- LOGIN
- MemberShip
- 2026-05-07 17:04:06
- Company
- Fidanacogene Elaparvovec designated as an orphan drug
- by Eo, Yun-Ho Jun 12, 2023 05:42am
- Fidanacogene Elaparvovec, a one-shot hemophilia treatment, has been designated an orphan drug. The Ministry of Food and Drug Safety recently announced that it has selected Fidanacogene Elaparvovec, Pfizer's hemophilia B gene therapy, as an orphan drug. Fidanacogene Elaparvovec is a method that combines adenoviral vector (AVV) capsid and highly active coagulation factor IX gene and is characterized by producing coagulation factor IX in one cycle instead of regular injection am. The drug has been designated as a Breakthrough Therapy, Advanced Regenerative Medicine and Therapeutics (RMAT), and Orphan Drug by the US FDA, and PRIority MEdicines and Orphan Drug by the European EMA. The phase 3 BENEGENE-2 study confirming the effectiveness of Fidanacogene Elaparvovec also received significant attention. The study evaluated the efficacy and safety of Fidanacogene Elaparvovec in patients with a factor 9 of 2% or less, and the participating patients were evaluated for 6 years based on a single intravenous injection. The main purpose of this study is to determine how much gene therapy reduces ABR compared to SOC. According to the topline results released recently, the Fidanacogene Elaparvovec group met the primary endpoint by demonstrating non-inferiority and superiority compared to standard therapy in ABR. From 12 weeks to 15 months, the average ABR of the Fidanacogene Elaparvovec group was 1.3, whereas that of the standard therapy group was 4.43. Gene therapy reduced ABR by 71%, confirming its superiority over standard therapy. The main secondary endpoint was the ABR measured based on treatment. The Fidanacogene Elaparvovec group reduced treatment-based ABRs by 78% and annual infusions by 92% compared to the standard-of-care group. The Ministry of Food and Drug Safety is operating an orphan drug designation system to support the development of treatments for rare and incurable diseases. Among drugs used for the purpose of diagnosing or treating rare diseases, drugs that cannot be replaced or that are significantly improved over drugs that can be replaced may be designated as orphan drugs. If designated as an orphan drug, you can receive benefits such as being subject to expedited review at the time of product approval.
- Company
- Chong Kun Dang hastens way into diabetes Tx market
- by Kim, Jin-Gu Jun 12, 2023 05:42am
- Chong Kun Dang is working to accelerate the expansion of its diabetes treatment portfolio. In addition to its own ‘Duvie (lobeglitazone),’ the company received approval for diabetes combination drugs that contain 'Januvia (sitagliptin),’ which it had acquired rights for in Korea. The release of such combination drugs is considered the company’s strategy to preoccupy the market before the patent expiry of Januvia. According to industry sources on the 12th, Chong Kung Dang received approval for its Duvie Tab from the Ministry of Food and Drug Safety on the 9th. The drug is a combination between sitagliptin and lobeglitazone. The company had received approval for Duvie, a TZD-class diabetes treatment in 2013 as the 20th new new homegrown drug in Korea. In May, it acquired the license for MSD's DPP-4 inhibitor class diabetes treatment, Januvia. It acquired all rights, including domestic sales, distribution, licensing, trademark, and manufacturing, for not only Januvia but also Janumet and Janumet XR. As a result, Chong Kun Dang now owns 2 original drugs in the diabetes market. Immediately after acquiring Januvia, Chong Kun Dang obtained approval for a series of combination drugs that contain lobeglitazone and sitagliptin. On the 2nd of last month, Chong Kun Dang obtained approval for DuviMet-S XR, a