiring in October 2030 (10-1737142) ▲Three patents expiring in April 2034 (10-1725469/10-2318207/10-2309654), etc. The patent that expires in November 2028 has drawn the most challenges. 13 companies, including Chong Kun Dang, Hanmi Pharm, PharmGen Science, DongKoo Bio&Pharma, Daehan Nupharm, Dongwha Pharm, Youngpoong Pharm, Withus Pharmaceutical, JW Pharmaceutical, Medica Korea, Boryung, Aprogen Biologics, and Korea Pharma filed invalidation trials, while Aju Pharm has filed a passive scope confirmation request. For the 2028 combination-use patent (empagliflozin + linagliptin), 9 companies, including Genuone Sciences, Boryung, Dongkook Pharmaceutical, Medica Korea, Aprogen Biologics, Korea Prime Pharm, Daewha Pharm, GC Biopharma, and Aju Holdings, have each filed for invalidation trials and passive scope confirmation trials. In addition, Hana Pharm and Aju Pharm filed avoidance trials for the 2030 empagliflozin + metformin related use and formulation patent, while Genuone Sciences filed an invalidation trial for the 2030 triple-combination patent (empagliflozin + linagliptin + metformin). However, Genuone Science voluntarily withdrew this trial. For the 3 patents expiring in April 2034, companies including Chong Kun Dang, Genuone Science, Korea Prime Pharm, Huons, and Cosmax Pharma are currently challenging them for invalidation or seeking to circumvent them. With this first trial decision regarding Jardiance's non-registered patents issued, the industry anticipates that similar pending cases will follow with first-instance conclusions. Indeed, in most cases, examiners have been assigned to panels for deliberation. If generic companies prevail in multiple disputes over non-listed patents, they could significantly reduce their patent risks. Non-listed patents are those registered with the Korean Intellectual Property Office but not included in the Ministry of Food and Drug Safety's Green list. Since they are not listed, they have no impact on generic product approvals. However, product launch is a different matter. The original drug’s company can claim infringement of unregistered patents. If the Patent Trial and Appeal Board or the Patent Court finds the generic company in infringement of the patent, the original drug’s company can then seek a preliminary injunction to halt product sales and claim damages for patent infringement. From the generic company's perspective, overcoming all unregistered patents is necessary to fully eliminate patent risk.
- Company
- "Expanded access to Pergoveris…for older infertile women"
- by Son, Hyung Min Nov 14, 2025 06:12am
- The paradigm of infertility treatment is shifting amid the increased age of mothers and the expansion of the patient population with poor ovarian response (POR). Notably, the utilization of FSH (Follicle-Stimulating Hormone) and LH (Luteinizing Hormone) combination therapy has surged since the reimbursement criteria for 'Pergoveris inj (follitropin alfa, lutropin alfa)' were eased last year. Consequently, a personalized ovulation induction strategy focused on older and low-responder patients is emerging as the core of domestic infertility treatment. In response to this change, Merck Korea held a 'Fertility Academy' on November 13 at the Parnas Hotel in Gangnam-gu, Seoul, to share the clinical value of Pergoveris and emphasize the importance of personalized treatment strategies. The event was held to commemorate Infertility Family Day (November 11), which was established to encourage infertile couples, raise social awareness, and share the clinical value and personalized treatment strategies associated with Pergoveris. The expanded insurance reimbursement for Merck's ovulation induction injection last April provided new treatment opportunities for many infertility patients. Professor Hee Jun Lee of CHA Gangnam Medical CenterPreviously, Pergoveris was only reimbursed (up to 2 vials/day) for assisted reproductive technology in patients with severe endogenous Luteinizing Hormone (LH) deficiency (concentration below 1.2 IU/L). However, the reimbursement criteria were expanded to cover 'cases where Pergoveris is administered (up to 2 vials/day) for ART in patients with LH deficiency.' Consequently, patients can now receive reimbursement benefits for Pergoveris without restriction based on LH levels. Pergoveris is the only approved combination product in Korea that mixes recombinant rFSH and r-hLH. It has shown significantly improved results across all indices, including pregnancy rate, cumulative pregnancy rate, and pregnancy rate per embryo transfer, compared to FSH monotherapy. These data were consistent in the real world. A large-scale analysis involving 10,000 poor ovarian response patients showed that Pergoveris resulted in a higher cumulative live birth rate compared to FSH monotherapy. The effect of Pergoveris was particularly pronounced in cases of severe poor ovarian response. Professor Hee Jun Lee of CHA Gangnam Medical Center's Department of Obstetrics and Gynecology assessed, "In older mothers and those with poor ovarian response, a treatment strategy that considers the balance of FSH and LH is paramount." He added, "It has been confirmed across multiple clinical trials and international expert consensus that co-administering recombinant LH from the beginning of ovulation induction improves