- LOGIN
- MemberShip
- 2026-05-17 08:10:41
- Company
- PCSK9i Praluent can be prescribed after 5 years of approval
- by Eo, Yun-Ho Jan 19, 2022 06:07am
- Praluent can be prescribed at general hospitals. As a result, practical competition for PCSK9 inhibitors began five years after domestic approval. According to related industries, Sanofi-Aventis Korea's Praluent (Alirocumab) passed the Drug Committee of medical institutions such as Seoul National University Bundang Hospital, Gangnam Severance Hospital, Chonnam National University Hospital, and Pusan National University Hospital, including Asan Medical Center. Praluent was approved in Korea in January 2017, but it was not until June 2021 that insurance benefits were listed. Amgen Korea's Repatha(Evolocumab) was approved in April 2017, and the willingness to register was stronger. Repatha was first listed as an indication for homogeneous family hypercholesterolemia (HOFH) in August 2018. There were two PCSK9 inhibitors, but in fact, Repatha was the only option. Since last year's registration, Sanofi has carried out the landing process at a general hospital, and the competition between the two drugs is expected to begin in earnest as they show results this year. Repatha, which has advanced into the benefit right, has already entered the medical institution code and is attracting prescriptions. It can be prescribed at major medical institutions nationwide, including Samsung Medical Center, Seoul National University Hospital, Seoul St. Mary's Hospital, Asan Medical Center, and Sinchon Severance Hospital. PCSK9 inhibitors had a wide range of areas requiring prescriptions other than HoFH, and in January 2020, Repatha succeeded in expanding the benefit criteria to patients with high risk of atherosclerotic cardiovasic disease (ASCVD), Heterozysocial Hypercholesterolemia (HeFH), and statin-insensitive patients. The competitive areas of the two drugs are actually expected to be ASCVD and HeFH. Praluent added HoFH indications in the United States in April last year. Meanwhile, Praluent tended to reduce the risk of all-cause death, and was approved in two doses of 75 mg and 150 mg, and patient-specific dose selection is possible by referring to patient status and LDL-C levels. Listed Repatha has already entered the medical institution code and is attracting prescriptions. Prescriptions are available at major medical institutions nationwide, including Samsung Medical Center, Seoul National University Hospital, Seoul St. Mary's Hospital, Asan Medical Center, and Sinchon Severance Hospital.
- Company
- 3 Korean companies accelerate Stelara biosimilar development
- by Ji Yong Jun Jan 19, 2022 06:06am
- Korean companies are rushing to get a share of Stelara’s autoimmune disease treatment market that brings in ₩9 trillion in annual sales. Samsung Bioepis, which had been late in starting the development of Stelara biosimilars than its competitors like Dong-A ST and Celltrion, completed patient recruitment for its Phase III trial and pulled forward its study completion date. All three companies are expected to complete clinical trials for the commercialization of their Stelara biosimilars within this year. According to ClinicalTrals.gov, the clinical information website operated by the US National Institutes of Health, on the 19th, Samsung Bioepis had recently completed patient enrollment for its global Phase III clinical trial for SB17 (Name of Stelara biosimilar in development). The company completed patient recruitment in only 6 months since the company started Phase III trials in July last year. This Phase III trial that is being conducted in patients with moderate-to-severe plaque psoriasis registered 503 patients, exceeding the company's initial goal of enrolling 464 patients. The expected completion date was also pulled forward from March 2023 to December this year. Such moves are interpreted as Samsung Bioepis’s effort to accelerate the development of SB17. Stelara is an IL-12 and 23 inhibitor developed by the multinational pharmaceutical company Janssen. It is used to treat autoimmune diseases such as psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis. In 2020, the drug raised $7,707,000,000 (approximately ₩9,155,900,000,000 based on exchange rates on the 18th) in global sales. However, Stelara will soon face competition with latecomers as its substance patent expires in the US in September 2023 and Europe in July 2024. In other words, the mega-market that brings in ₩9 trillion a year will soon be open for entry by biosimilars from September next year. With Samsung Bioepis pulling forward its clinical trial completion date, Celltrion and Dong-A ST are also planning to complete their clinical trials for their Stelara biosimilar within this year. Celltrion will be completing its trial in the first half of this