- LOGIN
- MemberShip
- 2026-05-01 23:17:59
- Policy
- Boryung’s Pomalyst generic first to be reimbursed in Korea
- by Lee, Tak-Sun Jan 20, 2025 05:54am
- Boryung will be the first in Korea to receive reimbursement for its generic version of Pomalyst (pomalidomide, BMS), a multiple myeloma treatment. As a product from an innovative pharmaceutical company, the drug also receives premium pricing. According to industry sources on the 17th, four dosage forms (1, 2, 3, and 4 mg) of Boryung's ‘Pomalikin Cap’ that contains pomalidomide will be listed for reimbursement on February 1. The insurance price ceiling was set at KRW 132,184 for the 1mg, KRW 132,493 for the 2mg, KRW 134,140 for the 3mg, and KRW 135,271 for the 4mg formulation. This is 53.55% of the highest list price, plus a 68% premium granted as a product by an innovative pharmaceutical company. Pomalikin Cap is the first generic version of the original Pomalyst Cap. This month, the cap was adjusted due to the termination of the reimbursement risk-sharing agreement for Pomalikin Cap. Before the adjustment, the ceiling price of the 1mg product was KRW 356,691, while the adjusted price was KRW 194,389, a 44.6% price adjustment. Compared to Pomalikin Cap 1mg, the adjusted price of the original product is around KRW 60,000 higher. So Boryung’s first generic can be seen as competitive in terms of price. The premium pricing markup for Pomalikin Cap will end in February 2026, and the price will fall to 53.55% of the highest price thereafter. Meanwhile, Pomalyst is a blockbuster product that posted sales of KRW 22.8 billion as of 2023 according to IQVIA data. It is indicated ▲for the treatment of multiple myeloma patients who have received at least one prior therapy, including lenalidomide, in combination with bortezomib and dexamethasone; and ▲for the treatment of relapsed or refractory multiple myeloma patients who have received at least two prior therapies, including lenalidomide and bortezomib, in combination with dexamethasone. By securing a pomalidomide-based treatment in addition to lenalidomide, Boryung is expected to strengthen its position in the multiple myeloma treatment market,
- Company
- Ziihera receives orphan drug designation in Korea
- by Eo, Yun-Ho Jan 20, 2025 05:54am
- The first HER2 bispecific antibody drug Ziihera has received an orphan drug designation in Korea. The Ministry of Food and Drug Safety (MFDS) recently announced so through the first orphan drug designation in the new year. Its specific indication is for the treatment of adult patients with previously treated unresectable locally advanced or metastatic HER2-positive (IHC3+) biliary tract cancer. Ziihera (zanidatamab), a bispecific antibody that targets the HER2 gene, received accelerated approval from the U.S. FDA in November last year after demonstrating efficacy in patients with biliary tract cancer. This is the first time that a bispecific antibody targeting the HER2 gene has been used as a second-line treatment option for patients with biliary tract cancer who are identified as HER2-positive (IHC 3+). Biliary tract cancer is a fatal disease with a poor prognosis and a low five-year survival rate of less than 5% for metastatic disease. The company demonstrated Ziihera’s efficacy through the single-arm phase IIB HERIZON-BTC-01 study. In the trial, the drug met the primary endpoint of confirmed objective response rate (cORR) by independent central review (ICR). The objective response rate was 52%, and the median duration of response (DOR) was 14.9 months. The safety profile of Ziihera was demonstrated in the HERIZON-BTC-01 trial in 80 patients. During the study, 53% of patients treated with Ziihera experienced an adverse event. The most common adverse events were diarrhea, infusion-related reactions, abdominal pain, and fatigue. Serious adverse events occurring in 2% or more of patients were biliary obstruction, biliary infection, sepsis, pneumonia, diarrhea, gastric obstruction, and fatigue. The trial results were presented at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting and published in The Lancet Oncology. Long-term follow-up data showing improvement in the duration of response were reported at the 2024 ASCO Annual Meeting. Meanwhile, BeiGene has the domestic rights to Ziihera. The drug is currently being studied in multiple cancers, including Phase III trials in gastroesophageal adenocarcinoma (GEA) and metastatic breast cancer (mBC). It is also currently being studied in the Phase III HERIZON-BTC-302 trial, which compares the combination of Ziihera and standard of care (Soc) to Soc alone in patients with HER2-positive biliary tract cancer.
