- LOGIN
- MemberShip
- 2026-05-02 02:35:23
- Company
- 'Global obesity drug market 79 times bigger than Korea'
- by Son, Hyung Min Nov 29, 2024 05:55am
- Kang-Bok Lee, Marketing & Sales Excellence Lead at IQVIA Multinational pharmaceutical companies have been achieving significant growth through a strategy that focuses on core industries and key therapeutic areas (TA)s. On the 14th, KoreaBIO held a seminar on trends and prospects of the bio-industry at Yangjae El Tower. On this day, Kang-Bok Lee, Marketing & Sales Excellence Lead at IQVIA, shared the trends of the global pharmaceutical industry. According to Lee’s data, the global pharmaceutical market was worth USD 1.44 trillion (about KRW 191.6 trillion) last year. This is 79.1 times the size compared to Korea's market, which is USD 18.2 billion (about KRW 25.5 trillion), The growth of the global pharmaceutical market was driven by diabetes, obesity, anticancer, and autoimmune disease drugs. In particular, GLP-1 class diabetes and obesity drugs showed remarkable growth. Eli Lilly's GLP-1 diabetes drug Mounjaro generated KRW 6.48 trillion in sales last year, up 971.2% from 2022. Launched in May 2022, Mounjaro grew rapidly last year after generating KRW 640 billion in sales the same year. Novo Nordisk's GLP-1 class of diabetes drugs also had a strong year. Ozempic generated sales of KRW 5.48 trillion last year, up 52% from the previous year. Sales of Rybelsus, an oral treatment with the same ingredient generated KRW 1.44 trillion, up 140% from 2022. The obesity drug Saxenda generated KRW 1.225 trillion in sales last year, up 9.8% year-on-year. Wegovy surpassed KRW 300 billion in sales last year despite facing supply challenges. "The growth of global innovation brands is being driven by a small number of countries, products, and companies. Premium, innovative new drugs such as Keytruda and obesity treatments have driven growth in the global pharmaceutical industry." Global pharmaceutical companies steadily launch new drugs..."Expect to focus on core industries and TA" He expects an average of 70 new drugs to be launched between next year and 2028, with anticancer and obesity drugs driving growth. "Immuno-oncology drugs like Keytruda have a wide range of indications. The development of one drug has the effect of developing multiple therapies. Therefore, multinational pharmaceutical companies are showing a trend of new drug development that focuses on core industries and core therapeutic areas." "Oncology and obesity are expected to be the fastest growing segments. Oncology is expected to grow at a CAGR of 15% and obesity at a CAGR of 26% through 2028." In particular, Lee noted that companies that focus on one area, such as Mounjaro, Wegovy, SKYRizi, and Keytruda, are seeing strong growth, driving label expansions and ingredient re-engineering. "Novo Nordisk, Lilly, AbbVie, and MSD are companies that have seen success by focusing on a single therapeutic area. We believe that their products will continue to lead the global pharmaceutical market in the future,” said Lee. "Just as GLP-1 obesity drugs were developed as diabetes treatments and then became highly successful as obesity treatments, we expect re-engineering and labeling to emerge as a global R&D trend in the future."