year, and Dong-A ST in the second half of this year. Celltrion’s Stelara biosimilar, CT-P43, is being developed the fastest. Celltrion started Phase III trials for CT-P43 in January last year and completed patient enrollment with 509 patients in May of the same year. The company is in its final stages of the trial and is expecting trial completion by May this year. Celltrion plans to commercialize CT-P43 in line with Stellara's substance patent expiry in the US and Europe. If the company keeps up its pace of development, the company is expected to be able to easily enter the global core market. Dong-A ST is also speeding up its clinical trial for DMB-3115, its Stelara biosimilar in development. Dong-A ST started global Phase III trials for DMB-3115 in April last year. In November of the same year, the company completed enrolling 605 patients for the trial. Dong-A ST plans to complete the Phase III trial for DMB-3115 in November this year. Dong-A Socio Holdings and Meiji Seika Pharma had been jointly developing DMB-3115 since 2013. In July 2020, the right to develop and commercialize DMB-3115 was transferred from Dong-A Socio Holdings to Dong-A ST. Currently, the drug is being jointly developed by Dong-A ST and Meiji Seika Pharma. The two companies have also completed preparations for the global commercialization of the drug. In July last year, Dong-A ST and Meiji Seika Pharma signed a global license-out agreement for DMB-3115 with Intas Pharmaceuticals, transferring the rights for approval and marketing of DMB-3115 in the global region, excluding Korea, Japan, and some Asian countries. An industry official said, “Biosimilars earn a profit by taking a piece of the original’s market share of the pie. Therefore, earlier entry is beneficial to accumulate prescription data, etc.”
- Policy
- Bill for Rotarix·RotaTeq to be covered by NIP
- by Lee, Jeong-Hwan Jan 19, 2022 06:06am
- A bill to promote the health of infants and young children while reducing consumer burden by including rotavirus vaccines in the National Immunization Program (NIP) is being promoted. If passed, the bill will turn GSK’s Rotarix and MSD’s RotaTeq that are already being used in Korea into free national vaccinations. Since the WHO recommends mandatory vaccination of the rotavirus vaccine and the nature of the virus that is highly infectious among infants and young children, attention is focused on the legislative direction of the bill. On the 18th, NA member Bae Hyun-jin of the People Power Party announced that she had submitted a bill as representative for the partial amendment of the ‘Infectious Disease Control And Prevention Act’ to incorporate such changes. The incidence of rotavirus infection is very high among children aged under 5 to the extent that most of these infants are infected at least once with the virus. NA member Bae Hyun-jin expressed concerns that some parents give up rotavirus vaccinations due to the high cost even though rotavirus vaccinations are recommended for all newborns after 6 weeks of age. According to the KDCA’s Immunization Registry System, 7.9%, 21,728 of the 274,221 newborns eligible for vaccinations did not receive rotavirus vaccines in 2020. Statistics over the past 5 years show that 14.8% of the infants and young children eligible for vaccinations – 222,565 – did not receive rotavirus vaccines. Rotavirus vaccines are relatively high-priced, costing around ₩200,000-₩300,000. The two rotavirus vaccines currently sold in the market are GSK’s Rotarix and MSD’s RotaTeq. Rotarix is given in 2 doses, RotaTeq in 3. The two vaccines cost ₩70,000-₩100,000 and ₩100,000-₩130,000 per shot, respectively. In total, Rotarix costs around ₩200,000-₩260,000 and RotaTeq ₩210,000-₩300,000 to get fully vaccinated. The issue regarding the price of rotavirus vaccines has risen several times intermittently. GSK and MSD had raised the price of Rotarix and RotaTeq by 12% and 17% respectively, increasing the burden borne by consumers. Rotavirus vaccines are considered essential for newborns, and therefore its consumers are highly sensitive to their change in price. As a result, some local governments have implemented policies to provide free rotavirus vaccines for infants. Bae believes that the government should pay for the cost of the rotavirus vaccine inoculations to prevent enteritis in infants and young children, contribute to promoting public health, and reduce the burden borne by the consumers. Therefore, the bill contains a plan to include rotavirus infection as a disease eligible for regular vaccinations. Bae said, “The reality is that the need for rotavirus vaccination has been discussed for a long time but was excluded from support due to its high cost. However, no infants nor young children should be left out, unable to receive essential vaccinations due to financial reasons.”