- Company
- [Reporter' View] Results from J.P. Morgan Conference
- by Lee, Seok-Jun Jan 20, 2025 05:53am
- The Annual J.P. Morgan Healthcare Conference (hereafter J.P. Morgan Conference) has ended. It is the largest funding event in the pharmaceutical and biotech industry and is held in January every year. Many Korean companies have also participated in the event, to the extent that many key R&D people in the Korean pharmaceutical and biotech industry were said to be not present in Korea. According to the J.P. Morgan Conference report, almost 30,000 one-on-one business meetings requested at this year's event were held, where 12,000 agreements have been met. The total market capitalization of 531 companies at the official presentation sessions is US$ 9.6 trillion. Consequently, the J.P. Morgan Conference presents an opportunity for Korean pharmaceutical and biotech companies. According to the J.P. Morgan Conference report, almost 30,000 one-on-one business meetings requested at this year's event were held, where 12,000 agreements have been met. The total market capitalization of 531 companies at the official presentation sessions is US$ 9.6 trillion. Consequently, the J.P. Morgan Conference presents an opportunity for Korean pharmaceutical and biotech companies. Companies referred to as conglomerates in the pharmaceutical industry have unveiled their specific accomplishments. For instance, Yuhan presented its latest data on the new lung cancer drug, Leclaza, through its partner, Johnson & Johnson (J&J). Joaguin Duato, CEO of J&J, said, "A combination therapy of Leclaza and Rybrevant demonstrated results of extending three-year life-expectancy of lung cancer patients over a year. It is a difference that may bring changes to the treatment paradigm." UK-based AstraZeneca's Tagrisso (osimertinib), the standard therapy used in patients with EGFR-mutated non-small cell lung cancer (NSCLC) has the median overall survival (mOS) of approximately 3 years. The combination therapy of Leclaza and Rybrevant is expected to have a mOS of over 4 years. J&J projected the yearly sales goal for Leclaza+Rybrevant to be over KRW 7 trillion. Samsung Biologics announced that it will begin the antibody-drug conjugate (ADC) service in the first quarter of this year. The company aimed to provide top-level Contract Development and Manufacturing Organization (CDMO) services in the ADC field. Moreover, the company announced the building of Plant 6 facility this year. Other companies, including Celltrion, Lotte Biologics, Hugel, and SK Biopharmaceuticals, presented their R&D vision. These companies have shared their success at the J.P. Morgan conference. In contrast, some companies showed a different stance before and after the conference. It is difficult to estimate, but based on yearly trends, more than half of the companies that promoted before the conference did not issue additional press reports afterwards. Despite using promotional keywords, such as promoting technology transports, disclosing growth strategies, conducting big pharma meetings, and establishing facilities, these companies failed to release feedback after attending the J.P. Morgan Conference. The J.P. Morgan Conference has ended. Companies must release their accomplishments following the conference if they genuinely wish to be acknowledged for their company values. Rather than issuing promotional press releases beforehand, they should share objective results afterward. If one issues a promotional subject, providing feedback is essential. Additionally, companies must announce their accomplishments. Disclosable materials are limitless, depending on how companies view them. This week is the best time for companies to showcase their successes at the J.P. Morgan Conference.