- Policy
- Concerns about National Bio Committee's foreign pharma exec
- by Lee, Jeong-Hwan Nov 29, 2024 05:54am
- President Yoon Suk Yeol has been appointed as chair of the National Bio Committee. The National Bio Committee, chaired by President Yoon Suk Yeol, is set to launch in December. However, reports indicate that a global pharmaceutical company executive has been included as a committee member, raising concerns. The committee comprises up to 40 members, including the chair, vice-chair, various ministers, and officials from the National Security Office. One of the appointed committee members is an executive from a foreign pharmaceutical company. Given that the committee's primary role is to formulate national strategies for the pharmaceutical and biotechnology industries, critics have argued that appointing an executive from a major multinational pharmaceutical company has the potential for government policies or confidential information to be leaked externally. According to the pharmaceutical industry on November 27, the list of committee members of the National Bio Committee included an executive from a multinational pharmaceutical company. President Yoon Suk Yeol was appointed as chair of the National Bio Committee, and Lee Sang-yeop, vice president of the Korea Advanced Institute of Science and Technology (KAIST), will serve as vice-chair. Civilian committee members include 20 experts, including Kim Bit-naeri, Director of the Institute for Basic Science, Koh Han-seung, CEO of Samsung Bioepis, and Kim Young-tae, President of Seoul National University Hospital. The first meeting is scheduled for next month. An issue has arisen from including an executive from a multinational company who has worked for nearly 20 years in a global pharmaceutical company. Criticism has been raised about including an executive from a foreign-based company representing global big pharma to the National Bio Committee for the Korean pharmaceutical industry is inappropriate. The National Bio Committee is responsible for reviewing South Korea's overall biotechnology policies, including research and development (R&D) and new drug approvals, to foster the high-tech pharmaceutical and biotechnology industry as a next-generation key sector. The committee comprises up to 40 members, including the President as Chair, a vice chair, ministers from various government departments, the senior secretary for Science and Technology, the Third Deputy Director of the National Security Office, and private experts from the field and academia. Criticism have been raised about including an executive from a multinational pharmaceutical company. Critics argue that such an appointment contradicts the committee's purpose of promoting domestic pharmaceutical innovation. They state that allowing a global "Big Pharma" executive to participate in discussions about policies shaping South Korea's pharmaceutical R&D, regulatory·pricing strategies undermines the committee's intent. Concerns also extend to potential security risks, as sensitive government policies or information on the pharmaceutical and biotechnology sectors critical to national security could be inadvertently·deliberately leaked externally. "The National Bio Committee is responsible for unveiling, formulating, discussing, and deciding on South Korea's R&D strategy in the pharmaceutical and biotechnology sectors. Including a multinational pharmaceutical company executive in a committee chaired by the President Yoon, where vaccine and pharmaceutical security issues may be discussed, is illogical. It is comparable to handing over our home territory," a representative from the domestic pharmaceutical industry said.
- Company
- Sales rights for Viviant·Tuvero·Akarb to be transferred
- by Kim JiEun Nov 28, 2024 05:55am
- Due to changes in sales rights for blockbuster items that are frequently dispensed, the pharmaceutical supply and demand are expected to be affected in the end-of-year period and early 2025. According to the pharmaceutical distributor industry on November 26, several pharmaceutical companies are expected to change their distributors at the end of this year or early next year. Medication with confirmed changes to sales rights is Pfizer's Viviant Tab. Pfizer Korea has recently sent an official letter to wholesale distributors, hospitals, and pharmacies notifying 'The changes to the supply chain for Viviant.' Pfizer stated in the official letter that Pfizer will be responsible for distributing Viviant Tab 200 mg, previously distributed by Handok, starting December 1. The company requested that any inquires about returns for remaining stock from previously distributed items to be maded to Handok, and any inquiries after December 1 be directed to Pfizer. Pfizer also announced that the distribution of Caverject Injection 10 ug and 20 mg, previously distributed through Novamedix will be changed to direct distribution effective December 1. Pfizer stated any inquiries about remaining distributing stock for this item could be made to Novamedix, and inquiries related to distribution after December 1 should be directed to Pfizer. In addition to these items, more products with expected changes to sales rights in early next year. Sales rights for most of Boryoung's items will be changed, and the company seems to be informing its pharmaceutical distributors. The wholesale distributor said that Daewon Pharmaceutical has been responsible for distributing Boryoung's Tuvero Tab and Akarb Tab, but Boryung will directly distribute them starting next year. Yungjin Pharmaceutical has been distributing Boryung Buspar Tab since 2018. Boryung is highly like to distribute the drug directly next year. As changes to distributors have been announced, it is expected to affect the end-of-year pharmaceutical supply and demand. "Special circumstances such as changes to the distributor are likely to affect the supply and demand. The effective date may be postponed beyond what the pharmaceutical companies have previously announced," a pharmaceutical wholesale employee said. "We must prepare for possibilities such as difficulty in returning remaining stocks and unstable supply and demand for such items in early next year."
