- LOGIN
- MemberShip
- 2026-05-02 12:39:16
- Policy
- Reimb standards that prevent abuse of HA Eye Drops imminent
- by Lee, Tak-Sun Nov 05, 2024 05:45am
- Health authorities are expected to establish reimbursement standards to prevent the misuse of hyaluronic acid eye drops after failing to reach a conclusion during last year's reimbursement adequacy evaluations. It is reported that the Ministry of Health and Welfare is preparing an administrative notice to announce the reimbursement standards set by HIRA. HIRA’s reimbursement standard plan is expected to focus on the issue of abuse and misuse of HA eye drops and includes some restrictions on the number of uses and combinations. According to industry sources on the 3rd, HIRA’s proposed salary standards for HA eye drops are expected to be administratively notified soon. HA eye drops received reimbursement reevaluations in 2023, but no conclusion was made at the time. In September last year, during the first deliberation by the Drug Reimbursement Evaluation Committee, the committee determined that its use for “exogenous diseases such as postoperative, pharmaceutical, trauma, and contact lens wear” is not adequate for reimbursement. On the other hand, endogenous diseases such as Sjögren's syndrome, skin mucosal eye syndrome, and dry eye syndrome were deemed reimbursable, and it was concluded that it was necessary to set reimbursement standards such as single prescription per patient visit and total number of prescriptions per patient per year to encourage its appropriate use. However, opposition arose due to reasons such as weakened access and increased costs for elderly patients who use HA eye drops, and the issue was raised during the NA audit. As a result, DREC was unable to make a conclusion during its second deliberation and decided to review it further. At the MOHW’s Health Insurance Policy Review Committee meeting, which was held in December last year, the committee also said, “In the case of HA eye drops, we will make a final decision at a later date based on the evaluation results as it is necessary to consider setting the reimbursement standard for disposable eye drops as a whole in consideration of the users’ switch to other disposable eye drops.” So in the light of the new year, the health authorities reportedly came up with a reimbursement plan focused on preventing misuse. “It is expected that there will be some restrictions on the number of doses, combined use, etc.,” said a pharmaceutical industry insider, adding, ”The new reimbursement standards will have more restrictions on its use than now, but I understand that they are more aimed at preventing abuse.” After the first round of deliberations last year, there was a proposal to limit the number of boxes containing 60 individual vials to 4 per year. The pharmaceutical industry opposed the proposal, saying it was too restrictive. The current proposal, while still limiting the number, is said to be more eased than the 4 boxes per year. HIRA reportedly prepared a reimbursement standard and reported it to MOHW. The industry believes that MOHW will issue an administrative notice soon after a long discussion. HA eye drop prescriptions amount to around KRW 230 billion a year. They are widely used for eye diseases. Therefore, it will be interesting to see how the proposed reimbursement standard will affect the drug market.