- Opinion
- [Reporter's View] Improving rare disease care environment
- by Eo, Yun-Ho Jan 17, 2025 05:54am
- A few patients can only make a small noise. Despite the need for the improvement of the rare disease treatment environment, which has risen year after year, the voice of its patients that implore difficulties has never ceased to exist. In particular, there are cases where a drug is available, but due to the small number of eligible patients, it is difficult to prove the drug’s cost-effectiveness and predict the potential financial expenditures, rendering the drug’s reimbursement listing difficult in Korea. According to the 'Status of Severe Diseases that were applied Special Calculation' released by the Health Insurance Review and Assessment Service, rare and incurable diseases accounted for 37%, 32%, and 33% of the distribution of the doctors’ office personnel, medical expenses, and reimbursement expenses per severe disease in 2022, respectively. From 2012 to 2019, the relative proportion of reimbursement paid for rare and incurable diseases among the severe diseases that were applied special calculation was around 33%. However, the government has a different story. The average reimbursement rate for rare disease drugs was 85.3% (2016-2020) and 100% in 2020. This would seem to indicate that patient access to rare disease drugs seems picture-perfect. However, why isn’t it so in reality? Results published by HIRA are based on the reimbursement rates for drugs that have undergone the review and evaluation process, which are different from the reimbursement rates of approved rare disease drugs. In other words, they exclude various factors such as applications of drugs that dropped out or made voluntary withdrawals. In order to increase the true reimbursement rate of rare disease drugs, the utilization of risk-sharing agreements and the special economic evaluation exemption system must increase. Rare diseases are those with a prevalence of 20,000 or fewer people, or where the number of patients is unknown due to difficulty in diagnosis. It is often difficult to conduct clinical trials due to the small number of patients. Due to the small number of patients, it is difficult to expect profitability in the market, making it difficult to actively develop new drugs, and even if a new drug is successfully developed, it is difficult to prove its cost-effectiveness through cost-effectiveness analysis. As a solution to this, the industry has been advocating the expansion of the pharmacoeconomic evaluation exemption system. The industry has been advocating for the expansion of the exemption system, such as applying the exemption system even if the drug is approved with placebo-controlled trial data if there is no alternative drug or applying the number of patient requirements same as those used for special calculation. However, it is also true that the government needs to worry about its limited finances. The government is even considering introducing a system to further control the listed drugs due to the increased number of drugs covered by the system. This means that the price of some drugs may be reduced even further from the current price. If the government wishes to reduce the risk, they also need to tend to the blind spots. As these are areas where there are few patients and no available medicines. It is important to hold a closer year to the small but desperate voices.
- Policy
- Drug expenditures surge due to the use of high-priced drugs
- by Lee, Tak-Sun Jan 17, 2025 05:53am
- Drug expenditures have risen significantly in 2023 due to the rising cost of high-priced anticancer drugs and rare disease treatments. Due to the aging population increasing the expenditures spent on treating chronic diseases, an urgent need has arisen to come up with a measure to reduce drug expenditures. According to the National Health Insurance Service (NHIS, President: Ki-suk Jung), the total drug expenditure in 2023 was KRW 26.196 trillion, an 8.5% increase from the previous year (KRW 24.1542 trillion). This is twice as high as the 4.7% year-on-year increase in total medical expenses (KRW 110.8029 trillion) in 2023. The proportion of drug expenses in medical expenses also increased by 0.8 percentage points year-on-year to 23.6%, which indicates that the increase in drug expenditures has exceeded the critical level. As of 2022, the proportion of drug expenditures in Korea's current health expenditures was 18.0%, 3.8 percentage points higher than the OECD average of 14.2%, and ranked 7th among OECD countries. One factor contributing to the rapid increase in drug expenditures is the recent increase in reimbursement expenditures for high-priced anticancer drugs and rare disease treatments. Under the Comprehensive National Health Insurance Plan, the government has been expanding health insurance reimbursement coverage for cancer and rare diseases with high drug costs and promoting drug reimbursement for essential drugs needed for treatment in comprehensive consideration of the social and clinical needs, cost-effectiveness, public acceptance, and financial condition. In 2022, 22 drugs, including the acute lymphocytic leukemia drug Kymriah, were covered, and the scope of use was expanded for 7 drugs, including immuno-oncology drugs such as Keytruda. In 2023, 24 drugs, including the spinal muscular atrophy drug Evrysdi, were covered, and the scope of