- Policy
- KIPO opposes to objections to new drug patent extended cap
- by Lee, Jeong-Hwan Nov 28, 2024 05:55am
- The Korean Intellectual Property Office (KIPO) The Korea Research-based Pharma Industry Association (KRPIA) and others have submitted an opinion disagreeing with the proposed legislation that would introduce a maximum cap for terms of patent protection for new drugs. The proposed legislation is intended to facilitate an earlier market launch for Korea-made new drugs. The Korean Intellectual Property Office (KIPO) stated its opposition to this issue at the 21st National Assembly. At that time, KIPO stressed that the original draft instead of a revised Patent Law that would limit the number of patents to a single (1 patent). A similar proposed legislation has been submitted to the 22nd National Assembly. Therefore, it is highly likely that KIPO would state an opinion agreeining with the original draft. On November 26, the Trade, Industry and Energy Small and Medium Venture Business Committee is scheduled to hold a small committee for legislative review. Rep. Koh Dong-jin, a member of the People Power Party, will review the draft of the revision to the Patent Law. Rep. Koh Dong-jin's proposal contains key messages, including ▲legislating a 14-year maximum (cap) on the effective duration of patent rights and ▲ limiting the number of patents eligible for extension to a single patent per drug. The proposed legislation is based on concerns that the current patent term extension system under Korean patent law differs from those in advanced countries such as the United States and Europe, potentially reverse discriminiate the domestic pharmaceutical industry. KRPIA "It worsens individual access to medications…disagreeing with the legislation" In Rep. Koh Dong-jin's draft of the revision, the Korean Research-based Pharmaceutical Industry Association (KRPIA), the Japan Pharmaceutical Manufacturers (JPMA), the Pharmaceutical Research and Manufacturers of America (PhRMA), the European Federation of Pharmaceutical Industries and Associations (EFPIA), the Biotechnology Innovation Organization (BIO), the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), and the Patent Information Initiative for Medicines (Pat-INFORMED) have shared opposing views. The opposing group argues that adopting only selective features of overseas practices regarding patent term extensions, rather than improving the entire system, could worsen public access to medicines and disrupt international collaboration. Specifically, regarding introducing a patent term cap, they highlight that South Korea recognizes only the 'domestic clinical trial period' for patent extensions, excluding the 'overseas clinical trial period' from the calculation of extension durations. They also argued that the scope of extended patent rights in South Korea is narrower compared to major countries. Specifically, there are conflicting court rulings on whether extended patent rights are limited to the indications approved at initial approval. It also remains to be seen whether the extended rights apply to all approved uses of the extended product. They also stated that there is almost no chance of appealing a rejection decision concerning patent term extension applications in South Korea. Before Rep. Koh Dong-jin's draft of the revision, KRPIA and others have previously opposed the bill proposed by Democratic Party Rep. Jung Il-young during the 21st National Assembly, which was abandoned because the legislative term expired. KIPO disagrees by citing U.S. examples During the 21st National Assembly, KIPO presented counterarguments to the objections raised against Rep. Jung Il-young's bill, advocating for the retention of the original proposal. KIPO will likely maintain the same position regarding Rep. Ko Dong-jin's proposal in the 22nd National Assembly. KIPO states that it cannot accept the opposing arguments, including the claim that South Korea does not include overseas clinical trial periods in calculating patent extension durations and the assertion that the scope of extended patent rights in South Korea is unclear compared to major countries. However, regarding the opposing argument that there is no opportunity to appeal rejection decisions on extension applications, KIPO partially accepted this concern, suggesting the need to review the introduction of a related system. KIPO disputed the claim that South Korea excludes overseas clinical trial periods and uniformly excludes the Ministry of Food and Drug Safety's (MFDS) approval supplementation period when calculating patent extension durations, stating, "Other countries do the same." KIPO explained, "The U.S. system, similar to South Korea's patent extension calculation method, includes only half of the clinical trial period and does not recognize overseas clinical trial periods." It emphasized that "maintaining the current practice for calculating extension durations is appropriate." "Considering the Supreme Court ruling, which excludes the applicant's compensation period during the MFDS's review of approval documents, we believe that the current method for calculating extension durations should remain unchanged," KIPO stated. "The length of the extension period is primarily influenced by variations in domestic clinical trial durations, which are affected by factors such as a country’s drug development capabilities and the size of its pharmaceutical market, rather than differences in extension systems." Regarding the opposition's argument that the scope of patent rights should be expanded to include the 'active ingredient' and included into patent law, KIPO countered by saying that the legislation should proceed as originally proposed. "The Supreme Court, considering the protection of patent holders and the purpose of the extension system, has ruled that the scope of extended patent rights should be determined based on the 'active ingredient and drug use based on the Patent Law,' similar to major countries," KIPO stated. KIPO partially agreed with the opposing view that there is no opportunity to contest rejection decisions on extension applications. "It is necessary to establish various concrete measures to contest rejection decisions to secure procedural rights for patent holders," KIPO stated. "However, these remedies should be introduced alongside measures to address rejection decisions related to delays in registration, which will require a more thorough review."