both implantation and pregnancy success rates." Professor Lee added, "Given the high proportion of older mothers in Korea, the preference for combination therapy is increasing in actual clinical practice. The FSH + LH combination, primarily with Pergoveris, is becoming a key treatment strategy. The expanded reimbursement for Pergoveris has been a great help to patients. However, support for oocyte cryopreservation is still lacking, and there is room for improvement." Professor Lee emphasized, "The importance of early treatment for infertility is continuously stressed. As the efficacy and safety of these injectable agents have been confirmed, it is necessary to start treatment early." Merck's efforts to improve access for Korean infertility patients The infertility sector is particularly important due to the unique situation of low birth rates and aging demographics. Korea's fertility rate was 0.75 last year, the lowest in the world, and the population is expected to halve by the end of this century. The country remains at the lowest level among OECD nations. Demand for infertility treatment is also rapidly increasing due to the rise in older mothers. As of 2022, women aged 35 and over accounted for 70% of those receiving infertility procedures. Female fertility rapidly declines after age 35, often leading to Low Ovarian Reserve (LOR). When ovarian function is reduced, ovulation is less regular, and oocyte quality decreases, lowering fertilization and implantation rates. Merck is involved in social activities to address the low birth rate, going beyond simply supplying treatments. Kim Wook, Healthcare Fertility Unit Head at Merck KoreaMerck currently supplies the ovulation inducers 'Gonal-F' and 'Luveris,' in addition to Pergoveris, and has developed treatments for preventing premature ovulation. Merck launched the 'Fertility Counts' initiative in 2023 to discuss and respond to the socioeconomic challenges arising from declining birth rates in the Asia-Pacific (APAC) region. Fertility Counts was established to support effective policy intervention by authorities in building a family-friendly society through relevant research and resource provision, emphasizing the need for improved fertility-related policies to help those who wish to have children. In particular, Merck plans to continue its environmental, social, and governance (ESG) activities in Korea through its Infertility Awareness Campaign. Last year, Merck and the Korean Infertility Family Association signed an MOU to address the growing social problem of infertility rates. Kim Wook, Healthcare Fertility Unit Head at Merck Korea, stated, "We are conducting various activities in collaboration with multiple stakeholders, including the Fertility Counts initiative, the Family-Friendly Future Forum, a network of companies, government, and academia, as well as collaboration with infertility patient groups," and added, "We will continue our efforts, focusing on Pergoveris, to contribute to healthy births for Korean mothers."
- Policy
- Pricing nego for Erleada underway…last hurdle to reimb
- by Jung, Heung-Jun Nov 14, 2025 06:12am
- Janssen Korea's prostate cancer treatment Erleada (apalutamide) has entered price negotiations, the final hurdle for its reimbursement expansion in Korea. Erleada passed the Drug Reimbursement Evaluation Committee review last October and was deemed appropriate to expand reimbursement coverage to ‘high-risk non-metastatic castration-resistant prostate cancer’. According to industry sources on the 13th, the National Health Insurance Service (NHIS) is in pricing negotiations for Erleada’s reimbursement expansion. It being a drug subject to reimbursement under the risk-sharing agreement, the contract will be finalized based on the refund rate determined during negotiations. Once the price negotiations are complete, Erleada will be covered for ‘high-risk non-metastatic castration-resistant prostate cancer (nmCRPC)’ in addition to its existing coverage for ‘metastatic hormone-sensitive prostate cancer (mHSPC)’. The hormone-sensitive metastatic prostate cancer (mHSPC) indication was granted reimbursement in April 2023 as a refund-type risk-sharing agreement drug at KRW 20,045. At that time, Janssen focused its efforts on securing reimbursement for Erleada, even voluntarily lowering the price of its own prostate cancer treatment, Zytiga. It also became the first androgen receptor targeted agent (ARTA) to receive essential reimbursement at a 5% coinsurance rate. About six months later, competition intensified when Astellas Pharma Korea's Xtandi Soft Cap (enzalutamide) was added to the list as an essential reimbursement drug. Another competitor is Bayer Korea's Newbeqa, which also indicates treating ‘high-risk non-metastatic castration-resistant prostate cancer’. The company submitted a reimbursement application for Newbeqa in June this year, but the drug’s clinical benefit is still under review. With Erleada poised for reimbursement expansion, its prescription scope is also expected to broaden. Consequently, attempts to expand reimbursement for competing prostate cancer treatments are likely to follow. According to the pharmaceutical market research institution UBIST, Erleada's sales last year reached KRW 31.2 billion, a 368% increase from the previous year's KRW 6.6 billion.