- Company
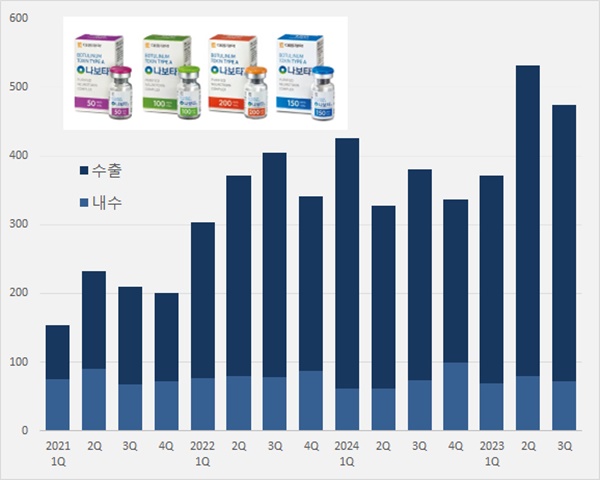
- 'Nabota' generated KRW 115.8B over 9 mths in foreign mkt
- by Chon, Seung-Hyun Nov 05, 2024 05:45am
- Daewoong's Nabota, which contains botulinum toxin, is expanding its presence in the foreign market. Its export amount surpassed KRW 100 billion up to Q3 2024. Over 80% of the overall sales were accounted for by sales generated in the foreign market. It is a cash cow export product. According to Daewoong on November 2, Nabota generated sales of KRW 47.5 billion in Q3, up 25.0% from KRW 38 billion Year-over-Year (YoY). Nabota's domestic sales amounted to KRW 7.2 billion in Q3, down 2.7% from the previous year. The export amount increased to KRW 40.3 billion, up 31.7%. Nabota's cumulative sales were KRW 137.8 billion in Q3, an increase of 21.6% from the previous year. Quarterly Nabota sales trend (unit: KRW 100 million, source: Daewoong). The export sales of Nabota have significantly increased due to its established credibility in the United States, following Daewoong's settlement with Medytox in a lawsuit for a stolen strain in 2019. In February 2021, Medytox settled a tri-party agreement with Daewoong's U.S. partnering companies, Evolus and AbbVie, for the sales of Nabota (marketed as Jeuveau in the United States) in the United States. Medytox and AbbVie will grant U.S. sales·marketing rights of Jeuveau to Evolus, and they will receive royalty payments. Nabota recorded export sales of KRW 6.4 billion in Q3 2020, an increase of over six-fold in four years. Nabota recorded cumulative export sales of KRW 115.8 billion until Q3 2024, up 23.7% Year-over-Year (YoY). In just 9 months, it nearly reached last year's export sales of KRW 117.4 billion. Export sales account for 84.0% of Nabota sales in Q3. The sales in the foreign market are substantial with continued exports between Q1 and Q3 this year at around 80%. Nabota ranks second in the cosmetic market for botulinum toxin in the United States, with a 13% market share. "In the first half of the year, Nabota's U.S. cosmetic sales surpassed Ipsen France's Dysport," the company said. "Nabota has recorded the highest growth rate among the cosmetic botulinum toxin brands. We continue establishing it as an outstanding brand with the highest quality overseas." In June, Evolus officially launched Nabota in Spain, expanding into the European market. Nabota is marketed as Nuceiva in Europe, and it has been launched in Spain, the UK, Germany, Austria, and Italy, continuing to expand to European countries in addition to the United States. In June, Nabota received marketing authorization from Argentina's National Administration of Drugs, Food and Medical Technology (ANMAT), and in September, it was launched in Malaysia. Nabota received marketing authorization from 68 countries worldwide, and the company has signed partnership agreements with 80 countries. Nabota is nearing its launch into the treatment market, which accounts for half of the global botulinum toxin market. Nabota's U.S. partnering company, iON Pharma, for the treatment indication is speeding up clinical trials for indications such as ▲chronic migraine ▲myotonia ▲gastroparalysis ▲PTSD. Daewoong has begun expanding its product plant ahead of increased demand for Nabota. The company invested KRW 101.4 billion in Hyangnam plant in Hwaseong, Gyeonggi Province to build the third plant with a yearly production size of 13 million vials. The third plant construction will commence in the first half of this year. When the third plant is completed, Daewoong will secure up to 18 million vials production capacity, including a yearly 5 million vials production from plants 1 and 2. "Nabota is a high-purity toxin manufactured using a patented and proprietary 'HI-PURETM Technology.' It provides an advantage of fast and accurate effects and safety towards drug tolerance," said Daewoong representative.