use was expanded for 8 drugs, including those for severe atopic dermatitis. As a result, as of 2023, the cost of reimbursed drugs used to treat cancer and rare and difficult diseases increased by 10.8% and 9.7% year-on-year to KRW 3.8402 trillion and KRW 2.5492 trillion, respectively, outpacing the growth rate of overall drug expenditures (8.5%). By age group, patients in their 60s accounted for the highest proportion (25.2%) of drug expenses at KRW 6.6 trillion, followed by those in their 70s (KRW 5.2 trillion), 50s (KRW 4.4 trillion), then 80s (KRW 3.1 trillion). Those aged 60 and over accounted for 58.1% of all drug expenses. By type of medical institution, pharmacies accounted for the highest amount of KRW 18 trillion (68.9%), followed by tertiary hospitals (KRW 3.8 trillion), general hospitals (KRW 2.2 trillion), and general clinics (KRW 1.1 trillion). By efficacy group, arteriosclerosis drugs (hyperlipidemia drugs) accounted for the largest expenditure of KRW 2.849 trillion, followed by anticancer drugs (KRW 2.7336 trillion), blood pressure lowering drugs (KRW 2 trillion), peptic ulcer drugs (KRW 1.3904 trillion), then diabetes combination drugs (KRW 1.3667 trillion). Due to the aging population and westernized dietary habits, hyperlipidemia drugs have taken the top spot in recent years, followed by drugs for chronic diseases (hypertension, diabetes, and hyperlipidemia). By ingredient group, the top-ranked drug was the combination of ezetimibe + rosuvastatin for hyperlipidemia, an atherosclerosis drug, posting KRW 605.8 billion. This was followed by choline alfoscerate (brain function enhancer, KRW 563 billion) > atorvastatin (hyperlipidemia drug, KRW 558.7 billion) > clopidogrel (antithrombotic drug, KRW 417.9 billion) > rosuvastatin (hyperlipidemia drug, KRW 337.7 billion). The second-ranked drug, choline alfoscerate, has seen a 104.3% increase in spending over the past 5 years (KRW 275.6 billion in 2018 → KRW 563 billion in 2023). The drug is undergoing clinical reevaluation by the Ministry of Food and Drug Safety to prove its therapeutic effectiveness through clinical trials, and for proper prescription management of the drug, HIRA has recommended institutions refrain from prescribing it for diseases other than dementia as a screened item from 2022, but expenditures on the ingredient have not decreased. “Drug expenditures are steadily increasing due to the inclusion of new drugs such as high-priced anti-cancer drugs and gene therapies in the reimbursement list and expansion of reimbursement standards, as well as the increase in chronic diseases caused by the aging population,” said an NHIS official. ”We will continue to increase coverage so that people can use the drugs they need for medical treatment on time, but will expand the analysis of drugs that are misused or unnecessarily prescribed to prepare management measures to protect health insurance finances while promoting public health.”
- Company
- Expanded reimb for 'Zejula'
- by Son, Hyung Min Jan 17, 2025 05:53am
- Dr. Jae-Weon Kim of Seoul National University Hospital Following the reimbursement expansion of Zejula for ovarian cancer, patient access to treatment has been improved. Experts suggest that Zejula will be more widely used in clinical practices based on demonstrated benefits in efficacy and safety in long-term treatment studies. On January 16, Takeda Pharmaceutical Korea held a press conference commemorating the reimbursement expansion of Zejula monotherapy as a first-line maintenance therapy of HRd-positive ovarian cancer. As of October 2024, the national health insurance reimbursement criteria for Zejula have been expanded to include treatment of homologous recombination deficiency (HRD)-positive ovarian cancer. Previously, Zejula has been covered by reimbursement only as a maintenance therapy for the first-line treatment of patients with ovarian cancer associated with BRCA who respond to platinum-based therapy. Due to the reimbursement expansion, Zejula became the only PARP (Poly ADP-ribose Polymerase)-inhibitor covered by reimbursement for the first-line maintenance therapy used in patients with HRd-positive ovarian cancer. HRd refers to homologous recombination deficiency, a DNA damage repair mechanism. When HRd is positive, cancer cells cannot efficiently repair DNA damage. It is particularly associated with BRCA1/2 gene mutations frequently observed in breast and ovarian cancers. Experts suggest that the clinical prevalence of HRd expression in ovarian cancer is over 50%. Based on a long-term follow-up study of 6.2 years, Zejula demonstrated significant benefits for patients with HRd-positive ovarian cancer. In the clinical study, the HRd patient group treated with Zejula recorded a progression-free survival of 24.5 months, which was markedly different from the 11.2 months of the placebo group. At the five-year mark of treatment, the number of patients who survived without disease progression in the Zejula group was twice as high as that in the placebo group. In the HRd patient group, a statistically significant difference in progression-free survival was observed between those treated with Zejula and those given a placebo up to five years of treatment. "Zejula has demonstrated long-term PFS benefits through the PRIMA trial, its approval study, and follow-up studies. In terms of safety, adverse events were consistent with previous clinical