three-drug combination for diabetes consisting of lobeglitazone, sitagliptin, and metformin. With the addition of Duvie-S to the lot, Chong Kun Dang's Duvie-based diabetes lineup has increased to amount to a total of 4: Duvie Tab which was approved in 2013, DuviMet XR Tab that was approved in 2016 (lobeglitazone + metformin), and DuviMet-S SR Tab and Duvie S Tab that were added this year. The company is expected to continue expanding its diabetes treatment portfolio for some time. Chong Kun Dang is currently conducting 4 clinical trials to treat diabetes: Phase III clinical trials for CKD-383, CKD-398, and CKD-371 and Phase I clinical trials for CKD-379. Among them, CKD-383 and CKD-379 are three-drug combination drugs for diabetes, presumably a combination product based on Duvie or Januvia. Chong Kun Dang The industry predicted that the imminent expiry of Januvia's patent was behind Chong Kun Dang's rapid expansion of its diabetes portfolio. Januvia's patent is set to expire in September. About 100 companies, excluding Chong Kun Dang and MSD, are expected to simultaneously release single and combination drugs that contain sitagliptin at the time of patent expiry. With such fierce competition being expected, Chong Kun Dang’s move is interpreted as a strategy to preoccupy the market by releasing a combination drug based on sitagliptin and lobeglitazone 2-3 months in advance. In particular, in a situation where the growth of both its Duvie series and the Januvia series are have been slowing down, attention is focused on how much synergy the combination of the two drugs will bring to the market. According to the market research institution UBIST, Duvie and Duvimet recorded a combined prescription amount of KRW 25.4 billion last year. This was a slight increase from the KRW 25.1 billion it made in 2021. In Q1 this year, the drugs recorded KRW 6.1 billion, down KRW 100 million from the previous year. The Januvia series recorded prescription sales of KRW 162.5 billion last year. The amount decreased by 8% compared to the KRW 176.3 billion the series had made in 2021. In Q1 this year, prescription sales were KRW 37.9 billion, down 9% from the previous year.
- Company
- Crystal Genomics supplies 2.6 billion Acelex to Russia
- by Lee, Seok-Jun Jun 09, 2023 05:36am
- Crystal Genomics will supply $2 million of Acelex, an osteoarthritis drug, to Russia. According to the company on the 7th, this order is based on a supply contract with PharmArtis International, a Russian state-run pharmaceutical company. This is the second order quantity. The contractual obligation to purchase is $43.86 million. Crystal Genomics will receive up to $77.6 million in milestones based on additional sales. PharmArtis is currently developing sales and marketing strategies and more. It will officially release Acelex in Russia next month at the earliest. The Russian COX-2 inhibitor market, to which Acelex belongs, is showing an annual growth rate of more than 30%. It is possible to expand the market to neighboring Eurasian Economic Community regions (Belarus, Kazakhstan, Armenia, Kyrgyzstan, etc.). The Eurasian Economic Community regional regulatory authorities are in the process of obtaining Acelex approval. Acelex is Korea's first bio-venture new drug. It selectively inhibits only COX-2 among cyclooxygenases (COX-1 and COX-2), two enzymes that act on the formation of prostaglandins that cause inflammation and pain. It was developed to overcome stomach cramps and gastrointestinal disorders, which are side effects of non-steroidal anti-inflammatory drugs. An official from Crystal Genomics said, "Following the shipment of the first volume at the end of March, we received an order for the second volume, so local sales are imminent. Sales in Russia are expected to improve performance."