- Company
- 'SGLT2·DPP4' comb market records robust growth
- by Kim, Jin-Gu Nov 04, 2024 05:48am
- Product photos of Qtern, Esgliteo, Esgliteo, and Sugadapa (clockwise from upper left). The market for combination drugs containing SGLT-2 inhibitor and DPP-4 inhibitor for treating type 2 diabetes is showing rapid growth. The industry expects the market size to expand by over KRW 30 billion this year. Products containing one or more original active ingredients are leading the market growth. Boehringer Ingelheim's 'Esgliteo' ranked top in prescription sales with cumulative sales of KRW 8.6 billion this year. It was followed by LG Chem's 'Zemidapa,' with KRW 6.1 billion, and AstraZeneca Korea's Qtern, with KRW 3.5 billion. 'SGLT2i+DPP4i combination drugs' generated KRW 9.5B in Q3…Will it surpass yearly KRW 30B? According to a pharmaceutical market research firm UBIST on October 2, two-drug combination drugs with SGLT-2 inhibitor and DPP-4 inhibitor generated KRW 9.5 billion in net sales in Q3 2024. The sales rose significantly over three-fold compared to KRW 2.8 billion of Q3 2023. This market was established with expanded reimbursement for diabetes combination therapy since April 2023. The government approved reimbursement of SGLT-2 inhibitor and DPP-4 inhibitor combination. After that, combination drugs containing different combinations were launched. In the same month, the patent of SLGT-2 inhibitor, 'Forxiga (dapagliflozin),' expired. Companies with original products with DPP-4 inhibitor have started to launch combination drugs with dapagliflozin class. In September, the DPP-4 inhibitor 'Januvia (sitagliptin)' patent expired. After that, generic companies without original active ingredients jumped into the competition. Quarterly prescription market size of SGLT2i+DPP4i combination drugs (unit: KRW 100 million, source: UBIST). Earlier in the launch, two-drug combination drugs generated below-expected sales. In 2023, the sales amounted to KRW 700 million in Q2 and KRW 2.8 billion in Q3. According to an analysis, the sales rose from Q4. It recorded KRW 5.3 billion in Q4 2023. This year, the sales significantly rose to KRW 7 billion in Q1, 8.2 billion in Q2, and 9.5 billion in Q3. The pharmaceutical industry expects sales to surpass KRW 10 billion for Q4 2024. The cumulative prescription sales in Q3 amounted to KRW 24.7 billion. This year's market size is expected to expand to over KRW 30 billion. Combination drugs with original products such as Esgliteo·Zemidapa show strong growth By products, products with original active ingredient are showing strong growth regardless of Korean pharmaceutical companies or global pharmaceutical companies. Boehringer Ingelheim's 'Esgliteo' generated the highest prescription sales in Q3 2024 with cumulative sales of KRW 8.6 billion. Last year, Boehringer Ingelheim launched two-drug combination drugs with its in-house originals, Jardiance (empagliflozin) and Tradjenta (linagliptin). It was followed by LG Chem's 'Zemidapa' with cumulative prescription sales of KRW 6.1 billion. Zemidapa contains LG-Chem's proprietary Zemiglo (gemigliptin) and dapagliflozin. The company launched this product after Forxiga's patent expired in April 2023. The next in sales is Astra Zeneca's 'Qtern' with KRW 3.5 billion. AstraZeneca combined its proprietary original Forxiga and Onglyza (saxagliptin). In South Korea, Ildong Pharmaceutical is responsible for the distribution of Qtern. AstraZeneca also has 'Sidapvia' with a different combination. Sidapvia's cumulative prescription sales are KRW 1.2 billion. It is a combination drug with dapagliflozin·sitagliptin. AstraZeneca collaborated with SK Chemicals to develop this drug and launched it in the market around the time of Januvia's patent expiration in September 2023. In South Korea, SK Chemicals is responsible for domestic production, and HK inno.N distributes the drug. The next drugs in rank are Chong Kun Dang's 'Exiglu-S' with KRW 1.1 billion, and Dong-A ST's 'Sugadapa' with KRW 1 billion. Chong Kun Dang launched Exiglu-S with a combination of Januvia, acquired from MDS, and dapagliflozin. Dong-A ST launched Sugadapa with a combination of its proprietary DPP-4 class Suganon (evogliptin) and dapagliflozin. The remaining 50 companies have generated cumulative prescription sales below KRW 500 billion up to Q3 2024. Most of these companies compete with products with dapagliflozin+sitagliptin. However, analysis suggests that their sales are below expected because they have entered the market later than other combination drugs with originals over five months, and many products were launched around the same time.