results, confirming its safety even with long-term use," Dr. Jae-Weon Kim of Seoul National University Hospital's Department of Obstetrics and Gynecology stated. In clinical practices, prescriptions for Zejula have been increasing since its reimbursement expansion. Ovarian cancer patients take two 100 mg tablets once daily, while Zejula is the only ovarian cancer treatment available with a once-daily dosing regimen. "Since the reimbursement expansion notice, no severe adverse events have been reported among HRd-positive patients continuing first-line maintenance therapy with Zejula monotherapy. Zejula, as an oral medication administered once daily, significantly enhances patient convenience," Dr. Jung-Yun Lee of Severance Hospital's Department of Obstetrics and Gynecology emphasized. Dr. Lee added, "HRd is a biomarker commonly observed during first-line maintenance therapy. With the reimbursement expansion of Zejula, there has been an increase in cases involving HRd testing, leading to higher diagnosis rates. Through diagnostic testing, many patients are expected to benefit from Zejula." Dr. Jung-Yun Lee of Severance Hospital
- Policy
- Pulmonary fibrosis drug 'Ofev' under consideration for reimb
- by Lee, Tak-Sun Jan 17, 2025 05:53am
- As 'Ofev Soft cap (nintedanib),' a treatment for pulmonary fibrosis, has been approved for reimbursement appropriateness, latecomer drugs are projected to enter the market quickly. Following the expiration of Ofev's usage patent, latecomer companies are anticipated to prepare for launching their drugs. According to industry sources on January 16, Daewoong's "Ofevvia tab 150 mg,' containing the same active ingredient as Ofev Soft Cap, was approved by the Ministry of Food and Drug Safety (MFDS). Last month, Yungjin Pharm's 'Nintebro Tab 150mg,' containing the same active ingredient, was approved. These companies aim to join the market early by changing the capsule formation of the original drug to tablets. They have successfully avoided the usage patent not registered as a patent in the MFDS. Therefore, latecomer drugs are projected to launch following the patent expiration of the substance patent, which is set to expire on January 25. However, latecomer companies are concerned that 'Ofev' has not been introduced the reimbursement list after it was approved in South Korea in October 2016. If Ofeb becomes introduced to the list, latecomer drugs that are considered products of development can be introduced to the list in three months after applying for reimbursement through the estimated course. Fortunately, Ofev received the green light from the Drug Reimbursement Evaluation Committee (DREC) of the Health Insurance Review and Assessment Service (HIRA). Both the original company and the generic company desire this outcome. The DREC committee determined Ofev's reimbursement appropriateness for the following indications: ▲ Systemic sclerosis-associated interstitial lung disease (SSc-ILD) ▲Treatment of chronic fibrosing interstitial lung diseases with a progressive phenotype. When the pharmaceutical company accepts the result, Ofev will be considered for the reimbursement list after undergoing drug price negotiations with the National Health Insurance Service (NHIS). Yet, the DREC excluded Ofev's first indication, idiopathic pulmonary fibrosis (IPF), for the reimbursement appropriates decision. IPF is a condition characterized by persistent and irreversible deterioration of lung function, leading to progressive scarring of the lungs and symptoms such as shortness of breath. As the disease progresses, lung function gradually and irreversibly declines. 18-32% of patients with IPF are known to progress to 'advanced pulmonary fibrosis,' where the lungs become stiffened, and fibrosis continues. The industry draws attention to whether Ofev's Boehringer Ingelheim will accept the reimbursement appropriateness decision for the rest of the indications, excluding the first indication for IPF. Even if Ofev gets introduced to the reimbursement list without the first indication, it will not pose a problem for latecomer companies. This is because the drug's use for IPF is patented, and IPF usage is not included in the currently approved efficacy and effectiveness. The efficacy and effectiveness of drugs from Yungjin Pharm and Daewoong are ▲Delay of pulmonary function decline in patients with SSc-ILD ▲Treatment of chronic fibrosing interstitial lung diseases with a progressive phenotype, excluding IPF. Analysis suggests that these companies could have avoided the usage patent if IPF is excluded from the efficacy·effectiveness section. Consequently, latecomer companies will likely determine the date to apply for reimbursement following Boehringer Ingelheim's next step. If Boehringer Ingelheim undergoes a negotiation process, companies are projected to aim for reimbursement listing as estimated drugs once the origunal drug is introduced to the list. Analysis indicates that companies must apply for reimbursement after negotiations if the original drug's reimbursement listing fails. Despite the non-reimbursement status in the domestic market, Ofev's sales have steadily increased. Based on IQVIA, Ofev generated KRW 1.8 billion in 2020, KRW 3.3 billion in 2021, KRW 5.6 billion in 2022, and KRW 5.7 billion in 2023. Ofev is projected to generate even higher sales when covered by national health insurance reimbursement.