- Company
- Korea’s new drug reimb rate is below OECD average
- by Eo, Yun-Ho Jun 09, 2023 05:35am
- A survey found that Korea’s new drug reimbursement rate does not amount to the OECD average. The Korean Research-based Pharmaceutical Industry Association (KRPIA, Chairman: Dong-Wook Oh) recently announced Korea’s new drug release status based on its ‘Global Access to New Medicines Report’ According to the report, it takes longer than the Organisation for Economic Co-operation and Development (OECD) country average for new drugs to be introduced to Korea after its global launch, and Korea’s release rate and reimbursement rate were also below the OECD country average. The report was based on the ‘Pharmaceutical Research and Manufacturers of America’ report that was published in April. PhRMA’s report subdivided 72 countries including Korea by G20, OECD, and region, and investigated the current status of new drug launches and health insurance coverage in each country. The report was prepared based on an investigation of a total of 460 new drugs that have received marketing authorization in the United States, Europe, and Japan over the past 10 years from 2012 to 2021. When taking a closer look, the report showed that the ratio of new drugs introduced to Korea within 1 year after the initial global launch was found to be less than half of the OECD average. While the average new drug introduction rate (non-reimbursement release rate) in OECD countries is 18%, Korea's level was only 5%, about a quarter of the OECD average. The countries with the highest new drug introduction rates were the United States (78%), Germany (44%), then the United Kingdom (38%). Also, Japan’s new drug introduction rate was nearly twice that of Korea, at 32%. By disease, it took about 27 to 30 months for anticancer drugs and new rare disease drugs to be released in Korea without reimbursement after their global launch. Compared to the average of 12 to 15 months it took in other advanced countries such as the UK and Germany and 18 to 21 months in Japan, it takes twice as long for new drugs to be launched in Korea. Moreover, it took a total of 46 months from the initial global launch of a new drug to reimbursement in Korea. The average time taken in other OECD countries was similar at 45 months, but compared to Japan (17 months) and France (34 months), it took from 10 months to more than twice longer for drugs to be reimbursed in Korea. Meanwhile, the share of new drugs covered by health insurance in Korea was 22%, which was below the OECD average (29%). This is only half that of Japan (48%) and the UK (48%). KRPIA said, “The report holds significance for allowing us to identify the rate and period of new drugs introduced and reimbursed in Korea after their global launch. Improvements should be made to Korea’s system and environment in advance to enable prompt reimbursement of global new drugs for patients in Korea.
- Company
- Non-covalent BTKi pirtobrutinib receives orphan drug desig
- by Eo, Yun-Ho Jun 09, 2023 05:35am
- The BTK inhibitor ‘pirtobrutinib‘ received an orphan drug designation in Korea. The Ministry of Food and Drug Safety recently announced that it had designated Lilly’s Bruton's Tyrosine Kinase (BTK) inhibitor Jaypirca (pirtobrutinib) as an orphan drug. The subject indication is as monotherapy for adult patients with relapsed or refractory mantle cell lymphoma who have been previously treated with a BTK inhibitor. The US FDA granted accelerated approval for Jaypirca in January. The drug received attention as the first non-covalent BTK inhibitor and as an alternative to patients who failed treatment with covalent BTK inhibitors like ‘Imbruvica (ibrutinib),’ ‘Brukinsa (zanubrutinib),’ etc. Jaypirca’s efficacy was evaluated through the BRUIN trial. Results showed that the overall response rate of patients that received 200mg of Jaypirca once a day was 50% (60 patients) among the 120 mantle cell lymphoma patients, with a complete response rate of 13% (15 patients) and a partial response rate of 38% (45 patients). The estimated median duration of response was 8.3 months, and the estimated duration of response rate at 6 months was 65.3%. The pooled safety analysis of the full BRUIN study population evaluated 583 patients with hematologic malignancies administered Jaypirca 200 mg daily as a single agent. In this pooled safety population, the most common adverse reactions (ARs) to Jaypirca therapy, occurring in 20% of patients or more, were decreased neutrophil count, decreased hemoglobin, decreased platelet count, fatigue, musculoskeletal pain, decreased lymphocyte count, bruising, and diarrhea. Meanwhile, the MFDS has been operating the orphan drug designation system to support the development of treatments for rare and intractable diseases. Among the drugs used to diagnose or treat rare diseases, drugs that are irreplaceable or those that significantly improved outcomes compared to their alternatives are designated as orphan drugs. Drugs designated as orphan drugs can receive benefits such as being eligible for accelerated review for marketing authorizations, etc.