- Company
- Will the polycythemia vera drug Besremi be reimb in KOR?
- by Eo, Yun-Ho Nov 04, 2024 05:48am
- Whether PharmaEssentia Korea’s new drug for polycythemia vera, ‘BESREMi,’ will be listed with reimbursement in Korea is gaining attention. The drug was approved for hydroxyurea-refractory or intolerant polycythemia vera in March last year but failed to overcome the CDDC barrier in July of the same year. At that time, the CDDC determined that there was insufficient evidence to determine the clinical utility of BESREMi as a second-line treatment. In response, PharmaEssentia resubmitted its application for reimbursement in March after adding domestic clinical data on BESREMi and supplementing the evidence on the drug’s efficacy in second-line therapy. As 50,000 people signed a petition for improved access to BESREMi to the National Assembly in February, whether the company will succeed in receiving reimbursement this time is gaining close attention. BESREMi is a next-generation interferon treatment that selectively removes JAK2 mutations that cause polycythemia vera. I It was developed to improve the purity and tolerability of existing interferons so that it can be administered every two weeks for the first 1.5 years and every four weeks thereafter. It is currently recommended for the treatment of PV in the National Comprehensive Cancer Network (NCCN) and European Leukemia Network (ELN) guidelines, regardless of prior treatment history. Polycythemia vera is a rare blood disorder where a somatic cell mutation in the bone marrow abnormally activates bone marrow function and produces excessive red blood cells. According to HIRA data, about 5,000 patients are affected with PV in Korea, and hydroxyurea is mainly used for the majority of patients. However, as the current reimbursed drugs are not curative and there are no new alternatives for patients who fail hydroxyurea treatment, there remains a high unmet need for the disease.
- Company
- K-Bios globally present immuno-oncology drugs
- by Son, Hyung Min Nov 04, 2024 05:48am
- The development achievements of the domestic pharmaceutical bio industry's immuno-oncology drugs will be presented at an overseas conference. Hanmi Pharmaceutical, GC Cell, Abion Bio, ST Cube, and Y-Biologics, among others, have completed preparations to emerge into the international stage by disclosing positive clinical trial results. In particular, some companies are conducting clinical trials for combination therapies that utilize their approved immuno-oncology and targeted therapy drugs. They are aiming to increase the chances of commercialization by combining the drugs with verified therapies. Bispecific antibodies emerge as a global R&D trend, Korean companies also make a bid into the industry According to industry sources on the 4th, The Society for Immunotherapy of Cancer’s annual meeting (SITC 2024) will be held in Houston, U.S., from April 6 to 10. STIC is the world's largest immuno-oncology society with more than 5,000 industry professionals from more than 70 countries around the world. Hanmi Pharmaceutical will present the clinical trial results for its bispecific antibody, which has emerged as a global R&D trend. The company is conducting a Phase I clinical trial on BH3120, a bispecific immuno-oncology drug candidate. BH3120 simultaneously targets PD-L1 and 4-1BB. Bispecific antibodies are drugs that can bind to two different antigens simultaneously, or to two different epitopes on the same antigen. Such antibodies that simultaneously target various biomarkers have the advantage of allowing BBB penetration through targeted binding to receptors on the surface of the blood-brain barrier. In particular, anticancer drugs need to penetrate the BBB to increase drug permeability. Recently, a growing number of companies have developed multispecific antibodies by combining antibodies that bind to antigens that regulate the activity of immune cells and antibodies that bind to specific antigens on tumor cells. In clinical trials, BH3120 showed a decoupling of immune activity between the tumor microenvironment and normal tissue, confirming its safety. In addition to immuno-oncology drugs, Hanmi Pharmaceutical and Beijing Hanmi Pharm are also exploring the possibility of combining the drug with other anticancer drugs. BH3120 incorporates Hanmi’s platform technology, “Pentambody.” This is a next-generation bispecific antibody platform technology that activates immune cells while attacking only target cancer cells. Since 4-1BB is activated only in immune cells surrounding cancer cells expressing PD-L1, BH3120 minimizes the toxic side effects of 4-1BB and has long-term anti-cancer effects that prevent recurrence. Y-Biologics will present clinical results from