- Policy
- Innovative Pharmaceutical Company certification to change
- by Lee, Jeong-Hwan Jan 17, 2025 05:53am
- The Ministry of Health and Welfare will improve the certification system for innovative pharmaceutical companies to a 'certification evaluation score system' that will increase the proportion of new drug research and development (R&D) and change the disqualification criteria to a point-based score system. The MOHW plans to issue an administrative notice next month after deliberations by the Pharmaceutical Industry Fostering and Support Committee. It will be interesting to see if the final notice will reflect the pharmaceutical industry's opinion that it is too harsh or unreasonable to cancel the certification of innovative pharmaceutical companies that encourage the creation of homegrown drugs and the development of the pharmaceutical industry based on illegal rebates in the past, etc. An MOHW official who requested anonymity made such remarks on the 15th, during a meeting with reporters from the Korea Special Press Association on Friday. While announcing its major work plan for 2025, the MOHW announced it would improve the core regulations for each industry to make the global leap. The improvement measures include overhauling the certification criteria for innovative pharmaceutical companies. The key is to add criteria for granting extra points for items such as a pharmaceutical company's R&D investment efforts and to allocate points for the disqualifying criteria, such as revoking the certification of innovative pharmaceutical companies based on the number of administrative penalties or the amount of illegal rebates. The industry has claimed that despite investing in R&D to create new drugs and its ethical management efforts, the revocation or decertification as an innovative pharmaceutical company due to rebate cases detected in the past dampens the companies’ will to develop new drugs. The current innovative pharmaceutical company certification system excludes innovative pharmaceutical companies based on the illegal rebate disqualification clause if they have received two or more administrative penalties for violating the Pharmaceutical Affairs Act in the past three years, or if the total amount of rebates exceeds KRW 5 million. A MOHW official said, “Under the current system if a company violates the standards, its innovative pharmaceutical company certification is revoked. We plan to introduce quantitative indicators to the disqualification criteria and add a premium for R&D efforts to allow companies to make up for the lost score” The MOHW is also considering the typification of the certification criteria for multinational pharmaceutical companies. The Korean Research-based Pharmaceutical Industry Association (KRPIA) has previously argued that open innovation collaboration with domestic research centers and companies, in addition to conventional clinical research, plays an important role in building global-level R&D capabilities. Therefore, the KRPIA has been requesting standards that can recognize and promote such activities. The MOHW has been preparing measures to reflect this. One of the improvements under consideration is to specify the reasons for rejection when notifying pharmaceutical companies of their innovative pharmaceutical company certification results. On the 10th, the 2nd Vice Minister of MOHW, Minsoo Park, said in a Q&A session on the 2025 Major Work Plan, “The most important criterion for selecting innovative pharmacuetical companies will be the R&D criteria, but we are looking at the specific condition and status of the current industry and trying to adjust the criteria accordingly.” He added, “We are considering expanding various quantitative evaluation criteria, creating customized criteria for each type of pharmaceutical company, and rationally improving the disqualification criteria,” he said. Such improvements must be reviewed by the Pharmaceutical Industry Fostering and Support Committee under the ‘Special Act on Fostering and Support of the Pharmaceutical Industry.’ The MOHW is currently preparing committee deliberations and plans to issue an administrative notice in February. Meanwhile, the benefits of being certified as an innovative pharmaceutical company include: (1) points for participating in government R&D; (2) corporate tax deduction for R&D expenses and tax deduction for investment in facilities to improve drug quality control; (3) When building research facilities, they are exempted from local regulations and levies on location; (4) companies that enter the KOSDAQ under the technology special exception or growth special exception are exempted from KOSDAQ listing requirements; and (5) drug prices are preferentially reduced for generic drugs, incrementally modified new drugs, and biosimilars, and reduce the actual transaction price reduction rate.