- Company
- Exporting medicines to North America ↑ 2.6 times in 2 years
- by Kim, Jin-Gu Jan 19, 2022 06:06am
- Exports of domestic drugs hit an all-time high last year, especially exports to the North American market surged, the report showed. Exports to the U.S. and Canada have increased 2.6 times in two years. 2017-2021 Amount of medicines exported to the North American market Germany is still the largest exporter, but it is estimated to have decreased slightly compared to last year. In addition, exports of medicines to Japan, Italy, and Vietnam have increased significantly. ◆Last year's US exports amounted to $1.1 billion, 71% compared to $600 million in 2020 ◆ According to the Korea Customs Service on the 16th, pharmaceutical exports amounted to $8.12144 billion last year, up 18% from $6.8935 billion in 2020. Exports to the North American market have increased significantly. U.S. exports amounted to only $643.04 million in 2020. Last year, it rose 71 percent to $1.097.85 billion. Canadian exports increased 130% from $32.99 million to $75.99 million during the same period. The total exports to the North American market increased 1.7 times from $676.03 million in 2020 to $1.173.83 billion last year. Compared to $44329 million two years ago, it has increased 2.6 times. ◆ Biosimilar, K-new drug and botulinum toxin are in full swing Celltrion and Samsung Biologics account for a large portion of exports to the North American market. Celltrion exports Remicade biosimilar "Remsima," Mabthera biosimilar "Truxima", and Herceptin biosimilar "Herzuma" to the North American market through Celltrion Healthcare. Celltrion Healthcare's cumulative exports to the North American market in the third quarter of last year amounted to 491.4 billion won. Remsima occupies the highest market share among Infliximab biosimilars in the US market. Samsung Biologics entrusts and produces antibody drugs from global pharmaceutical companies. Last year's exports amounted to 377.6 billion won until the third quarter. The annual exports to North America in 2020 exceeded 289.5 billion won early. On top of that, domestic new drugs and domestic botulinum toxin drugs have been adding strength since last year. Sales of SK Biopharm's new epilepsy drug Xcopri (Cenobamate) in the North American market have begun in earnest. SK Biopharm's cumulative exports in the third quarter of last year amounted to 187.9 billion won, up 19 times from the cumulative 9.9 billion won in the third quarter of 2020. Almost all are estimated to be Xcopri sales. SK Biopharm received Xcopri marketing approval from the U.S. FDA in November 2019. It went on sale in May 2020. Since then, sales in the North American market began in earnest last year, and SK Biopharm's exports have also soared. The full-fledged sale of domestic botulinum toxin in the U.S. market after the "Strain Dispute" agreement is also cited as the background of the increase in exports to the region. Last year, domestic botulinum toxin exports amounted to $235.85 million, up 15% from $2528 million in 2020. In particular, exports to the United States increased significantly. Domestic botulinum toxin exports to the U.S. stood at $15.8 million in 2020, but more than doubled to $31.3 million last year. Early last year, Daewoong Pharmaceutical and Medy Tox concluded the dispute over botulinum toxin strains through an agreement. From the second quarter, sales of Daewoong Pharmaceutical's "Nabota (Jeubeau)" in the North American market began in earnest. Sales of Xcopri (left) in North America have begun in earnest North American market. ◆Germany, the largest exporter, fell 5% year-on-year, while exports to Japan and Vietnam surged Although exports to the U.S. have soared, Germany is still the largest exporter of domestic medicines. Last year, exports of medicines to Germany amounted to $1.76922 billion. Compared to 2020, it decreased by 5%. Germany's share of pharmaceutical exports fell from 26.9% to 21.8% during the same period. Exports to Japan, Vietnam, and Italy surged. Last year, exports of medicines to Japan amounted to $48699 million. It increased 42% from $265.17 million in 2020. Exports to Vietnam increased 70% from $144 million to $253.11 million , while exports to Italy rose 187% from $65.28 million to $187.2 million.