AR092, one of its T-cell bispecific antibody pipeline. AR092 is a drug candidate generated from Y-Biologics’s proprietary next-generation T-cell bispecific antibody platform, “ALiCE.” Y-Biologics confirmed the potent anti-cancer effect and low toxicity of AR092 against solid tumors through a preclinical trial. SITC 2023(Source= SITC). Developing immuno-oncology drugs targeting novel mechanisms of actions Cell therapies and immuno-oncology drugs targeting new mechanisms of action will also be introduced at SITC 2024. G-Cell will present preclinical results of its NK cell therapy candidate “GCC4001” in combination with Merck's EGFR antibody therapy Erbitux. In preclinical studies, GCC4001+Erbitux demonstrated approximately 2 times the anti-tumor activity of Erbitux monotherapy. GC Cell believes that the combination may represent a novel treatment alternative for recurrent or metastatic head and neck cancer. GC Cell will also present unique findings on its proprietary NK cell culture technique based on eHuT-78 CDV feeder cells. ST Cube will present clinical results on its “nelmastobart,” which targets a novel biomarker, BTN1A1. BTN1A1 is a protein that regulates the immune response to cancer cells by inhibiting the activity of T-cells. This biomarker is not expressed in normal cells, but is strongly expressed in cancer cells and is mutually exclusive with PD-L1. By targeting BTN1A1, ST Cube is developing an immuno-oncology drug that may represent a new treatment option in refractory cancers. Currently, ST Cubde is conducting Phase Ib/II clinical trials in the U.S. and South Korea for nelmastobart in combination with paclitaxel in patients with relapsed or refractory extensive-stage small cell lung cancer (ES-SCLC). The company is also exploring the potential of nelmastobart in combination with capecitabine for the treatment of Stage III or higher metastatic colorectal cancer. Abion Bio is presenting preclinical results on “ABN202,” an antibody-cytokine fusion protein (ACFP) that fuses an interferon-beta variant to a tumor-targeting antibody. ACFP is a novel drug candidate that is expected to address the systemic toxicity of interferon-beta thereby offering a strong safety profile. The company will present nonclinical data confirming immuno-oncology efficacy in immunosuppressive solid tumor mouse models. Abion Bio is also exploring the potential for combination therapy with existing immuno-oncology agents.
- Company
- Reimb of Vocabria+Rekambys for HIV gains attention
- by Eo, Yun-Ho Nov 04, 2024 05:48am
- The industry’s eyes are on whether the long-acting HIV combination therapy ‘Vocabria+Rekambys’ will be reimbursed by the end of the year in Korea. According to industry sources, GSK Korea and Janssen Korea have completed the pharmacoeconomic evaluation of their HIV drugs Vocabria (cabotegravir) and Rekambys (rilpivirine) combination therapy and are awaiting the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee’s review. It remains to be seen whether the drug will be reviewed by DREC by the end of the year and progress onward. As it has been more than 2 years since the drug’s domestic approval, the industry’s eyes are on whether it will be reimbursed this time. The MFDS previously approved the two drugs in February 2022 as a combination therapy for the treatment of HIV-1 infection in adult patients who are virologically suppressed, have no history of virologic failure, and have no known or suspected resistance to either cabotegravir or rilpivirine. The Vocabria+Rekambys combination was approved in Korea as a once a month or once-every-two-month injection regimen. The advantage of the combination is convenience. Previous HIV treatments required patients to take tablet formulations once a day, so every day, but with the approval of the two injectables, patients will be able to take intramuscular injections once a month or every other month, reducing the frequency and increasing patient satisfaction. The two drugs were originally developed as oral formulations and then were developed as injectables. The long-acting injectables do not cure HIV infection, but they work by targeting white blood cells to help lower and maintain levels of the AIDS virus. The combination was approved in Europe in December 2020 after demonstrating efficacy and safety in clinical trials in groups receiving the combination once every 4 weeks or once every 8 weeks. In the clinical trials, the most frequent adverse events observed in the Vocabria+Rekambys arm were injection site reactions, headache, pyrexia, nausea, fatigue, asthenia, and myasthenia. It remains to be seen if the benefit of convenience of the combination therapy will be accepted by the health authorities and be listed for reimbursement.