- Company
- Humira with sales of 100 billion won
- by Chon, Seung-Hyun Jun 09, 2023 05:35am
- In the autoimmune disease treatment drug ‘Humira’ market, biosimilar products are not showing remarkable activity in the early stages. Although it has been two years since domestic companies penetrated the market, the market share of ‘Humira’ biosimilar has passed two years, but the market share was only 12%. It is analyzed that the price of original drugs and biosimilars did not show an explosive rise because the insurance price was similar. According to IQVIA, a pharmaceutical research institute on the 7th, the market for adalimumab ingredients in the first quarter was 24.5 billion won, up 9.8% from 22.3 billion won in the same period last year. Adalimumab is a TNF-alpha inhibitor that treats autoimmune diseases such as rheumatoid arthritis and psoriasis. AbbVie's Humira is the original drug. Humira posted sales of 104 billion won in 2019, 91.2 billion won in 2021 and 85.8 billion won last year, respectively. Samsung Bioepis and Celltrion released Humira biosimilars in 2021 and last year, respectively. The adalimumab market showed a moderate upward trend after falling 24.9% from 27.5 billion won in the first quarter of 2021 to 20.7 billion won in the second quarter. The sharp decline in sales in the second quarter of 2020 was due to a reduction in drug prices following the entry of biosimilars, which reduced the size of the market. When Samsung Bioepis listed Humira biosimilar Adaloche as a benefit in May 2021, Humira's upper limit also fell a month later. As of June 7, 2021, Humira's insurance cap has been reduced by 30%. The price of three drugs, Humira Pen 40mg/0.4mL, Humira Prefilled Syringe 40mg/0.4mL, and Humira 40mg vial, fell 30% from 411,558 won to 288,091 won, and Humira Prefilled Syringe 20mg/0.2 mL went down from 224,002 won to 156,801 won. Humira has maintained sales at a similar level after drug price cuts following biosimilar registration. Sales of Humira in the first quarter of last year were 21.5 billion won, up 1.5% from the same period last year. It increased by 4.0% from 20.7 billion won in the second quarter of 2021, right after the drug price cut. This means that sales volume has decreased due to drug price cuts, but sales since then have maintained the previous year's level. Analysts say that the pace of market penetration of biosimilar products is slow. In the first quarter of last year, Adalloce of Samsung Bioepis and Yuflyma of Celltrion posted a total of KRW 3 billion in sales. Adalloce and Yuflyma posted sales of 2.6 billion won and 400 million won, respectively. Adalloce posted its first sales of 400 million won in the third quarter of 2021 and exceeded 1 billion won in the first quarter of last year. Adalloce's sales in the first quarter of this year increased by 134.7% compared to the same period last year, but its share in the adalimumab market was only 10.6%. Adalloce's domestic sales will be handled by Yuhan Corporation. Yuflyma posted sales of 200 million won in the third quarter of last year, and sales in the fourth quarter of last year and the first quarter of this year were only around 300 million won. Celltrion Pharmaceuticals is in charge of the domestic sales of Yuflyma. The sales share of Adalloce and Yuflyma in the adalimumab market in the first quarter was 12.2%. It exceeded 10% for the first time in the fourth quarter of last year and recorded 12.5%, but this year it has decreased somewhat. The market share recorded two years after biosimilar penetration is analyzed as a disappointing report card. The upper price of Adalloce PFS 40mg/0.4ml and Yuflyma 40mg/0.4ml is KRW 244,877 each, and the gap between the drug price of Humira PFS 40mg/0.4ml and Humira Pen 40mg/0.4ml of KRW 288,091 is only 15.0%. In principle, under the domestic drug price system, biosimilars can receive up to 70% of the original drug price before the patent expires. From October 2016, up to 80% of products are guaranteed for 'items developed by a company that has entered into a joint contract between an innovative pharmaceutical company, an equivalent company, a domestic pharmaceutical company and a foreign company, or items for which Korea is the first licensed country or items produced in Korea. When biosimilars are released for original drugs whose patents have expired, insurance prices are automatically reduced to 70-80% of the previous level. Even if biosimilars are listed at a price that is 30% cheaper than the price of the original drug before the patent expires, it is not easy to secure price competitiveness as the drug price of the original drug is also reduced. There is also an analysis that medical staff and patients have no choice but to prefer new drugs from multinational pharmaceutical companies that have accumulated reliability for a long time in a situation where drug prices do not differ greatly due to the nature of autoimmune disease drugs used for severe diseases.