- Company
- Ultomiris is reimbursed for aHUS in Korea
- by Son, Hyung Min Jan 16, 2025 06:14am
- Dr. Jinseok Kim, professor of hematology at Severance Hospital. Ultomiris has been reimbursed in Korea for atypical hemolytic uremic syndrome (aHUS) since January. While experts have welcomed the news of its reimbursement, they have also raised the need for systematic improvements to the stringent conditions for reimbursement, including the preliminary review requirement. On the 10th, AstraZeneca Korea held a press conference at JW Marriott Dongdaemun Square Seoul to celebrate the reimbursement coverage of Ultomiris for aHUS in Korea. Starting this month, Ultomiris will be reimbursed by health insurance for patients with aHUS with thrombotic microangiopathy (TMA) and kidney damage. This is expected to improve access to treatment for patients with aHUS whose symptoms can worsen rapidly and lead to end-stage renal disease (ESRD). Ultomiris is a next-generation C5 complement inhibitor with a half-life approximately four times longer than Soliris. While Soliris requires dosing every 2 weeks, Ultomiris has an extended dosing interval of 8 weeks, improving treatment convenience. When complement C5 is activated on the surface of bacteria, it produces a membrane-attacking complex that causes holes in the cell membrane. If the normal immune system process of complement activation continues, vascular endothelial cells are disrupted, causing related diseases. Ultomiris has a mechanism of action that inhibits this process. aHUS is an acute rare disease in which the immune system's complement is overactivated due to a genetic defect and causes thrombotic microangiopathy, which can lead to severe damage to multiple organs, especially the kidneys. aHUS refers to hemolytic uremic syndrome not associated with E. coli. The efficacy and safety of Ultomiris were confirmed in Phase III Study 311 in adult patients who were not previously treated with complement inhibitors. At 26 weeks of treatment, Ultomiris demonstrated improvement in TMA-related markers, including platelet and LDH levels, in 53.6% of patients. The treatment also demonstrated sustained terminal complement inhibition by maintaining serum-free C5 concentrations of < 0.5 μg/ml. In the Phase III Study 312 in pediatric patients, Ultomiris demonstrated complete resolution of TMA in 94.4% of patients at 50 weeks of treatment. “In a study of pediatric aHUS patients who switched from Soliris to Ultomiris, the renal and hematologic parameters remained stable over one year, confirming the efficacy of the switch,” said Dr. Jinseok Kim, professor of hematology at Severance Hospital. “As clinicians, we are excited to be able to offer hope to our patients with the introduction of this new treatment option.” Ultomiris and Soliris are currently available for aHUS in Korea, but both agents are only available to patients who have been approved through the prior review process. Clinicians have called for improved reimbursement, including moving to a post-review process, as patients' conditions can worsen if they are not properly dosed. The coverage of aHUS was implemented in 2018 with the introduction of Soliris. However, according to an analysis of the results of the preliminary review system from July 2018 to October last year, 56 out of 321 cases were approved, showing an average approval rate of 18%. Kim added, “Although Ultomiris has been reimbursed, like Soliris, it is subject to the same restrictions as Soliris, which requires preliminary review. We hope that systemic improvements are also made so that aHUS patients can receive treatment in a timely manner.”
- Company
- Last year's pharma export sales recorded KRW 11T
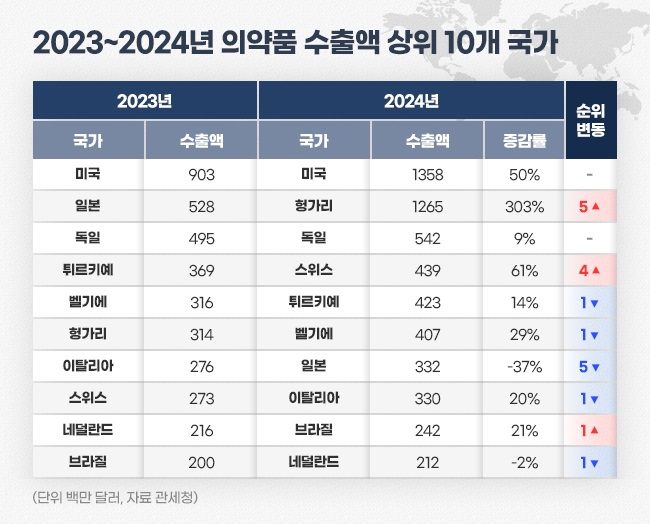
- by Kim, Jin-Gu Jan 16, 2025 06:14am
- Last year's export sales of Korea-made pharmaceuticals amounted to US$ 7.5 billion (approximately KRW 11 trillion), up 29% from the previous year. Analysis indicates that a significant increase in biopharmaceutical exports of Samsung Biologics and Celltrion has contributed to the growth. The largest exported country was the United States, with exports totaling US$ 1.3 billion (approximately KRW 1.98 trillion), up 50% from the previous year. Export sales to Hungary increased more than fourfold over the year, reaching US$ 1.2 billion (approximately KRW 1.85 trillion), making it the second largest after the United States. Pharmaceutical export sales reached US$ 7.54 billion last year…second largest in the history According to the Korea Customs Service on January 16, last year's Korean pharmaceutical export sales amounted to US$ 7.53 billion. After the announcement of the endemic, export sales expanded to the largest size. The export sales of domestically produced