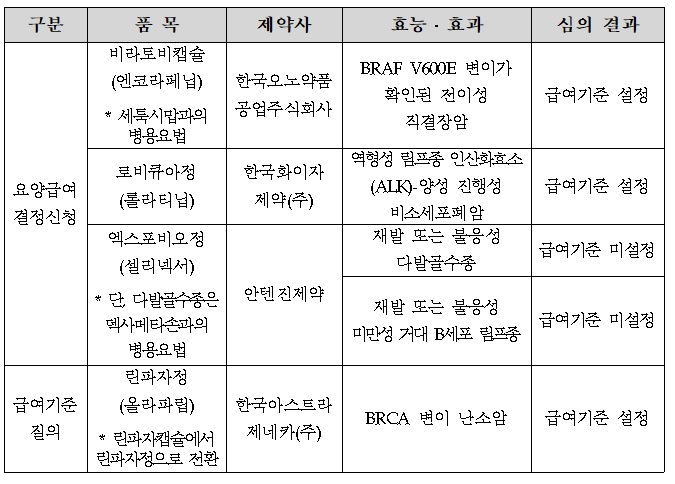
- Policy
- The benefit standard for Braftovi & Lorviqua was set
- by Lee, Hye-Kyung Jan 19, 2022 06:06am
- At the first Severe (Cancer) Disease Review Committee held this year, the benefit standards for Braftovi, a direct bowel cancer treatment of Ono Pharmaceutical Korea, and Pfizer's Lorviqua, were set. The HIRA (Director Kim Sun-min) today (12th) released the "Result of Review on benefit Standards for Drugs for Cancer Patients" reviewed by the 1st Cancer Disease Review Committee in 2022. Today's cancer screening was newly participated by members of the 9th committee formed in November last year, and Braftovi and Lorviqua, who applied for medical care benefit decisions, set benefit standards, and Antigene's Xpovio did not set standards. Braftovi, which standard was set, was approved by the MFDS on August 19 last year for the use of combination therapy with Erbitux in adult patients with direct bowel cancer who had previous treatment experience and confirmed BRAF V600E mutation. Since Lorviqua was designated as a rare drug in March 2020, it is approved in Korea in July 2021 for use in cases treated with Xalkori and at least one other ALK inhibitor, or with a primary ALK inhibitor as a monotherapy for the treatment of adult patients with ALK-positive progressive non-small cell lung cancer. Xpovio has not established a benefit standard for both permits, such as recurrent or refractory multiple myeloma and recurrent or refractory giant B cell lymphoma. The standard was set for Lynparza tab's standard. Meanwhile, in accordance with Articles 5 and 5-2 of the National Health Insurance Medical Care Benefit Standards, the HIRA may publicly announce drugs prescribed and administered to severely ill patients after deliberation by the Severe Disease Review Committee. The benefit criteria for the drug may be set differently according to clinical literature, domestic and foreign guidelines, and expert opinions within the scope of the efficacy and effectiveness of the MFDS' permission, and the benefit criteria may change during the follow-up procedure.
- Policy
- Daewoong tx are being developed for skin side effects
- by Lee, Tak-Sun Jan 18, 2022 06:06am
- Daewoong PharmaceuticalDaewoong Pharmaceutical has started to develop a treatment for skin side effects caused by anticancer drugs. This treatment is known as a stem cell-based drug. The MFDS approved the phase 2 clinical trial plan for DWP708 submitted by Daewoong Pharmaceutical on the 17th. This clinical trial is conducted to explore the efficacy of DwP1708 and evaluate safety for the treatment of EGFR inhibitor-related skin side effects. The test methods are multicenter, randomized, double-blinded, placebo-controlled, therapeutic search phase 2. It will be held at Gyeongsang National University Hospital and Dong-A University Hospital for 96 patients in Korea. EGFR inhibitor is mainly used in the treatment of arsenic lung cancer. It has a mechanism to reduce cancer cell proliferation by inhibiting EGFR, and types of anticancer drugs include treatments containing Erlotinib, Cetuximab, and Gefitinib. These anticancer drugs, however, are known to have frequent serious skin side effects. 50-90% of patients administered EGFR anticancer drugs suffer from skin side effects. Mainly, skin rashes, follicles, and dry skin occur in the face and upper body. Treatment of skin side effects is said to be managed with steroids or antibiotics, but the effect is not significant. Accordingly, Daewoong Pharmaceutical has been conducting research on related drugs, believing that EGFR inhibitory anticancer drugs block normal cell proliferation such as keratinocytes, causing skin side effects. According to the list of Daewoong's R&D tasks, DWP708 is indicated as a stem cell treatment. The study started in 2007 and received approval for phase 2 clinical trials this time and will begin full-scale human application research. The new drug candidate is known to be the first anticancer drug to be applied as a skin side effect treatment. Daewoong Pharmaceutical expects that it will contribute to controlling the dose of anticancer drugs and reducing the discontinuation rate due to skin side effects as a mechanism to suppress skin epithelial regeneration and inflammation. It is not possible to determine the exact market size as there are no anticancer drug skin side effects treatments, but if the drug is commercialized, it is expected to significantly improve the quality of life of cancer patients administering EGFR treatments.
