- LOGIN
- MemberShip
- 2026-05-15 21:37:10
- Curocell begins commercialization process for Korea's first CAR-T
- by Cha, Ji-Hyun | translator Hong, Ji Yeon | 2026-05-15 14:44:27
Curocell is pursuing market entry following the approval of South Korea's first domestically developed chimeric antigen receptor T-cell (CAR-T) therapy. Based on its domestic R&D and manufacturing infrastructure, the company aims to improve patient access to the CAR-T treatment landscape that has previously relied mostly on overseas facilities. Furthermore, the company plans to strengthen its next-generation pipeline through a strategy of expanding indications.
On May 14, Curocell held a press conference at the Four Seasons Hotel in Seoul to commemorate the approval of its CAR-T therapy, 'Rimqarto Inj' (ingredient name: anbalcabtagene autoleucel). The company unveiled Rimqarto's clinical value, domestic commercialization strategy, and directions for subsequent clinical development. The event was attended by Curocell CEO Gunsoo Kim, Professor Won Seog Kim of Hemato-oncology at Samsung Medical Center, Curocell Executive Director Seungwon Lee, and Su-hee Cho, Head of Curocell’s Clinical Development Center.
Rimqarto is an autologous CD19-targeting CAR-T therapy that uses a patient's own T cells. The process involves collecting T-cells from the patient's blood, introducing genes that allow them to recognize cancer cells, proliferating them ex vivo, and reintroducing them into the patient. It has been designed with Curocell's proprietary OVIS platform, designed to maximize therapeutic effects by simultaneously suppressing the expression of PD-1 and TIGIT, immune checkpoint receptors that inhibit the anticancer activity of T cells.
Previously, on April 29, the Ministry of Food and Drug Safety (MFDS) granted marketing authorization for Rimqarto as a treatment for adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and primary mediastinal large B-cell lymphoma (PMBCL) after two or more lines of systemic therapy. With this, Rimqarto has been listed as the 42nd novel drug developed in Korea and the first CAR-T therapy developed by a Korean company.

CEO Kim shared in his opening remark that "The approval of Rimqarto is more than just one new drug entering the market. Curocell has step-by-step built a foundation that allows the entire process(from R&D to clinical trials, production, quality control, and licensing) to be performed domestically."
CEO Kim emphasized that Rimqarto is a treatment option that can expand patient access in the CAR-T treatment landscape, which has been highly dependent on overseas supply. "For patients with relapsed or refractory DLBCL, treatment options are limited and disease progression can be rapid, making timely treatment critical," and added, "While existing CAR-T therapies faced barriers before reaching Korean patients in clinical settings, we will do our best to improve treatment accessibility and establish a stable supply base through Rimqarto."
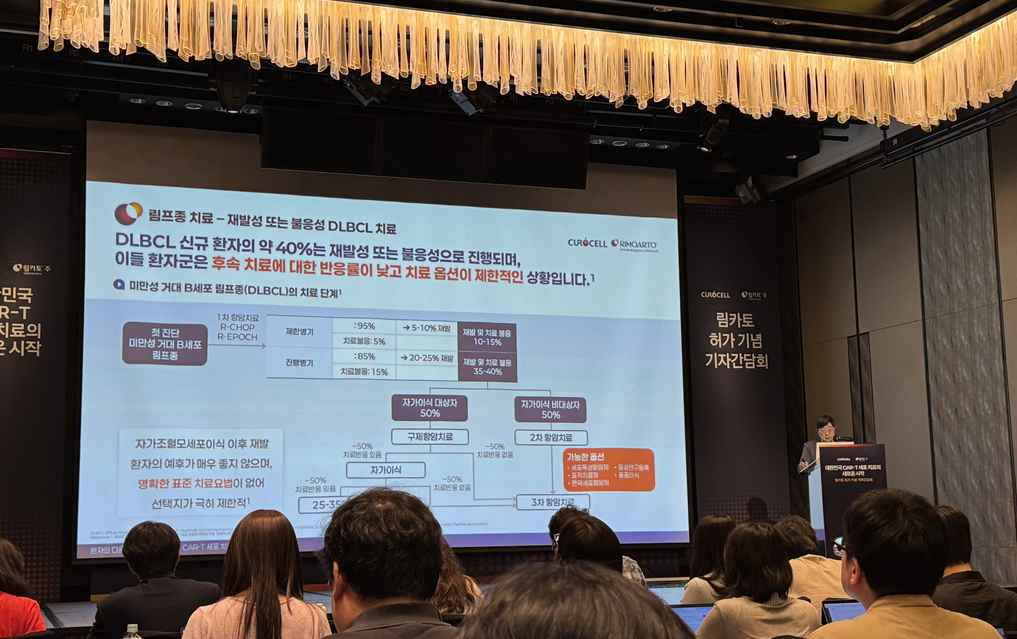
Professor Won Seog Kim, the first presenter, explained the unmet needs in DLBCL treatment and the clinical value of Rimqarto. According to Professor Kim, approximately 6,000-6,500 cases of malignant lymphoma occur annually in Korea, with DLBCL being the representative disease, accounting for 40% of cases. However, 35–40% of DLBCL patients experience recurrence after standard treatment, and patients reaching the third-line treatment stage number around 700 annually in Korea, carrying a poor prognosis.