pharmaceuticals significantly increased during the COVID-19 pandemic. In 2019, the export value was US$ 3.69 billion, which soared by 81% over the year to reach US$ 6.68 billion in 2020 as the pandemic intensified. By 2021, this figure had further increased by 22%, reaching US$ 8.12 billion, setting an all-time high, primarily driven by the export sales of domestically developed COVID-19 vaccines. After that, the sales decreased for two consecutive years until 2023. In 2022, it decreased to US$ 6.27 billion, down 23% compared to the previous year. In 2023, it further shrank to US$ 5.85 billion. Last year, export sales rebounded successfully. Compared to 2023, export sales rose by 29%. It is the second largest after 2022. Yearly pharmaceutical export sales (unit: US$ 1 million, source: Korea Customs Service) The pharmaceutical industry considers that the export growth resulted from a significant increase in biopharmaceutical exports to the United States and Europe, primarily driven by Samsung Biologics and Celltrion. As of Q3 last year, Samsung Biologics' cumulative export sales reached KRW 3.29 trillion, up 26% compared to KRW 2.62 trillion in the Year-over-Year (YoY). This represents an annual export growth of over KRW 600 billion. Considering its order backlog, Samsung Biologics is projected to achieve record-breaking yearly export performance. Since 2015, Samsung Biologics has secured US$ 14.23 billion in orders as of Q3 last year. Among this, US$ 7.49 billion of products have been delivered, leaving an order backlog of US$ 6.73 billion. If the clients successfully develop their products, the estimated value of the order backlog could rise to US$ 12.31 billion. Celltrion's cumulative export sales for Q3 also increased by 30%, from KRW 623.5 billion in 2023 to KRW 810 billion in 2024. Additionally, SK Biopharm, GC Biopharma, Yuhan, Hugel, Dongwha, Daewon, and Boryung also had significant increases in export sales, contributing to the overall growth of pharmaceutical exports. Pharmaceutical import sales reached US$ 8.98 billion, marking a 5% decrease compared to the previous year. The pharmaceutical trade deficit narrowed with a significant increase in exports and a decline in imports. The trade deficit improved from US$ 3.58 billion in 2023 to US$ 1.45 billion last year. Exports to the US represent largest amount over three consecutive years…Hungary, a four-fold increase over the year Korea has exported the most to the United States the most. Last year's pharmaceutical exports to the United States reached US$ 1.35 billion. The United States accounts for 18% of the total pharmaceutical export sales. Export sales to the United States quickly rose over the past two years. It increased 7% from KRW 843.94 million in 2022 to KRW 933.0 million in 2023. Last year, it increased over 50%. In 2022, the United States became the largest export destination, replacing Germany. Since then, it has maintained the top rank for three consecutive years. Hungary ranked second in export sales after the United States. Last year's export sales to Hungary reached US$ 1.26 billion, surging more than four-fold compared to US$ 316.27 million in 2023. The top countries by pharmaceutical export sales 2023-2024 (unit: US$ 1 million, source: Korea Customs Service) One of the reasons for the significant increase in exports to Hungary is Celltrion's expanded biosimilar export to Europe. Hungary is Celltrion's export hub for the European market. The company announced its plans to establish a direct sales system using its Hungarian subsidiary as the gateway for European exports. As part of this strategy, shipments of Celltrion's biosimilars for the European market have been concentrated through its Hungarian subsidiary. Celltrion's biosimilars, primarily Remsima SC, performed strongly in Europe last year. The company's cumulative European export sales for Q3 of last year reached KRW 444.9 billion, up 57% compared to KRW 283.5 billion during the same period the previous year. This growth is attributed to the expanded export coverage of Remsima SC to additional countries. Followed in ranking were Germany, Switzerland, Türkiye, Belgium, Japan, Italy, Brazil, and the Netherlands. Export sales increased YoY for all countries except Japan and the Netherlands. Exports to Germany amounted to US$ 542.14 million, up 9% from US$ 495.40 million in 2023. Exports to Switzerland recorded US$ 438.78 million, up by 61%. Exports to Türkiye rose 14% to US$ 422.80 million, while Belgium saw a 29% increase, recording US$ 406.59 million. Exports to Japan declined by 37% from US$ 528.47 million in 2023 to US$ 331.77 million. In 2023, exports to Japan ranked second after the United States. However, it went down to No.7 for last year.