- Policy
- Anticipating reimb for DPP4·SGLT2 combination drugs
- by Lee, Jeong-Hwan Nov 01, 2024 05:51am
- The government has announced a plan to address the necessity of the National Health Insurance reimbursement·expansion for two-drug combination drugs containing DPP-4 inhibitors and SGLT-2 inhibitors for the treatment of type 2 diabetes. In South Korea, reimbursement for combination drugs containing DPP-4 inhibitors and SGLT-2 inhibitors is only provided for three-drug combination therapy that involves an additional prescription of metformin. Approval of reimbursement for two-drug combination therapy, excluding metformin, is expected to offer more treatment options and convenience for diabetes patients while preventing unnecessary national health expenditures. On October 31, the Ministry of Health and Welfare (MOHW) and Health Insurance Review and Assessment Service (HIRA) responded to the question regarding the National Health Insurance coverage of type 2 diabetes treatments asked by Rep. Seo Miwha, a member of the People Power Party, during the parliamentary audit. Rep. Seo pointed out that reimbursement of combination drugs containing SGLT-2 inhibitors and DPP-4 inhibitors for treating diabetes must be approved to broaden treatment choices for diabetes. Rep. Seo stated that the current reimbursement for combination drugs is complicated for physicians to prescribe and restricts treatment choices for patients. Since May 2023, the scope of reimbursement for combination drugs containing DPP-4 inhibtors and SGLT-2 inhibitors prescribed to patients with type 2 diabetes has been expanded·changed so that it can be reimbursed when metformin is additionally prescribed. The problem is that the use of SGLT-2 inhibitors and DPP-4 inhibitors in combination or a combination drug is not covered by the National Health Insurance reimbursement. The current reimbursement policy is illogical for diabetes treatment because patients who can benefit simply from two-drug combination therapy are unnecessarily prescribed three-drug combination therapy because three-drug combination therapy is reimbursed, but two-drug combination drug are not. The HIRA responded to such criticism that they would review the need to expand the National Health Insurance reimbursement for a two-drug combination containing SGLT-2 inhibitors and DPP-4 inhibitors or a combination drug. "We will assess the safety·clinical utility of oral combination drugs for treating diabetes and communicate with related associations so that improved policy for two-drug combination drugs can be generalized," the MOHW said. "However, reimbursement expansion of diabetes drugs requires an impact on the National Health finance. We will discuss this with related associations." The HIRA explained the reason for the exclusion of two-drug combination drugs from the current reimbursement list, and they announced a plan to pursue improvements to the scope of reimbursement. "In 2018, we have written reimbursement criteria for the concurrent use of SGLT-2 inhibitors and DPP-4 inhibitors by discussion with an advisory committee. However, we retracted our stance that reimbursement of concurrent use by drug classes is appropriate and decided to maintain the current policy," the HIRA said. "After the previous discussion, we have checked the updated approvals from the Ministry of Food and Drug Safety (MFDS) and assessed the revision plan by comprehensively reviewing the latest resources and expert opinions, such as clinical practice guidelines, clinical research documents." "In addition to restricting the overall general policy for diabetes drugs, we plan to review issues related to reimbursement expansion for the combination therapy containing SGLT-2 inhibitors+DPP-4 inhibitors, prescription ease, and patient choices by discussing with related associations and experts," they added.
