- LOGIN
- MemberShip
- 2026-05-14 14:14:14
- Domestic CAR-T Limcato speeds toward reimbursement
- by Hwang, byoung woo | translator Alice Kang | 2026-05-14 09:28:26

With Korea’s first domestically developed CAR-T therapy Limcato (anbalcabtagene-autoleuce) receiving regulatory approval, attention is now focused on the timing of its reimbursement.
Curocell expects to accelerate market entry through the pilot program that allows parrallel operation of approval, reimbursement evaluation, and price negotiation. With the fast-track system in place, commercialization may be possible as early as the second half of the year.
However, some observers note that it remains to be seen whether the reimbursement listing will proceed strictly according to schedule. While Limcato holds symbolic significance as the first domestically developed CAR-T therapy, it is still an ultra-high-cost, one-time treatment costing hundreds of millions of won.
This implies that factors such as risk-sharing, performance-based post-marketing management, and reimbursement conditions, in addition to the drug price, could serve as variables in the negotiation process.
Ultimately, the key focus regarding Limcato’s reimbursement is shifting from “whether it will be reimbursed” to “under what conditions and how quickly it will be listed.”
While the likelihood of reimbursement itself is viewed relatively positively, analysis suggests that the actual speed of commercialization may vary depending on the terms of the negotiations.
Reimbursement prospects are optimistic… Speed expected to rise via parallel track
Limcato is an autologous CD19-targeted CAR-T therapy developed by Curocell. Its approved indications are for the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and primary mediastinal large B-cell lymphoma following two or more prior line of systemic therapy.
The Ministry of Food and Drug Safety (MFDS) granted marketing authorization for Limcato on the 29th of last month. This marks the first instance of a CAR-T therapy developed by a domestic company receiving approval.
Previously, the MFDS designated the drug as a “Bio-Challenger” candidate and the 33rd entry in the “Global Innovative Product on Fast-Track (GIFT)” system, providing tailored consultation and expedited review from the early stages of development.
The outlook for reimbursement is quite promising. Limcato has been selected as a drug for the Ministry of Health and Welfare’s “Pilot Project for Parallel Application for Approval, Reimbursement Evaluation, and Drug Price Negotiation.”
Previously, the process could take over 300 days, including 120 days for MFDS approval, 150 days for HIRA reimbursement evaluation, and 60 days for NHIS drug price negotiations, but the pilot project aims to shorten this timeline.
The company also maintains a positive outlook. Regarding drug price negotiations, the company has mentioned the possibility of an adjustment to a level that is the same as or slightly lower than the prices of existing CAR-T therapies, and therefore does not view the drug price itself as a factor that will cause significant delays.
In Korea, Novartis’ Kymriah has already established a precedent for CAR-T reimbursement, making this scenario more feasible.
Last January, the National Health Insurance Review and Assessment Service (HIRA) established reimbursement criteria for Gilead Sciences’ Yescarta for the indication of “treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and primary mediastinal B-cell lymphoma (PMBCL) following two or more prior systemic therapies.”
Although Limcato is a late entrant, it benefits from accumulated experience in prior CAR-T evaluations and negotiations.
However, the industry notes that reimbursement likelihood and negotiation difficulty are separate issues. Even if the likelihood of entering the reimbursement system is high, given the nature of ultra-high-cost one-time treatments, there may be significant debate surrounding the conditions for listing.
Conditions, not price, may be the key variable… focus on risk-sharing discussions
Pricing is an important starting point for Limcato. Since Kymriah is already reimbursed, the CAR-T therapy’s price is expected to serve as a benchmark.
If Limcato adopts a relatively lower pricing strategy, this could strengthen its case for reimbursement. Its domestic manufacturing base also adds positive policy value.
However, industry experts suggest that risk-sharing and post-management conditions may be more critical than price itself.
CAR-T is an ultra-high-cost therapy that is expected to produce therapeutic effects with a single administration. If a sufficient response does not occur after administration, or if relapse or death occurs within a certain period, it will be crucial to determine how to link the financial burden on the national health insurance system to treatment outcomes.
According to Market Access (MA) experts, the likelihood of Limcato being covered by insurance is considered relatively high. This is because there are precedents for the coverage of existing CAR-T therapies, and if the company proposes a price that is equal to or lower than that of existing treatments, there is a strong possibility that the Health Insurance Review and Assessment Service (HIRA) and the National Health Insurance Service (NHIS) will positively consider the necessity of listing the drug.
However, the pharmaceutical industry views that, given the high likelihood of Limcato being discussed within the category of ultra-high-cost one-shot therapies, conditions such as performance-based reimbursement, patient-specific tracking, efficacy evaluation over a set period, and submission of post-treatment management data may be attached.
This is assessed not so much as a factor that makes reimbursement itself difficult, but rather as a variable that influences the duration of negotiations and the burden of actual commercialization preparations.