- Policy
- Trial for targeted drug 'VERT-002' for NSCLC begins in KOR
- by Lee, Hye-Kyung Nov 28, 2024 05:55am
- A clinical trial of 'VERT-002,' an innovative targeted treatment for degrading a c-MET gene, will begin in South Korea. C-MET gene is known as one of the cancer-inducing factors of non-small cell lung cancer (NSCLC). On November 27, the Ministry of Food and Drug Safety (MFDS) has granted approval of 'a first-in-human (FIH) multi-center, dose-escalation, dose-expansion Phase I/II clinical trial with VERT-002 for patients with locally advanced or metastatic solid tumors, including NSCLC.' This clinical trial will be conducted at the Asan Medical Center in Seoul and the Severance Hospital. The VERT-002 candidate product was developed by Vertical Bio, a Swiss-based biotechnology company specializing in developing antibodies for cancer treatment. The French pharmaceutical company Pierre Fabre Laboratories acquired Virtual Bio last year, adding a pipeline of cancer agent discovery. A degrader of MET, VERT-002 is a monoclonal antibody with a unique and differentiating mechanism of action. The company anticipates that VERT-002 will likely become a best-in-class treatment for cancer associated with MET alterations. Global clinical trials for the candidate are being conducted worldwide, including in South Korea. The Phase ½ clinical trial for a first-in-human (FIH) multi-center, dose-escalation, and dose-expansion in patients with NSCLC harboring MET alterations began in October. NSCLC is the most common form of lung cancer, accounting for about 85% of newly diagnosed lung cancer cases. Also known as the hepatocyte growth factor receptor (HGFR), MET is a factor regulating tumor genes in a patient sub-group with NSCLC. 1-4 MET exon 14 (METex14) skipping mutations and MET amplification have been identified as the major tumor gene, and MET amplification appears to be mechanisms of tolerance to selected targeted treatment. With its unique and differentiating mechanism of action, VERT-002 works by targeting clinically proven tumor-inducing factors and induces degradation of the c-MET tumor gene. Meanwhile, Pierre Fabre recorded sales of 467 million euros in 2020 just with anticancer agents.
- Policy
- Handok’s thrombocytopenia drug Doptelet approved in KOR
- by Lee, Hye-Kyung Nov 28, 2024 05:54am
- Handok's new rare disease drug ‘Doptelet (avatrombopag)' was approved in Korea on the 26th. The Ministry of Food and Drug Safety approved Doptelet as a treatment for thrombocytopenia for patients with liver disease. Handok is reaping the benefits of forging a strategic partnership with the global biopharmaceutical company Sobi-Handok last year. At that time, the company decided to introduce two new drugs, Empaveli (pegcetacoplan) and Doptelet, a rare disease treatment, into the country, both of which have now been approved in Korea. Doptelet is an orally administered second-generation thrombopoietin receptor agonist (TPO-RA) approved in the U.S. for the treatment of thrombocytopenia in adult patients with liver disease. Patients with moderate-to-severe thrombocytopenia are at increased risk for serious or fatal bleeding events. Patients with thrombocytopenia are commonly known to receive platelet transfusions to increase their platelet count prior to surgery. The approval of Doptelet was based on safety and efficacy data from two Phase III clinical trials on 435 patients. Doptelet is the first oral treatment option for patients with chronic liver disease which offers the advantage of eliminating the need for platelet transfusions prior to surgery for many patients. Meanwhile, Handok formed a joint venture with Sobi in April this year, following a strategic partnership last year. Sobi-Handok will be responsible for developing and selling Sobi’s rare disease drugs in Korea, with a 49:51 shareholding ratio between Handok and Sovi, respectively.