- Policy
- Discord in system hinder coverage of rare disease drugs
- by Lee, Jeong-Hwan Jan 18, 2022 06:06am
- A suggestion has been raised that it is irrational for a drug that received an orphan drug designation to be ineligible for insurance benefits that were set for rare disease treatments to improve patient accessibility because its disease was not designated as a rare disease or set for special exemption of calculation. In other words, the treatment and disease burden born by the patients are worsening because the tools for orphan drug reimbursement evaluations such as the risk-sharing agreement system (RSA) and the pharmacoeconomic evaluation exemption system cannot be applied due to discord between the designation of orphan drugs and rare diseases, and this issue must be resolved. In particular, the benefits of reimbursement tools such as the RSA are too concentrated on anticancer drugs, leaving orphan drugs to be neglected from the system. Criticism also followed that the national orphan drug policy is more focused on providing convenience to the administrative system rather than being patient-focused in its operations. On the 17th, Jong Hyuk Lee, Professor of Chung-ang University’s College of Pharmacy, wrote so in a special column of ‘Angel Spoon,’ a magazine published by the Korean Organization for Rare Disease. Professor Lee argued that Korea should spend more on rare drugs as the current rate falls much below the global rate. As of 2018, Korea spent 370 billion won on rare disease drugs, which is 2.1% of its total drug expenditure. The global rate of expenditures on rare disease drugs exceeds 14%. Lee added that there is a need to improve the reality that stops patients from benefitting from systems that can reduce their medical costs, such as the RSA and the pharmacoeconomic evaluation exemption system that are in place in Korea. Patients cannot receive these benefits if their condition is not designated as a rare disease. Even if a drug is designated as an orphan drug used to treat rare diseases, the drug may not go through the RSA or pharmacoeconomic evaluation exemption track when undergoing evaluation for reimbursement benefits if its indicated disease is not recognized as a rare disease. This in turn triggers failure in reimbursement and increases the burden borne by patients. Also, the operation of the NHI coverage enhancement policy is too focused on anticancer drugs, harming the reimbursement of rare disease drugs in the process. 78%, or 32 of the 41 drugs that are applied RSA, a system that plays the biggest role in enhancing coverage of new drugs, were anticancer drugs. Another issue that was raised was that the system for rare disease drugs was designed to convenience government ministries rather than the patients. Orphan drug designation/approval is carried out by the Ministry of Food and Drug Safety; rare disease designation/ insurance registration/ special exemption of calculation by the Ministry of Health and Welfare and operated by the Korea Disease Control and Prevention Agency, the Health Insurance Review and Assessment Service, and the National Health Insurance Service; medical expenses support by each city/town/district; and the catastrophic medical expense support project operated by NHIS, rendering the processes too complicated for patients to follow. In the column, Lee emphasized that we need to expedite the approval of rare disease treatments to increase accessibility for the patients and increase special systems like the pharmacoeconomic evaluation exemption system for such diseases in the course of granting their reimbursement. Also, Lee added that social discussion should be held on diversifying finances for the coverage of rare diseases that are mainly sourced by NHI finances by raising funds for rare diseases, etc. Lee said, “We need to carefully examine whether any patients are left neglected due to institutional issues that create gaps between the designation of rare diseases and orphan drugs. The entities that operate such systems for rare diseases vary and the procedures are also very complicated. Therefore, the system should move away from such convenience-focused administrative practices and become more patient-centered.”