- Company
- Generic company challenges flu drug Xofluza’s patent
- by Kim, Jin-Gu Nov 14, 2025 06:12am
- Roche's influenza treatment Xofluza (baloxavir marboxil) has become a generic company’s target for patent challenge. This marks the first patent challenge against Xofluza. Industry observers predict related patent challenges will continue in the near future. According to industry sources on the 13th, Kwangdong Pharmaceutical recently filed a passive scope of rights confirmation trial against Shionogi & Co. regarding Xofluza’s formulation patent. Shionogi is the original developer of Xofluza, and Roche Korea is currently in charge of its domestic sales. A total of 4 patents related to Xofluza are currently registered. These include two substance patents expiring in 2031 and 2036, respectively, and two formulation patents expiring in 2038 and 2039, respectively. Among these, Kwangdong Pharmaceutical has filed a circumvention trial against the formulation patent (for a solid formulation with excellent stability) that expires in April 2039. The industry anticipates that patent challenges against Xofluza will likely expand in the future. Kwangdong Pharmaceutical is expected to file an additional challenge against one of the remaining formulation patents. If Kwangdong Pharmaceutical successfully avoids both formulation patents, it could bring forward the launch date for Xofluza generics to September 2036. There is also speculation that other generic companies may follow suit with patent trial applications. Under current regulations, to satisfy the requirement of being the “first to file a trial request” and obtain exclusive marketing rights (first generic exclusivity rights), companies only need to file the same trial request within 14 days. As a result, it is projected that patent challenges from other generic companies besides Kwangdong Pharmaceutical will continue until the end of this month. Previously, a total of 10 companies participated in the patent challenge against another influenza treatment, Peramiflu inj (Peramivir). Xofluza is an influenza treatment approved in Korea in 2019. Roche introduced Xofluza as a successor to its existing influenza treatment Tamiflu (oseltamivir). Unlike Tamiflu, which requires oral administration over 5 days, Xofluza offers improved convenience in administration by enabling treatment with a single injection. However, Xofluza is currently a non-reimbursed drug, resulting in limited domestic sales performance. If it succeeds in gaining reimbursement status, projections indicate it could absorb Tamiflu's sales. According to the Ministry of Food and Drug Safety, Xofluza’s domestic import sales were around USD 1.6 million in 2022-2023 but surged significantly to USD 7.38 million (approximately KRW 10.8 billion) last year.