- Company
- Daewoong,"promoting to change Fexuprazan's partner"
- by Kim, Jin-Gu Jun 08, 2023 07:47pm
- Daewoong Pharmaceutical is going to change partners in charge of the development and commercialization of Fexuprazan in the North American market. Daewoong Pharmaceutical announced on the 5th that it has terminated the exclusive license agreement with Neurogastrx of the United States for the clinical development and commercialization of Fexuprazan in the US and Canada markets under an agreement between the two companies. The two companies signed a contract in June 2021. Daewoong Pharmaceutical explained that it decided to terminate the contract under the judgment that it needed a strategic partner that can quickly develop Fexuprazan for various indications in the North American market. Neurogastrx is said to have agreed to terminate the contract after determining that the development of Fexuprazan no longer aligns with its strategic business plan through pipeline re-evaluation. Upon termination of the contract, Daewoong Pharmaceutical has withdrawn all rights to clinical development and commercialization of Fexuprazan in the US and Canada. However, the 5% stake in Neurogastrix acquired by Daewoong Pharmaceutical at the time of the contract in 2021 is not returned. Daewoong Pharmaceutical started looking for a new partner. In particular, it has entered into negotiations with multiple multinational pharmaceutical companies that can simultaneously conduct clinical development not only in North America but also in global big markets such as Europe and Japan. Jeon Seung-ho, CEO of Daewoong Pharmaceutical, said, “With P-CAB receiving new attention from medical professionals around the world, we are communicating with multiple multinational pharmaceutical companies that can quickly settle Fexuprazan in the global market.” CEO Jeon Seung-ho emphasized, "We will secure additional indications for Fexuprazan in the global big market and accelerate the development of formulations to achieve 1 trillion won in sales by 2030 and develop it into a Best-In-Class within the family." In July of last year, Daewoong Pharmaceutical launched Fexuclue, a gastroesophageal reflux disease treatment in the P-CAB (Potassium Competitive Gastric Acid Blocker) class, as the 34th domestic new drug. Fexuprazan improves the disadvantages of existing PPI drugs and quickly and stably suppresses gastric acid secretion without activation by gastric acid.
- Company
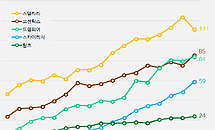
- Competition for interleukin inhibitors heats up in Stelara
- by Kim, Jin-Gu Jun 08, 2023 05:34am
- Stelara, Cosentyx, Skyrizi, and Tremfya (clockwise from top left) The interleukin inhibitor market also grew rapidly in the first quarter. Compared to the first quarter of last year, the market size increased by 26% in one year. While Stelara, the market leader, posted sales of KRW 11.1 billion, up 9.8% YoY, generics are rapidly catching up. Skyrizi saw a 69.7% year-over-year increase in sales and Tremfya's 43.4%. In particular, as generics are scrambling to add new content and expand the scope of reimbursement, competition in this market is expected to intensify in the future. The interleukin inhibitor market increased by 26% in 1 year, and Stelara remains No. 1 in the market for 12 years According to IQVIA, a pharmaceutical market research institute on the 5th, the size of the interleukin inhibitor market in the first quarter was 36.4 billion won. Compared to 28.8 billion won in the first quarter of last year, it increased by 26.4% in one year. Interleukin inhibitors are primarily used to treat psoriasis. It is divided into the IL-17 family and the IL-23 family according to the pathway that inhibits interleukin (IL). Janssen Stellara, which is classified as a first-generation product, is a mechanism that inhibits IL-12/23. Second-generation products, Novartis Cosentyx and Lilly Taltz inhibit IL-17, while Janssen's Tremfya and AbbVie Skyrizi, two successors, inhibit IL-23. This market was formed in 2012 when Stelara was added to the health insurance coverage list. Since 2015, successor products have been added one after another, and it is growing very rapidly. The actual