- Policy
- The gov will push for a refund on losses from suspension
- by Kim, Jung-Ju Jan 18, 2022 06:05am
- The government will push for a revision of the law that will put a brake on the suspension of execution filed by pharmaceutical companies in drug price lawsuits. If companies fight against the government over drug prices and finally win the case, the insurer will refund the drug cost equivalent to the loss so far. This is a revision of the legal unit, so it is highly likely to take effect immediately. The revision bill will three times shorten the negotiation period for the reduction of the original drug price of patent expiration, and includes amendments to omit drugs with a history of negotiations in the future. The MOHW announced today (17th) the "Partial Amendment to the Rules on the standards of national health insurance medical benefits" and began inquiring opinions. The revision is a plan already reported by the government to the Health Insurance Policy Deliberation Committee late last month, and is a follow-up to calls by the National Assembly to prevent abuse of execution and rationalize industry negotiations. ◆Introduction of the loss refund system according to the outcome of the dispute =The government explained that the purpose of the system is to protect the rights and interests of drug manufacturers and prevent financial losses in health insurance by applying for suspension of execution when pharmaceutical companies file drug lawsuits. The main contents of the amendment stipulate that the chairman of the National Health Insurance Service pays the manufacturer, etc. the loss if the drug manufacturer or the administrative trial's citation ruling is confirmed on whether the Minister of Health and Welfare is eligible, adjustment of upper limit price, and changes in details. On the contrary, if the government wins, a revision is currently pending in the National Assembly to recover the insurer's loss to the pharmaceutical company that filed the lawsuit. ◆Regulation of negotiation period and procedure for registered drugs and follow-up measures = The maintenance and measures were designed to prevent confusion in the pharmaceutical industry and secure predictability caused by unclear negotiation procedures and follow-up measures since October 2020. The main contents include allowing negotiations to be omitted for drugs with a history of existing negotiations and reasonably reducing the negotiation period from the current 60 days to the next 20 days in consideration of health insurance finances when the government adjusts the original drug ex officio. However, if the negotiations break down, renegotiation will be allowed after deliberation by the Drug Benefit Evaluation Committee, and if the negotiations break down, the drug will be excluded from medical care benefits. The MOHW plans to conduct an opinion inquiry until March 18 and implement it as originally planned if there is nothing special.
- Company
- Pharma exports exceed ₩9 trillion in 2021
- by Kim, Jin-Gu Jan 18, 2022 06:05am
- Pharmaceutical exports in Korea have exceeded ₩9 trillion last year. With exports increasing around biopharmaceuticals, sales of the drugs increased over 2.2 times over the past 2 years. Imports have also reached record-high, influenced by the increased import of Pfizer and Moderna’s COVID-19 vaccines. On the other hand, the export of diagnostic kits that have surged after the COVID-19 outbreak has slowed down. ◆Export of pharmaceuticals exceed $8 billion… led by Samsung Biologics and SK Bioscience According to the Korea Customs Service on the 16th, pharmaceutical exports last year amounted to $8,121,440,000 (approximately ₩9.69 trillion) (based on the exchange rate on the 16th of $1 to ₩1,190). This is a 18% increase from the $6,893,550,000 (approximately ₩8.20 trillion) in 2020. With such a surge in exports for 2 consecutive years, exports increased 2.2 times last year compared to 2 years ago. In 2020, exports increased 87% from the $3,695,910,000 (approximately ₩4.4 trillion) in 2019. Annual Pharma exports 2010-2021 The export of biopharmaceuticals has led the increase in exports. In 2021, export of biopharmaceuticals recorded $6,007,230,000 (approximately ₩7.15 trillion). This is a 24% increase from the $4,906,870,000 (approximately ₩5.84 trillion). In the same period, export of chemical drugs increased only 3% from $1,986,680,000 (approximately ₩2.36 trillion) to $2,049,150,000 (approximately ₩2.44 trillion). This increased export of biopharmaceuticals was led by Samsung Biologics and SK Bioscience. The accumulated exports of Samsung Biologics by Q3 last year was ₩930 billion, a 50% increase from the ₩620.4 billion in Q3 2020. In the same period, exports of SK Bioscience increased by over 14 times from ₩11.3 billion to ₩161.3 billion. CMOs of COVID-19 vaccines are expected to have directly influenced this increase in exports in the two companies. Samsung Biologics signed a CMO agreement with Moderna for the COVID-19 vaccine in May last year. Manufacture of Moderna’s vaccine began in earnest from Q3, sharply increasing the company’s exports this year. SK Bioscience signed a CMO contract with AstraZeneca to manufacture its COVID-19 vaccine from July 2020. Although the contract has expired at the end of last year, the company had recorded record amounts through exports of vaccine solutions and finished products. This year, the company plans to manufacture the Novavax COVID-19 vaccine in full this year. ◆Import of pharmaceuticals exceeds ₩10 trillion … due to the import of COVID-19 vaccine Imports of pharmaceuticals in Korea had