- Opinion
- [Reporter's View] Address Actions to drug substitution
- by Lee, Jeong-Hwan Nov 01, 2024 05:51am
- "We will foremostly activate drug substation to resolve the drug shortages issue," Cho Kyoo-hong, Minister of Health and Welfare (MOHW), promised during the parliamentary audit session commenced by the National Assembly's Small Committee for Health and Welfare. This is an answer from the MOHW director to an issue of essential medicines frequently in shortage, leading to chaos in pharmacies and lower patient access to medications. The issue has been unresolved for several years. Rep. Seo Youngseok and Rep. Nam In-soon of the Democratic Party of Korea have requested the implementation of active ingredient prescriptions for cold medicines and essential medicines to resolve the issue of frequent drug shortages. Minister Cho repeatedly answered that he would improve the current situation by enabling drug substitution rather than active ingredient prescription, as the latter requires agreements between officials. According to Minister Cho, activating drug substitution beyond the current 'lower-cost drug substitution incentive' policy by the Health and Welfare Committee requires specific administrative actions, policy establishment, and implementation. In other words, their plan cannot be attained by the way MOHW said to actively participate in the legislative session to review the National Assembly submitted bill for simplifying drug substitution procedure. Passing the plenary session of the National Assembly for the bill for implementation of drug substitution that has been submitted to the National Assembly would take several months after undergoing the legislative process. It has not been considered for the Legislation and Judiciary Small Committee for Health and Welfare. During the legislative procedure, it is unknown whether the bill would face opposite opinions, overcome oppositions, and pass all legislative hurdles. During the parliamentary audit, the MOHW answered a question about the restricted implementation of active ingredient prescriptions for cold medicines by stating, "It is the terms of the medical reform agreement. Therefore, the issue requires societal discussion and a procedural agreement." Then, the MOHW must consider detailed and active administration to implement drug substitution to resolve frequent drug shortages of cold medicines and pediatric medicines. In particular, lower-cost drug substitution must be increased because the government provides incentives to strengthen the stability of the National Health Insurance finance. According to the Health Insurance Review and Assessment Service (HIRA), the incentive for drug substitution increased temporarily during the COVID-19 pandemic. After the announcement of 'With COVID-19," it returned to that of pre-COVID-19. It shows that drug substitution policy has been conservative and passive unless drug substitution is inevitable, such as during the pandemic period. Not only during this audit but also during the previous parliamentary audit, the MOHW had agreed to the necessity of supporting drug substitution and increasing the incentive rate for lower-cost drug substitution. However, the concern is that they have not suggested a concrete method to specifically address when and how drug substitution can be actively implemented. As Minister Cho promised during the parliamentary audit that he would actively implement drug substitution to resolve the drug shortages, the MOHW must provide incentives and establish an administrative policy so that pharmacies can dispense drug substitution for drug shortages without hesitation. We must not passively wait for the legislative procedure after agreeing to the bill to simplify drug substitution, which is to be reviewed by the National Assembly. Aside from passing the bill to the National Assembly, we hope to hear about MOHW's updated policy to support drug substitution in resolving the drug shortage issue by actively implementing it.