interleukin inhibitor market size was only 10.4 billion won in the first quarter of 2019, but it expanded 3.5 times in 4 years to 36.4 billion won in the first quarter of this year. Analysts say that while Stelara, the market leader, is in good health, generics contributed to the expansion of the market by rapidly increasing sales. Stelara recorded sales of 11.1 billion won in the first quarter of last year. It increased by 9.8% from 10.1 billion won in the first quarter of last year. However, it decreased compared to 12.4 billion won in the fourth quarter of last year, which is interpreted as a result of drug price cuts according to PVA at the end of last year. Since last November, the government has lowered the price of Stelara by 0.2-4.4% by content. 2nd-generation drugs such as Cosentyx, Tremfya, and Skyrizi, heating up competition to expand coverage. The second-generation products released following Stelara are fiercely competing as their sales have increased significantly. Tremfya, which Janssen launched as a successor to Stellara, saw a 43.4% increase in sales within a year. Sales increased from 5.8 billion won in the first quarter of last year to 8.4 billion won. Novartis Cosentyx increased by 13.3% from 7.5 billion won in the first quarter of last year to 8.5 billion won in the first quarter of this year. In particular, with the rapid growth of the competing product Tremfya, the gap between the two products narrowed from 1.7 billion won to 100 million won in one year. AbbVie's Skyrizi, which was the latest to enter the market, is seeing the steepest growth in sales. Skyrizi's sales in the first quarter of last year were 5.9 billion won, up 69.7% from 3.5 billion won in the first quarter of last year. AbbVie launched this product in the second quarter of 2020. Cosentyx, Trempier, and Skyrizi are scrambling to expand their ranges, foreseeing even fiercer competition in the future. For Cosentyx, the high-dose product, which was newly approved in November last year, was additionally applied as a benefit from last month. In addition to the existing 150mg dose, up to 300mg dose is applied as compensation, aiming to expand sales. Tremfya has been covered for palmar and plantar pustulosis since this month. Skyrizi's benefit was expanded in March. Previously, benefits were recognized only for psoriasis, but from March, benefits were applied to active and progressive psoriatic arthritis. Lilly Taltz saw a modest increase in sales from 1.8 billion won in the first quarter of last year to 2.4 billion won in the first quarter of this year.
- Company
- K-Bio appealed for its competitiveness in Bio USA
- by Hwang, Jin-joon Jun 08, 2023 05:33am
- Major domestic pharmaceutical bio companies participated in the '2023 Bio International Convention (Bio USA)', a global partnering event, and introduced their competitiveness. Samsung Biologics disclosed an advanced target operation time for Plant 5 and a plan to build an ADC-only production plant. Lotte Biologics has sought to win biopharmaceutical CMO contracts with major pharmaceutical bio companies at home and abroad. Celltrion's goal to participate in the event was to increase its brand value through strengthening partnering. We discovered partners and held discussions related to new drug development. Bridge Biotherapeutics, Onconic Therapeutics, Inventage Lab, Abion, Vigencell, and Genome & Company announced the competitiveness of their new drug development pipelines. According to the industry on the 7th, major domestic pharmaceutical bio companies participate in Bio USA and introduce their competitiveness. Bio USA was held in Boston, USA on the 5th (local time). It will be open until the 8th. BioUSA is a global event in the biofield held every year by the American Bio Association. About 15,000 biopharmaceutical companies will attend, display new technologies and biotechnology products, and hold business meetings. According to the American Bio Association, 289 major domestic pharmaceutical bio companies participated in Bio USA this year. Of these, 41 opened publicity booths. Samsung Biologics CEO John Rim announced that BioUSA would shorten the target operating period for Plant 5 from September 2025 to