- Company
- AZ runs Lung Health Check Bus…screens Changwon residents
- by Son, Hyung Min Nov 14, 2025 06:11am
- The ‘Lung Health Check Bus’ was operated in the ‘2025 Anti-Aging BIOHealth Expo’ that was held at the Changwon Exhibition and Convention Center (CECO) from the 6th to 8thAstraZeneca Korea announced on the 12th that it operated a ‘Lung Health Check Bus’ equipped with AI-based chest X-ray technology at the ‘2025 Anti-Aging BIOHealth Expo (ABEXPO 2025)’, which was held at the Changwon Exhibition and Convention Center (CECO) in Gyeongsangnam-do from the 6th to the 8th, in collaboration with the Korea Tuberculosis Association and Maihub. ABEXPO 2025 is Korea’s premier healthcare exhibition dedicated to advancing healthy longevity and innovation in the anti-aging industry in response to an aging society. The event showcased next-generation anti-aging solutions that integrate cutting-edge technologies such as AI, biotechnology, and digital healthcare. The Lung Health Check Bus was showcased as a representative example of digital healthcare applying AI to enable early screening of lung diseases, embodying the starting point of anti-aging medicine, “disease prevention through early detection.” Notably, Changwon, the host city of the exhibition, officially entered a super-aged society in June, with more than 20% of its population aged 65 or older, a demographic particularly vulnerable to cardiovascular and pneumonia-related risks. AstraZeneca Korea participated in this expo with the aim of expanding healthcare access beyond the capital region to rural areas. Moving forward, it plans to continue touring the nation with the Lung Health Check Bus, supporting diverse citizens to easily check their lung health. During the expo, approximately 570 visitors came to the booth to experience AI-based chest X-ray screening. Visitors were able to directly check their lung health status through the interpretation reports provided on-site. Fifty-seven percent of the examinees were aged 50 or older. The AI reports detected abnormal findings, such as lung nodules (165 individuals), in 238 people (42%). Among those with abnormal findings, 11% (26 individuals) had a family history of lung cancer. The results of the specialist's detailed interpretation were sent to each individual within a week. Abnormal findings detected on chest X-rays can be associated with various lung and cardiovascular diseases, including lung cancer, making early detection and prompt management crucial. Confirming uncertain lung nodules is also essential for the early diagnosis of lung cancer. Lung cancer was the leading cause of cancer-related mortalities in Korea in 2023. Beyond smoking, it can arise from various factors, including family history, secondhand smoke, and environmental exposures like air pollution. Lung cancer often presents with minimal early symptoms or, even when the disease has progressed, may show no significant symptoms beyond coughing and sputum production. At the time of diagnosis, 41.4% of patients already have distant metastases. According to 5-year relative survival rate statistics from 2018-2022, the survival rate is only 12.9% when lung cancer metastasizes to other sites. However, if detected at a localized stage, the survival rate significantly increases to 79.8%. The current national lung cancer screening program targets high-risk individuals aged 54 to 74 with a smoking history of 30 pack-years or more, utilizing low-dose chest CT scans. Additionally, AI-based chest X-rays are being regarded as an effective tool for lung cancer diagnosis due to their high detection rate of pulmonary nodules. At the Changwon ABEXPO, a Lung Health Check Bus was operated as a way for non-smokers and low-risk citizens to check their lung health through AI chest X-rays. Jiyoung Ahn, Executive Director of Strategy and External Cooperation at AstraZeneca Korea, stated, "AstraZeneca Korea is creating practical solutions to enhance access to early diagnosis and treatment through collaboration with domestic AI healthcare companies. The Lung Health Check Bus is a prime example demonstrating how cutting-edge AI technology can directly contribute to public health.“ She added, ”We hope the AI-integrated Lung Health Check Bus will address the screening gap for lung cancer in non-smokers and serve as an opportunity to spread awareness about the importance of early screening. AstraZeneca Korea will continue to contribute to raising awareness for early lung cancer diagnosis and improving the quality of public health through science-driven collaboration and practice." Min-seok Shin, President of the Korea Tuberculosis Association, stated, “We hope this campaign enables more citizens to easily check their lung health and detect diseases early. The Korea Tuberculosis Association will continue its efforts to promote nationwide lung health and expand screening accessibility.” Hyeok Yang, CEO of Maihub, said, “Digital healthcare technology is a crucial means of innovating personal health management and increasing access to medical services. The Lung Health Check Bus, which utilizes AI chest X-ray technology, holds significance as it demonstrated how advanced technology can contribute to actual public health improvement. We will continue to strive to build a healthy society based on public benefit and innovation.” Visitors to the ‘2025 Anti-Aging BIOHealth Expo’ held at the Changwon xhibition and Convention Center from the 6th to the 8th are receiving on-site lung health interpretation reports after having AI-equipped chest X-rays taken at the Lung Health Check Bus. As a member of the global non-profit collaboration ‘Lung Ambition Alliance (LAA)’, AstraZeneca Korea has been continuing various initiatives to create ‘a world where lung cancer is no longer a cause of death’. Last year, AstraZeneca Korea and the LAA launched the ‘Lung Cancer Zero’ campaign. In collaboration with the British Chamber of Commerce in Korea and the British Embassy Seoul, they expanded the reach of lung cancer awareness activities across various sectors of society. Last June, AstraZeneca Korea signed a tripartite Memorandum of Understanding (MOU) with the Korea Tuberculosis Association and Maihub to successfully operate the ‘Lung Health Check Bus’ campaign. It is leading the way in early detection of lung diseases by operating mobile screening buses nationwide, equipped with AI chest X-ray imaging capabilities. Participating in this Changwon Expo is part of the effort to create an early screening environment accessible to everyone, extending beyond the capital region to local communities. AstraZeneca Korea has been expanding its collaboration with Korea’s AI and digital healthcare ecosystem. Specifically, it is conducting joint research and pilot projects with various domestic startups across diverse fields, including: ▲AI image analysis (OncoSoft, AVIS), ▲Precision medicine and genomic analysis (3Billion), and ▲digital patient community and health data-based management solutions (Humanscape, Maihub), to build innovative healthcare models applicable in real clinical settings.