In particular, Limcato is the first domestically developed CAR-T therapy to be commercialized directly by a Korean biotech company. Compared to multinational pharmaceutical companies, the organizational burden may be relatively greater in areas such as managing risk-sharing agreements, establishing collateral, managing refunds, responding to hospitals, the National Health Insurance Service (NHIS), and the Health Insurance Review and Assessment Service (HIRA), and managing long-term follow-up data.
The industry is considering two scenarios regarding this point.
The first scenario is if the company accepts the drug price and post-marketing management conditions relatively quickly. In this case, leveraging the intent of the pilot program, which combines approval, evaluation, and negotiation, it appears possible to secure reimbursement listing and achieve commercialization in the second half of the year.
If Limcato secures price competitiveness compared to existing CAR-T therapies and negotiations proceed without major disagreements regarding post-marketing management conditions, the speed of market entry could accelerate.
The second scenario is if negotiations over risk-sharing conditions drag on. Even if the drug price can be adjusted relative to existing treatments, discussions regarding reimbursement conditions, long-term follow-up criteria, and performance evaluation methods could take a long time.
In this case, rather than the reimbursement listing itself falling through, it is more likely that the timing of the listing and the actual pace of prescription expansion will be delayed.
The initial key point to watch is the timing of the submission to the Cancer Drug Deliberation Committee. Even if the drug enters the parallel approval, evaluation, and negotiation track, it must still undergo deliberation by the Cancer Drug Deliberation Committee. The industry is closely watching whether it will be submitted in May.
Some observers believe there will be no Cancer Drug Deliberation Committee meeting scheduled for June, so the outline of the reimbursement schedule for the second half of the year is expected to become clearer depending on the timing of the agenda’s submission.
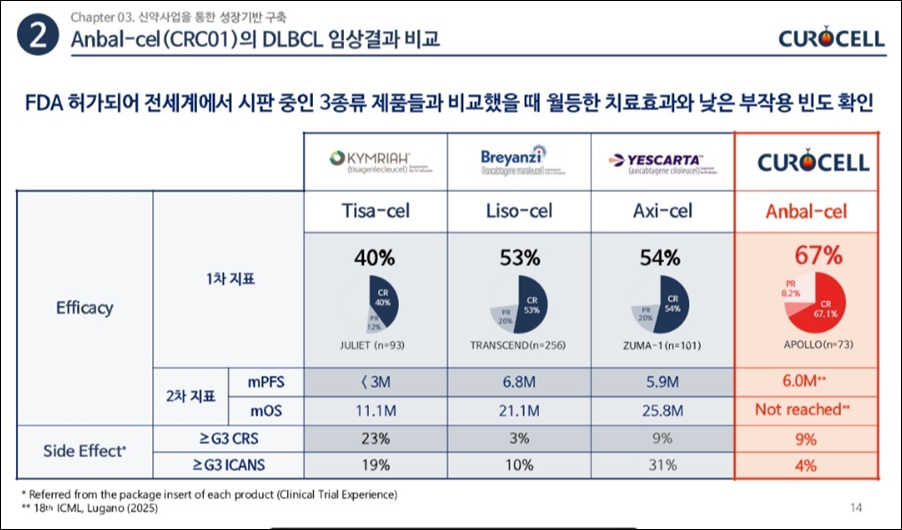
Supply speed is a strength… Differentiation through domestic manufacturing infrastructure
In the commercialization phase following reimbursement, Limcato’s supply competitiveness is expected to emerge as a key differentiator.
Existing CAR-T therapies followed a structure where patient cells were collected, sent to overseas manufacturing facilities, and the finished product was then imported back into the country.
In this process, manufacturing, logistics, and customs clearance procedures overlapped, requiring a certain amount of time before treatment could begin. Particularly for patients with rapidly progressing terminal blood cancers, the waiting period itself has been cited as a barrier to treatment accessibility.
In contrast, Curocell has established a system for domestic manufacturing and supply based on its CAR-T-dedicated GMP production facility in Daejeon. In fact, the company highlights the ability of Limcato to reduce waiting times through this domestic production and supply system as a key strength.
Curocell has also established “CuroLink,” an integrated solution for commercial operations. CuroLink is a cell therapy supply management solution that links information in real time between hospitals, manufacturing sites, and logistics providers. It is structured to track and manage patient-specific information from prescription through leukocyte collection, cell manufacturing, shipment, and administration.
Curocell also believes that its domestic production base will help expand patient access.
A Curocell official stated, “Since Limcato is manufactured at domestic GMP facilities, we can promptly respond to supply requests even from regional hospitals. Once a hospital decides to prescribe the drug after it is listed for reimbursement, the product supply process can be carried out through the system prepared by the company.”
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- [Reporter's View] The dark side of improved diabetes med convenience
- Reporter's view | Son, Hyung Min
- [Reporter's View] Contradiction of "K-passing" and a new drug powerhouse
- Reporter's view | Lee, Jeong-Hwan