April of the same year. It is expected to advance construction by about a year compared to the 180,000-liter third plant of the same size. According to Samsung Biologics, the early operation of Plant 5 is a strategy to preemptively respond to growing CDMO demand. In addition, the fact that new contracts with customers and the volume of existing agreements are increasing also influenced the decision to operate early. Starting with the 5th factory's construction, the 2nd Bio Campus will begin in earnest. Additional production facilities and an open innovation center will be built on the 2nd Bio Campus. The total investment is 7.5 trillion won. CEO John Rim also announced that he would build a production facility exclusively for ADC biopharmaceuticals to participate in the ADC market, which has recently attracted attention. Considering the market growth potential, it plans to start production of ADC commercial products within 2024. Lotte Biologics promoted the Syracuse plant's manufacturing technology, process development service, and quality system. It also revealed plans to build a large-scale factory in Korea by 2030 by spending 3 trillion won. Lotte Biologics prepared a meeting table and a private meeting room inside the booth. We discuss partnering for biopharmaceutical CMOs with about 30 major domestic and foreign pharmaceutical companies and bio companies that have been coordinated in advance. Celltrion participated in Bio USA and set up a promotional booth. (Photo by Celltrion) Celltrion and other bio companies engaged in the development of new drugs have also started to discover partners and introduce pipelines. Celltrion promoted its technological competitiveness and brand to leap forward as a new drug development company. An open meeting space and a private meeting room were set up inside the booth to discuss business. Potential partners were also explored to discover new modalities and new drug candidates. In the future, discussions for joint development are also planned. An official from Bridge Biotherapeutics introduces their new drug pipeline. (Photo by Bridge Bio) Bridge Biotherapeutics presented its corporate competitiveness and pipeline to industry officials. Business Development Director Pavel Princeev introduced major clinical challenges in the field of lung cancer and fibrosis, which the company is strategically focusing on. The fourth-generation lung cancer drug candidate 'BBT-176' phase 1 clinical trial data and self-discovered BBT-207 were shared. Innovative diagnosis technology recently acquired was also disclosed. Onconic Therapeutics sought additional technology transfer for its major pipeline P-CAB-type gastroesophageal reflux disease drug candidate 'Zastaprazan'. In addition, it introduced follow-up pipelines such as 'OCN-201', a dual inhibitor target anti-cancer drug. We plan to discuss building partnerships with domestic and foreign pharmaceutical bio companies. Inventage Lab introduced pipelines 'IVL3001' and 'IVL3002' using the long-acting injectable platform technology 'IVL-Drug Fluidics'. Data on the Alzheimer's treatment candidate 'IVL3003', drug/alcohol addiction candidate 'IVL3004', and prostate cancer treatment candidates 'IVL3008' and 'IVL3016' were also announced. It also presented a blueprint to enter the lipid nanoparticle (LNP) CDMO business based on the gene therapy development platform technology 'IVL-Genefluidic'. Abion promoted the technology transfer of 'ABN401', a c-MET treatment for non-small cell lung cancer, which is undergoing global phase 2 clinical trials, and 'ABN501', a candidate material in the non-clinical stage. Participating in K-Bio Showcase, a side event of Bio USA, introduces the representative pipeline to global investors and industry officials. Genome & Company will unveil its microbiome immuno-oncology treatment pipeline. Through more than 20 meetings, it has started strengthening business partnerships with domestic and foreign pharmaceutical companies. We introduce the development status of GEN-001', a major pipeline, and new target immuno-anticancer drug candidates GENA-104 and GENA-111. In addition to technology transfer, meetings related to joint development will be held.