- Policy
- Lagevrio prescription age raised from 60 to 70
- by Kang, Shin-Kook Nov 28, 2024 05:54am
- The prescription age for the government-supplied COVID-19 treatment, Lagevrio, will be raised from 60 to 70 from the 27th. According to the Korea Disease Control and Prevention Agency (KCDA), the three COVID-19 drugs (Paxlovid, Lagevrio, Veklury Inj) that were initially supplied by the government, were planned to be included in Korea’s national health insurance system and transitioned to a general drug status within the year. Accordingly, Paxlovid and Veklury Inj have already been granted marketing authorization in Korea and have been covered by Korea’s health insurance since the 25th of last month. Change in prescription target for Lagevrio On the other hand, Lagevrio has not completed the marketing authorization process in Korea yet and is still being used under emergency use authorization. A considerable period of time may be needed before the approval process is resumed due to the need for additional evidence. The KDCA stated that although a marketing authorization is required to include Lagevrio in the general medical system, it plans to continue supplying Lagevrio on a limited basis within the scope of the government inventory for the time being, considering the COVID-19 winter epidemic and expert opinions. However, the government will limit prescriptions to those for whom domestic and international studies have shown increased efficacy, and will only allow Lagevrio prescriptions to those aged 70 years and older. National support for Lagevrio will continue throughout the winter season, after which the government will monitor the COVID-19 pandemic and comprehensively review the situation, including adjustments to the COVID-19 crisis phase, which is currently in the “interest” stage of concern. Youngmee Jee, Commissioner of KDCA, said, “We will continue to work to ensure a stable supply of COVID-19 drugs, including Lagevrio, to effectively protect high-risk populations from the COVID-19 winter pandemic.”
- Company
- Lilly appoints Stacie Liu to head the CardioMetabolic Health
- by Eo, Yun-Ho Nov 28, 2024 05:54am
- Stacie Liu, Senior Director, CardioMetabolic Health Business Unit, Lilly Korea Lilly Korea has named Stacie Liu (37), as the new Senior Director of its CardioMetabolic Health Business Unit On the 1st, Lilly Korea announced that it had appointed Stacie Liu as the Senior Director of its CardioMetabolic Health Business Unit. In this role, Liu will be responsible for setting and executing the business strategy for Lilly's diabetes and obesity portfolio in Korea, including the company's type 2 diabetes and obesity treatment, Mounjaro. Lilly's decision is notable in that Liu is a young, foreign, female leader. This is in line with Lilly's culture, which believes in the power of diversity, equity, and inclusion. As of 2023, one in two (49%) of Lilly’s executives were women. This represents a 4%p increase over 4 years from 2019. Five of the 12 Board of Directors are also women, with their ages ranging from early 50s to late 60s. “At Lilly, our goal is to help people live better everyday lives through dedicated and exploratory development of new medicines, and we need the best and brightest minds with the most creative ideas to address the world's most pressing health challenges,” said Liu. “Lilly's success today is founded on the corporate culture that embraces diversity and values equity. Based on this culture, Lilly Korea will be committed to delivering life-changing medicines promptly to patients in Korea.” Liu has previously held positions at Lilly's Shanghai office and U.S. headquarters and has a bachelor's degree in International Relations and Affairs from Fudan University in Shanghai, China. Fudan University is a world-renowned university, ranked 31st in the world and 3rd in China as of 2022.
- Company
- ‘The new ATTR-CM treatment can improve patient survival’
- by Whang, byung-woo Nov 27, 2024 05:50am
- With the possibility for reimbursement coverage for the ultra-rare disease, transthyretin amyloid cardiomyopathy (ATTR-CM) rising, excitement is also rising in the clinical site. Due to the short life expectancy of ATTR-CM - 2-3.5 years after diagnosis – and no clear treatment option available, the benefits of a new treatment option are expected to be significant. As an extremely rare disease with a small number of patients, challenges still remain in identifying and diagnosing patients early. Dailypharm met with Dr. Junho Hyun, Professor of Cardiology at Asan Medical Center in Seoul, to discuss Korea’s treatment environment for ATTR-CM and the changes made since the reimbursement of its treatment option. Junho Hyun, Professor of Cardiology, Seoul