CAR-T therapies are considered an option that has changed the treatment paradigm in this relapsed/refractory DLBCL field. Professor Kim said, "In a patient group where the possibility of long-term survival was previously around 10%, data showed that CAR-T treatment could offer another treatment opportunity. For this reason, we could proceed quickly to obtain reimbursement, and added, "However, even with current commercial CAR-T therapies, the expectation for a complete cure remains at about 40%, leaving room for further improvement."
Professor Kim presented Curocell's proprietary OVIS technology as Rimqarto’s distinguishment. One reason existing CAR-T therapies fail is that T-cells become exhausted or immune checkpoint mechanisms such as PD-1 or TIGIT become active, preventing the sufficient elimination of cancer cells.
"Curocell’s OVIS technology works by loading small RNA fragments into the CAR-T to suppress the expression of immune checkpoints like PD-1 and TIGIT," Professor Kim explained, "Rimqarto is a differentiated, next-generation CAR-T product in that it can overcome treatment failure caused byover-expression of immune checkpoints."
Clinical data also confirms Rimqarto’s competitiveness. In Phase 2 trial, Rimqarto demonstrated an objective response rate (ORR) of 75% and a complete response (CR) rate of 67% based on independent review committee evaluations. Regarding safety, the incidence of Grade 3 or higher cytokine release syndrome (CRS) was 9%, and neurotoxicity was 4%. Professor Kim said, "The response rate of over 75% and a CR rate of 67% as evaluated by the committee are highly encouraging results. It showed data that is manageable not only in terms of efficacy but also safety."
Starting with this approval, Curocell plans to accelerate the commercialization of Rimqarto. Related to the core pillars of the commercialization strategy, Executive Director Lee Seung-won presented ▲rapid insurance reimbursement listing ▲expanding patient accessibility. "As CAR-T therapies are perceived as high-priced treatments, the speed of reimbursement listing is directly linked to patient access," Lee explained, " Rimqarto was selected for the parallel 'Approval-Evaluation-Negotiation' pilot project, listed on a track that can shorten the time from approval to reimbursement by approximately 90 days." Curocell is currently responding to supplementary data requests for the drug reimbursement evaluation and designing price-negotiation scenarios to support a reimbursed launch as early as September.
The company will also pursue its domestic production base to build a supply chain and expand patient access. Lee stated, "Existing global CAR-T therapies involve a structure where a Korean patient’s cells are sent to an overseas manufacturing site and then brought back, which takes weeks and carries international transport risks. Rimqarto can achieve a fast 'vein-to-vein' time based on its domestic production facility." Curocell plans to establish a system within the year allowing Rimqarto treatment at 30 hospitals nationwide and will manage the entire process (from ordering to collection, manufacturing, delivery, and administration) through its online tracking platform, 'CuroLink.'

The company is also strengthening its next-generation pipeline by expanding Rimqarto's indications. As a follow-up development strategy, Curocell is considering expanding indications to adult acute lymphoblastic leukemia (ALL), systemic lupus erythematosus (SLE), and second-line treatment for DLBCL. Director Cho Su-hee explained, "There are currently no clear treatment options after standard therapy for patients over the age of 25, who make up the majority of adult ALL patients," and added, "Curocell has been preparing to expand the indication to adult ALL since 2022 and is currently in the final stages of Phase 1." The company plans to enter Phase 2 shortly and is also pursuing an expansion of clinical trials in Japan to strengthen global capabilities.
The company is also pursuing expansion into autoimmune diseases and earlier lines of treatment. Director Cho stated, "To provide a better life for patients with lupus nephritis, we have started Korea's first autoimmune disease CAR-T clinical trial. Based on the experience of administering Rimqarto to lupus nephritis patients through "approval for use for therapeutic purposes," we expect encouraging results," and added, "Since Rimqarto showed a high response rate compared to other agents in third-line DLBCL, moving it up to second-line treatment would help even more patients."
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- [Reporter's View] The dark side of improved diabetes med convenience
- Reporter's view | Son, Hyung Min
- [Reporter's View] Contradiction of "K-passing" and a new drug powerhouse
- Reporter's view | Lee, Jeong-Hwan