- Company
- Commercialization of 3rd new P-CAB drug near
- by Kim, Jin-Gu Jun 08, 2023 05:33am
- The launch of the third product is imminent in the P-CAB (potassium competitive acid blocker) class gastroesophageal reflux disease treatment market that has grown to post sales of KRW 140 billion a year. With Onconic Therapeutics, a new drug development subsidiary of Jeil Pharmaceutical, filing an application for the marketing authorization approval of Zastaprazan, competition between this drug and the existing K-Cab (tegoprazan) and Fexclue (fexuprazan) is expected to intensify from next year at the earliest, triggering a 3-way race. The industry predicts that competition for indications between the three drugs will intensify along with the approval of Zastaprazan. P-CAB market nears sales of KRW 150 billion a year... 3rd drug applies for approval According to the pharmaceutical industry on the 7th, Onconic Therapeutics submitted a new drug application (NDA) for Zastaprazan to the Ministry of Food and Drug Safety. The company predicts the product will be approved next year. Therefore, the competition between drugs in this class could develop into a three-way race with HK Inno.n’s K-Cab and Daewoong Pharmaceutical’s Fexclue from next year at the earliest, P-CAB drugs have been showing explosive growth n the gastroesophageal reflux disease treatment market. According to UBIST, a pharmaceutical market research institute, outpatient prescriptions of P-CAB drugs in Korea last year recorded KRW 144.9 billion. This market was formed when HK Inno.N released K-Cab in Q1 2019. K-Cab, which posed KRW 30.4 billion in prescriptions that year, surpassed the KRW 100 billion mark in 2021, just 2 years later. Last year, the market expanded further with the addition of Daewoong Pharmaceutical’s Fexclue. Daewoong Pharmaceutical released the product in Q3 last year and recorded KRW 12.9 billion in prescriptions in the half year. In Q1 this year, K-Cab recorded KRW 35.7 billion and Fexclue recorded KRW 10.8 billion, respectively. If this trend continues, the market will expand to exceed KRW 150 billion by the end of the year. Quarterly sales of K-Cab and Fexclue (Unit: KRW 100 million, Data: UBIST) The industry predicts the P-CAB- market to continue its growth for a while even after Onconic's new product is added to the market. The expectations are that Zastaprazan’s sales will grow not by stealing the market share of K-Cab or Fexclue, but by replacing existing PPI (Proton Pump Inhibitor) drugs. K-Cab showed a 15% increase in prescriptions compared to the previous year, despite the rapid growth of Fexclue in Q1 this year. On the other hand, most of the PPI drugs show slower sales performance. Hanmi Pharmaceutical’s Esomezol (S-omeprazole), a representative PPI-type drug, recorded KRW 15.4 in prescriptions, up 5% from the previous year, and Nexium (esomeprazole) recorded KRW 8.7 billion, up 4% from the previous year. This is in contrast to the double-digit growth the drugs had made every year until the introduction of P-CAB drugs. The analysis is that P-CAB drugs replace existing PPI drugs with advantages such as faster drug expression and intake independent of meals. Competition to expand indications intensify… Zastaprazan plans to add gastric ulcer indication after erosive esophagitis The pharmaceutical industry predicts that the 3 drugs, including Zastaprazan, to compete more fiercely to expand their indications. Onconic plans to first receive approval as a treatment for erosive gastroesophageal reflux disease and then add indications for gastric ulcers. Onconic started a Phase III trial for gastric ulcer patients in May last year. An official from Onconic Therapeutics explained, “Clinical trials are underway to add indications to Zastaprazan.” Major indication extensions plans of P-CAB class drugs Currently, K-Cab has 5 indications: ▲erosive gastroesophageal reflux disease ▲non-erosive gastroesophageal reflux disease ▲gastric ulcer ▲antibiotic combination therapy for eradication of Helicobacter pylori ▲maintenance therapy after treatment of erosive gastroesophageal reflux disease (25mg). In addition, Phase III trials are underway as a preventive treatment for non-steroidal anti-inflammatory drug-induced gastric and duodenal ulcers. After first receiving approval as a ‘treatment for erosive gastroesophageal reflux disease', Fexuclue added an indication for 'improvement of gastric mucosal lesions in acute/chronic gastritis (10mg)'. Daewoong plans to secure 3 more indications for Fexuclu in the future. Phase III trials are already underway for the drug’s use as a 'maintenance treatment after treatment of erosive gastroesophageal reflux disease' and 'prevention of non-steroidal anti-inflammatory drug-induced gastric ulcer.’ In addition, the company plans to enter into clinical trials related to the eradication of Helicobacter pylori.