Asan Medical Center ATTR-CM is a rare, progressive disease where a protein called transthyretin (TTR) misfolds and forms harmful amyloid deposits in the heart. Although it is caused by genetic issues, it can also occur with aging. In particular, ATTR-CM is a rare disease that is most commonly found in patients over the age of 65, so experts believe its patient population could grow as Korea enters an aged society. "The exact prevalence of ATTR-CM patients in Korea is unknown, but what is known is that it is expressed in a different form in Korea than in other countries," said Professor Hyun. "In the United States, the number of affected patients was identified through the patient registration system and the introduction of its treatment was also fairly quick. Korea needs to improve the overall treatment environment for ATTR-CM patients, including its management, diagnosis, and treatment," said Dr. Lee. "The most common cause of cardiac amyloidosis is AL amyloidosis, followed by ATTR amyloidosis, so we test for these 2 possibilities at the time of initial diagnosis," he said. "We also use nuclear scintigraph to diagnose ATTR-CM, and then genetic testing to differentiate between the hereditary and wild-type. In some cases where nuclear scintigraphs are not available, we test the heart tissue." According to Professor Hyun, the diagnosis of ATTR-CM patients in the United States is slowly increasing. He believes that this is due to increased awareness, which, like other diseases, has led to the development of treatments. In the past, patients with hereditary ATTR-CM often died suddenly without knowing the cause of their disease due to low awareness, but early identification of patients has led to higher diagnosis rates the patients’ families can also be diagnosed and treated. From a treatment standpoint, prior to the approval of Vyndamax (tafamidis) in 2020, the doctors had lacked options - using diuretics to relieve symptoms or carrying out heart and liver transplants. However, despite the emergence of a treatment, its high price remains a hurdle. Recently, Vyndamax’s reimbursement agenda passed the review of the Drug Reimbursement Evaluation Committee of the Health Insurance Review and Assessment Service 4 years after its approval, leaving only drug price negotiations with the National Health Insurance Service. "With a median survival of only 2-3 years, patients with ATTR-CM despair due to the fact that treatment options are available but unaffordable. It's very unfortunate that there is a cure, but it's not covered by insurance, which limits treatment," added Hyun. While he understands the limitations that exist due to government budgets, Hyun believes that reducing mortality through early treatment of ATTR-CM patients is more cost-effective. "In Korea, the deterioration of ATTR-CM symptoms slowed down or even improved in some cases in patients who used the drug through patient support programs. However, it is less effective if the timing of treatment is delayed due to delayed access and the disease progresses, so it is necessary to set reimbursement standards so that the drug can be used from an early stage in addition to patients with severe symptoms." "ATTR-CM is like cancer in that it has a very short median overall survival, so it is very unfortunate that its treatment options are being restricted because it is a rare disease with a small number of patients," said Professor Hyun. "The medical gap that exists due to the high treatment cost is a social problem, which requires serious government consideration.”
- Company
- Active development of new drugs for myasthenia gravis
- by Son, Hyung Min Nov 27, 2024 05:50am
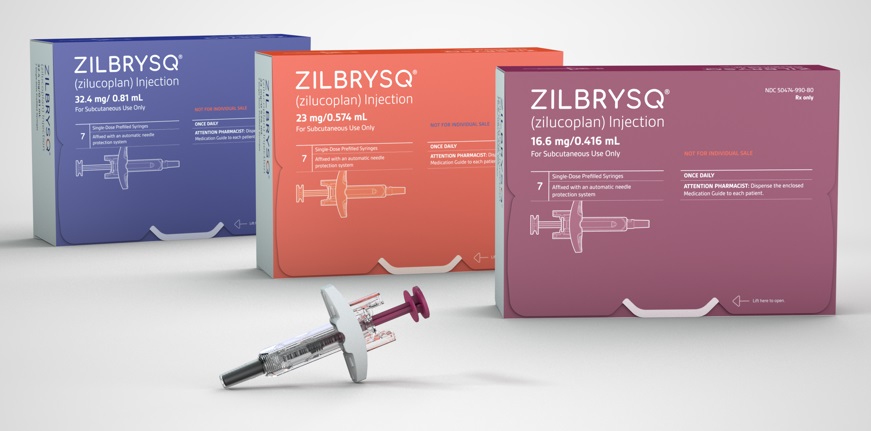
- Competition in the pharmaceutical market is heating up to secure myasthenia gravis indication. UCB's new drug was approved in South Korea, and Janssen finished the phase 3 trial, aiming to acquire regulatory approvals worldwide. In South Korea, Handok seeks to enter the market with its acquired new drug. HanAll Biopharma is conducting a Phase 3 trial and investigating the potential of commercialization. According to industry sources on November 27, UCB's 'Zilbrysq' was approved in South Korea on November 21. Zilbrysq can be used as an add-on to standard therapies, such as cholinesterase inhibitors, steroids, and immune checkpoint inhibitors. This therapy is a complement C5 inhibitor and works by inhibiting the complement-mediated damage to the neuromuscular junction (NMJ). Product photo of UCBDue to this approval, Zilbrysq became the first treatment that can be self-administered, unlike other domestically approved treatments for adult myasthenia gravis. 