- Company
- New AML drug Vyxeos can be prescribed at Big 5 hospitals
- by Eo, Yun-Ho Nov 01, 2024 05:51am
- The new acute myeloid leukemia drug ‘Vyxeos’ may now be prescribed at general hospitals in Korea. According to industry sources, Vyxeos (daunorubicin+ cytarabine), a treatment for adults with acute myeloid leukemia AML, has passed the drug committees (DCs) of the ‘Big 5’ tertiary hospitals in Korea including the Samsung Medical Center, Seoul National University Hospital, Seoul St. Mary’s hospital, Sinchon Severance hospital, several medical institutions in Korea including the National Cancer Center, Samsung Medical Center, Seoul National University Hospital, and Asan Medical Center, and Sinchon Severance Hospital. The company also made progress in the drug reimbursement discussion. Vyxeos passed the Health Insurance Review and Assessment Service’s Drug Reimbursement Evaluation Committee review in August. If the company succeeds in completing pricing negotiations with the National Health Insurance Service and the drug is listed for reimbursement, it is expected to quickly lead to actual prescriptions. Vyxeos was developed by an Ireland-based global pharmaceutical company Jazz Pharmaceuticals headquartered in Ireland, and Handok owns the exclusive rights to its sales in Korea. Treatment-related adult AML and AML with myelodysplasia-related changes have a poor prognosis and are associated with lower remission rates and shorter overall survival (OS) when treated with intensive chemotherapy compared to other AML. Intensive chemotherapy, the 7+3 regimen of cytarabine and daunorubicin, has remained the standard therapy for over 50 years since its introduction in the 1970s. This is why an unmet need existed in the field for some time. Vyxeos is a liposomal formulation of a fixed combination of daunorubicin and cytarabine in a 1:5 molar ratio. Vyxeos liposomes accumulate and persist at a higher concentration in the bone marrow, where they are preferentially taken up intact by leukemia cells, maximizing synergistic antitumor activity. In a Phase III trial that demonstrated the efficacy of Vyxeos, the median overall survival of patients with t-AML or AMLM-MRC who were treated with Vyxeos was 9.6 months, which was longer compared with the 6 months in patients that received the 7+3 arm. Also, 48% of the Vyxeos-treated patients achieved complete remission (CR) and complete remission with incomplete neutrophil or platelet recovery (CRi), which was higher than the 33% in the 7+3 arm. The safety profile in both arms was similar.
- Policy
- Hyundai withdraws application for Mifegymiso approval again
- by Lee, Hye-Kyung Nov 01, 2024 05:50am
- Hyundai Pharm has reportedly voluntarily withdrawn the application it had filed for the marketing authorization of Mifegymiso (mifepristone and misoprostol), which the company had applied for approval as the first abortion pill in Korea. Hyundai Pharm has requested approval for the abortion drug twice in the past, first in 2021 and then in 2023, but the Ministry of Food and Drug Safety has repeatedly asked for supplemental data, due to which the company has not been able to receive proper review. According to the MFDS’s written response to a written inquiry made during the NA Audit by the Health and Welfare Committee on the 31st, the approval and review process for Mifegymiso was recently temporarily suspended due to the company’s withdrawal. “Currently, there are some conditions that only be reviewed and approved after the Criminal Act and the Mother and Child Health Act is amended to allow drug-induced pregnancy termination and the permissible period of pregnancy termination are established by law,” said MFDS. In other words, if the Criminal Act and the Mother and Child Health Act are not amended, it would be difficult to review Mifegymiso’s effect/efficacy, and risk management plan, among others. The MFDS said, “Currently, the approval review process has been temporarily suspended (withdrawn) by mutual recognition of the applicant and the MFDS,” adding, “If the relevant laws are amended in the future and the applicant submits data accordingly, we plan to speed up the review and promptly make a decision.” Hyundai Pharm signed an exclusive marketing and distribution agreement for Mifegymiso’s supply in Korea with the UK-based Linepharma International in 2020, but the company has not been able to launch the drug in Korea due to disruptions in the approval process. Mifegymiso comes in a combination pack that contains 1 200mg mifepristone tablet and 4 misoprostol 200ug tablets. The drug inhibits the action of progesterone, the progestational hormone that sustains pregnancy, while misoprostol works to contract the uterus. It is only indicated for use in the first trimester of pregnancy, up to 9 weeks, and is typically prescribed as one tablet of mifepristone followed by 4 tablets of misoprostol 36-48 hours later. In April 2019, the Constitutional Court ruled the criminal offense of abortion (termination of pregnancy) unconstitutional, citing respect for women's right to bodily self-determination. As of January 1, 2021, abortion was effectively legalized. Upon the ruling, the Constitutional Court ordered the National Assembly to come up with an alternative legislation to reflect the decision, but 4 years have passed without such legislation being enacted. In the 21st National Assembly, 7 bills to abolish the abortion law were introduced, including amendments to the Criminal Code and the Mother and Child Health Act, but were discarded with the closure of the 21st National Assembly.