also exceeded ₩10 trillion for the first time. Import of pharmaceuticals recorded $9,828,760,000 (₩11.7 trillion) last year, a 32% increase from the $7,432,960,000 (₩8.85 trillion). Such a great increase in exports is analyzed to have been greatly influenced by the import of COVID-19 vaccines. The import of COVID-19 vaccines in Korea had recorded $345,230,000 (₩410 billion) in 2020 and increased over 6 times last year to record $2,355,260,000 (₩2.8 trillion). Monthly imports showed that the amount was around $300,000,000 (₩360 billion) until March before Pfizer’s COVID-19 vaccine started to be imported in earnest, then jumped to $500,000,000 (₩600 billion) since April. Since June when Moderna’s vaccine was added to the imports, the amount surged to exceed $1,000,000,000 (₩1.19 trillion). Vaccine imports in September last year recorded the highest amount with $6,800,000,000 (₩8.9 trillion). ◆Export of the once-leading export product ‘K-diagnostic kits’ fell 6% … due to intensified global competition Sales of diagnostic kits that had settled as a leading export item in Korea since the COVID-19 outbreak had somewhat faltered. Its amount fell 6% from the $2,170,870,000 (₩2.58 trillion) in 2020. Last year, export of diagnostic kits recorded $2,046,670,000 (₩2.44 trillion). This was a 6% decrease from the $2,170,870,000 (₩2.58 trillion) in 2020. The domestic diagnostic kits have enjoyed a surge in sales since April 2020, when the COVID-19 broke out in full. Demand had risen across the globe to the extent that was unable to be met by the short supply. The performance of domestic diagnostic kit companies that quickly entered the market had improved greatly. However, since Q4 2020, the prolonged COVID-19 outbreak had increased the competition and reduced the unit price of products, consequently reducing the export amount. In fact, the export weight of domestic diagnostic kits had increased 48% from 5832 tons in 2020 to 8659 tons last year. In other words, the export weight increased by 1.5 times but the amount earned from exports had actually decreased.
- Opinion
- [Desk] The task of reimbursement for ultra-high-priced drugs
- by Kim, Jung-Ju Jan 18, 2022 06:05am
- The evaluation of the adequacy of the super-high-priced Kymriah's benefit is approaching insurance coverage. This is because the agenda to expand the health insurance standards for Kymriah, a treatment for acute lymphocytic leukemia and lymphoma CAR-T, and Keytruda, a non-small cell lung cancer immuno-cancer drug, passed side by side at the HIRA meeting on the 13th. Until now, the biggest obstacle when it was decisively blocked by cost-effectiveness in the process of reimbursement for high-priced drugs was the discussion process of the Pharmacist Evaluation Committee or the Cancer Disease Review Committee. Whenever the reimbursement priority and adequacy were discussed, the effect and necessity were recognized, but the cost was too high, so Kymriah's registration was delayed for 11 months, and Keytruda was also in progress for 4 years and 4 months. Patients' struggles to ensure these drugs were also tearful. Patients and their families who need immediate medication as life-related drugs have protested in front of the company to urge rational fiscal sharing, issued press conferences and statements, and filed petitions against the government, so the committee's passage will be impressive. Drug price negotiation procedures are still needed, and considering the period required for deliberation and resolution by the Health Insurance Policy Deliberation Committee, two to three more months must be waited. As such, it is a drug that is essential to patients, but it is an innovative drug that the general public cannot afford, and as technology continues to develop, similar cases like this are expected to continue to appear in the future. In fact, the government is aware of this and has prepared various mechanisms such as positive list system to strengthen the coverage of not only drugs but also medical technology, but it cannot cover all innovations with limited finances. Patients argue that a rapid health insurance registration system should be introduced for new drugs directly related to life to protect their lives at the national level, but the difficulty of accepting them from the government's point of view may be due to predictability and financial problems. As mentioned earlier, ultra-high-priced drugs such as Kymriah and Keytruda will be released every time, and the speed will increase as technology develops. Currently, in the face of such a problem, the authorities demand a rational fiscal sharing plan between the company and the government (insurer) from the developing company. There are already many cases empirically that such an agreement is not premised in the drug price negotiation process, which is another major obstacle. The government sought to strengthen the coverage and accessibility of ultra-high-priced drugs, but has repeatedly expressed its intention to use the existing negotiation mechanism under the positive list system. It is necessary to effectively establish a reasonable and predictable finance, that is, a voting structure and procedures to share mutual risks, and a mechanism to prevent delays in discussions without damaging the screening registration system, the central axis of drug registration. Academia should actively and actively conduct related research to present evidence and solutions to patients, the government, and companies, and the government and the National Assembly should consider them in practice to find solutions that our society can accept.