'Ultomiris,' a complement C5 inhibitor used to treat existing myasthenia gravis, is an intravenous injectable that requires administration at hospital visits every 8 weeks. Generalized myasthenia gravis (gMG) is a chronic autoimmune rare disease in which nerves fail to transmit signals to muscles, leading to skeletal muscle weakness. This disease induces fluctuating weakness of systemic muscles, affecting daily activities such as standing, swallowing, and breathing. Zilbrysq's approval is based on the Phase 3 'RAISE' study. The study evaluated the efficacy and safety of Zilbrysq in 174 adult patients with anti-acetylcholine receptor (AchR) positive gMG. The primary endpoint was the 'Myasthenia Gravis-Activities of Daily Living (MG-ADL)' for gMG. Typically, an MG-ADL score of 2 or higher or a Quantitative Myasthenia Gravis (QMG) score of 3 or higher indicates clinically significant improvement. The clinical results showed that the Zilbrysq group showed a clinically significant reduction by a total score of 4.39 from baseline at 12 weeks compared to the score of 2.30 of the placebo group. In addition to working on Zilbrysq, UCB is preparing to expand approvals of 'Rystiggo,' UCB's additional treatment for myasthenia gravis, in countries. Rystiggo obtained the U.S. Food and Drug Administration (FDA) approval in June 2023 and was also approved in Europe in January of this year. Janssen has recently completed the Phase 3 trial of 'nipocalimab,' a new drug candidate for myasthenia gravis and the company is preparing to obtain approval. Janssen applied to obtain approvals from the FDA in August and the European Medicines Agency (EMA) in September. Nipocalimab works by a novel mechanism that blocks the Fc receptor (FcRn), a protective immunoglobulin G (IgG) receptor. This mechanism reduces IgG antibodies that cause the disease and inhibits their recycling process. Nipocalimab binds to FcRn to prevent the degradation of IgG antibodies. The Phase 3 Vivacity-MG3 study demonstrated the efficacy of nipocalimab in combination with standard therapy compared to the placebo. The clinical study confirmed a statistically significant result, with nipocalimab recording an MG-ADL score of 4.70. Also, nipocalimab improved the myasthenia gravis patients' muscular strength and function compared to the placebo. Handok·HanAll aim to commercialize their treatments with a similar mechanism to nipocalimab In South Korea, Handok is preparing for the new drug approval for the treatment of myasthenia gravis. In August. Handok signed an agreement with argenx, the Belgium-based developer of Vyvgart. Handok is now responsible for domestic approval registration, applying for reimbursement, and exclusive distribution. Like nipocalimab, Vyvgarth is a new drug targeting the FcRn. It has been approved in several countries, including the United States, Europe, the United Kingdom, Israel, and China, for treating adult patients with generalized myasthenia gravis. The clinical efficacy of Vyvgarth was demonstrated in the Phase 3 clinical trial named ADAPT. It was shown that Vyvgarth recorded a 68% responder rate based on the MG-ADL scale, significantly higher than the 30% of the placebo. Additionally, Vyvgarth demonstrated an improved score compared to the placebo group on the QMG scale. HanAll Biopharma is also developing an FcRn antibody treatment candidate, batoclimab (HL161), as a subcutaneous injectable. In December 2023, HanAll Biopharma's Chinese partner, Harbour BioMed, secured Phase 3 clinical trial results and submitted an approval application to Chinese regulatory authorities. Harbour BioMed said that batoclimab improved symptoms in myasthenia gravis patients based on MG-ADL and QMG scales. HanAll Biopharma is conducting a phase 3 trial for batoclimab in the United States. HanAll Biopharma's U.S. partnering company Immunovant finished registering patients for the Phase 3 clinical for myasthenia gravis. Immunovant plans to present top-line results of the phase 3 clinical trial in the first half of 2025. HanAll Biopharma and Immunovant plan to investigate the potential of batoclimab for various autoimmune disease indications, including thyroid eye disease and chronic inflammatory demyelinating polyneuropathy. In addition to developing batoclimab, both companies will develop new drugs for additional myasthenia gravis indication. HanAll Biopharma and Immunovant are conducting a phase 1 trial of IMVT-1402, a new FcRn antibody drug candidate product. Unlike conventional FcRn antibody treatments, IMVT-1402 is not known to affect the LDL-cholesterol level. In the Phase 1 clinical trial, subcutaneous injection of IMVT-1402 and placebo are administered to randomized healthy adults. Each patient group receives a single-dose or multiple doses with a dose-escalation.