- Policy
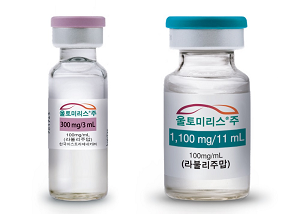
- Ultomiris gains reimbursement approval for 'NMOSD·gMG'
- by Jung, Heung-Jun Nov 14, 2025 06:11am
- Product photo of Ultomiris AstraZeneca's Ultomiris (ravulizumab) will receive reimbursement coverage for Neuromyelitis Optica Spectrum Disorder (NMOSD) this month, and its reimbursement scope will expand to generalized myasthenia gravis. Notably, the Ministry of Health and Welfare (MOHW) has requested reimbursement coverage for myasthenia gravis. Therefore, it was considered a separate agenda item for the Drug Reimbursement Evaluation Committee (DREC) held on November 6. According to industry sources, Ultomiris has passed the DREC, and it will be covered for reimbursement of 'anti-acetylcholine receptor (AchR) positive generalized myasthenia gravis (gMG)' starting December 1. Although it was not included in the meeting report disclosed by the DREC; however, it was discussed as a separate supplementary agenda. It appears that the MOHW's request for reimbursement for gMG treatments has played a role. During this year's MOHW Parliamentary Inspection, access to treatment for rare, severe diseases, including gMG, was highlighted. According to the report that Democratic Party Rep. Young-seok Seo received from the MOHW, there are no gMG medicines eligible for reimbursement. In response, the MOHW stated, "The MOHW continues to identify rare disease treatments that require urgent reimbursement coverage through seeking clinical field." It appears that the inclusion of the agenda and approval in the DREC was influenced by the critiques from the Parliamentary Inspection and the government's policy to enhance patient access. As of November 1, Ultomoris is covered by reimbursement for its indication to 'treat anti-aquaporin-4 (AQP-4) antibody-positive neuromyelitis optica spectrum disorder (NMOSD).' Eligible patients include those aged 18 or older who test positive for anti-aquaporin-4 (AQP-4) antibodies. There have been continued requests for reimbursement coverage for Ultomiris by patient organizations and petitions to the National Assembly. With the reimbursement scope expanding between November and December, more patients are likely to be prescribed in the future.
- Policy
- Pharmaceutical post-market management system to be enhanced
- by Jung, Heung-Jun Nov 14, 2025 06:11am
- The National Health Insurance Service (NHIS) plans to revise its drug price negotiation and post-market management systems next year, with a focus on stable pharmaceutical supply. The NHIS will actively enforce penalties stipulated in its regulations if pharmaceutical companies discontinue drug supply without notification in violation of agreements reached during price negotiations. This is a response to criticism raised during this year's parliamentary inspection that the agency was not adequately managing the supply obligation despite having the 'Drug Price-Reimbursement Agreement.' Se-rim Oh, Head of the Negotiation and Post-Management Unit at the Department of Drug Management.On November 11, the NHIS held a 'Drug Price Negotiation and Post-Market Management System Briefing' at its headquarters in Wonju, sharing the direction for management reinforcement with industry stakeholders for next year. The NHIS emphasized that if a company must inevitably suspend supply in violation of the agreement, it must formulate patient protection measures and consult with the NHIS beforehand. Se-rim Oh, Head of the Negotiation and Post-Management Unit at the Department of Drug Management, said, "The NHIS plans to strengthen management focusing on supply issues. If a company withdraws approval and suspends supply, it must ask pharmaceutical associations and formulate patient protection measures." Starting next year, the NHIS will actively consider imposing penalties on companies that violate their supply obligations, based on a fixed formula. The current formula sets the penalty amount per day of violation as: "(Previous Year's Annual Claim Amount of the Violating Drug) X 1/n X1/365X50%." Furthermore, companies will be required to submit data to the NHIS within 40 days of the end of each quarter to verify the fulfillment of their supply obligation (including monthly production volume, import volume, requested supply volume, and actual supply volume). The existing clause requires a company to pay a KRW 1 million fine for failure to mee these requirements. This is in response to criticism during the parliamentary inspection that the clause was not being enforced. The NHIS plans to actively enforce this rule. Oh explained, "If fulfilling the agreement is difficult, you must communicate and consult with the NHIS beforehand. Some companies have already agreed to the penalty clause after prior consultation due to the circumstances of their contract manufacturers." Oh added, "Companies failing to submit (data) are supposed to pay KRW 1 million, but this hasn't been enforced. The NHIS is aware of this. Please comply with it going forward." Eliminating reporting requirements for new efficacy and effectiveness...will review streamlining the requirement to report dosage The current obligation for companies to report any additional efficacy and effectiveness secured overseas after signing a contract with the NHIS will be exempted starting in the first half of next year. Reporting obligations when adding a new dosage will also be streamlined. Instead, if the domestic introduction of the new strength is necessary, the details will be stated in the agreement. Oh said, "The agreement includes the obligation to report when an indication is added. We are reviewing the exemption of this reporting obligation starting in the first half of next year," and added, "We are also considering simplifying the reporting obligation for adding new dosage strengths. However, if domestic introduction of a strength already listed for overseas reimbursement is necessary, we plan to address this by explicitly stating the details in the agreement. We will be gathering opinions on this matter." The NHIS is also considering the partial disclosure of information regarding Risk-Sharing Agreement (RSA) drugs. They are currently conducting consultations on disclosing a list of drugs subject to the refund-type RSA. Oh said, "If a reimbursement decision application is made to HIRA, we will ensure that information about the refund rate can be provided. However, a non-disclosure agreement must be signed," and added, "We are also conducting consultations on publicly disclosing the list of drugs subject to the refund-type RSA." PVA 'one-time refunds' will be allowed temporarily next year...new guidelines for negotiaing scope expansion will be established Hae-hee Moon, Head of the Volume Management Unit at the Department of Drug Management.The NHIS will also improve its drug utilization management plan next year. The agency plans to restrict the operation of the "one-time refund" mechanism, which was implemented during the COVID-19 pandemic, and establish new guidelines for negotiating the expansion of a drug's usage scope. Hae-hee Moon, Head of the Volume Management Unit at the Department of Drug Management, said, "We plan to limit the products eligible for the one-time refund contract starting next year. We are currently discussing this with the Ministry of Health and Welfare (MOHW). We plan to gather opinions from the pharmaceutical association afterward." Moon explained, "We are also preparing guidelines due to the need for written instructions in the process of negotiating the expansion of usage scope. We plan to conduct consultations this week and will refine these guidelines next year." The NHIS is also reviewing plans for introducing a dual pricing system with the MOHW. Moon said, "There is a request from pharmaceutical companies to introduce a dual pricing system due to the U.S. MFN policy. We are reviewing system improvements and implementation plans with the MOHW." Revision of price cap adjustment guidelines...separate adjustments for emergency imported drugs Hyung-min Kim, Head of the New Drug Management Department.The guidelines for adjusting the ceiling price during drug price negotiations will also be partially revised. The consideration for the estimated claim amount, which was previously based on the 'claim volume and growth rate of the negotiated drug over the past 3 to 5 years,' will be changed to the 'claim amount generated by the negotiated drug over the past 3 to 5 years.' Furthermore, a clause will be added to ensure that drugs urgently imported through the Korea Orphan and Essential Drug Center (KODC) will have a separate price adjustment procedure. In addition, the clause specifying 'when there is a need for public healthcare' as an exception to the three-year limit on price adjustment applications after the initial adjustment will be further specified. Hyung-min Kim, Head of the New Drug Management Department, explained, "We will define the need for public healthcare as a request for cooperation from central administrative agencies, and revise the regulation to require the Pharmaceutical Benefit Evaluation Committee to notify the NHIS of a price readjustment application," and added, "A clause will be inserted requiring measures such as refunds if the contract falls short of the mandatory production volume agreement," emphasizing the ministry's commitment to stable supply.