- LOGIN
- MemberShip
- 2026-04-24 08:27:12
- Policy
- Prices of top-priced generics could drop 32% upon pricing reform
- by Chon, Seung-Hyun Mar 18, 2026 09:11am
- The government’s announced tiered pricing system for generics is expected to function as a powerful mechanism that significantly lowers generic drug prices. Concerns have been raised that the faster exposure to the tiered pricing system compared to the current system could dampen the momentum for later-entering generics. It is estimated that the price of the 13th generic to enter the market will drop to half the current level, as the discount rate increases for products that fail to meet the highest-price criteria, such as those failing bioequivalence tests.Analysis suggests that even if a product meets the highest-price criteria, if more than 13 products are launched simultaneously, a new price reduction mechanism, which applies an additional 15% price cut one year later, will be triggered, causing the price of the first generic to fall by 32% compared to current levels.Tiered pricing system with 15% reduction applied from the 13th generic onward... Drug prices plummet due to stricter eligibility requirementsAccording to industry sources on the 16th, the Ministry of Health and Welfare presented a principle at the HIPDC subcommittee meeting held on the 11th that, under the price reform, the 13th generic would be subject to pricing. The price reduction rate applied to each tier would be 15%.The price drop for generics become greater with the reformThe tiered pricing system is structured such that the insurance ceiling price decreases on a monthly basis, the later a generic enters the market. Although it was abolished in 2012, the system was reinstated with the 2020 drug pricing reform. Under the current system, if there are more than 20 pre-listed products of the same formulation, the price of generics entering the market as latecomers is reduced by 15% at each step.When reporting the drug pricing system reform to the Health Insurance Policy Deliberation Committee on November 11 of last year, the Ministry of Health and Welfare proposed a policy to grant the first generic a price reduced by 5 percentage points (p) from the calculated price starting from the 11th listing of the same formulation. This effectively means the Ministry presented a relaxed version of the stepped pricing system just four months after its initial report.However, because the tiered system would now begin applying from the 13th generic instead of the 21st under the current system, products would be exposed much earlier to this additional price-cutting mechanism.The impact of the tiered pricing system becomes even greater as the generic pricing benchmark itself is lowered. The government has proposed lowering the benchmark for calculating generic drug prices from the current 53.55% to the low to mid-40% range, a reduction of about 10 percentage points. If the generic drug pricing benchmark drops from 53.55% to 43%, the maximum generic price would be reduced by 19.7% in absolute terms.For example, under the current drug pricing system, when the maximum generic price is KRW 53.55, the 21st generic’s price cannot exceed KRW 45.52, which is a 15% reduction. The price of 22nd and 23rd generics then drops to KRW 38.69 and KRW 32.89, respectively. The 24th generic would be KRW 27.95, and the 25th would be KRW 23.76, meaning drug prices decrease as generics enter the market later.Under the revised pricing system, where the generic drug pricing benchmark is set at 43%, if the maximum price is KRW 43, the price of the 13th and 14th generics drops to KRW 36.55 and KRW 31.07, respectively, following a 15% step-down reduction. The price of the 15th generic drops to KRW 26.4. Under the current drug pricing system, the 15th generic drug is not subject to the tiered pricing structure and can maintain a price of 53.55%, but under the revised system, its price drops to less than half that level.As the stricter maximum price requirements under the revised system will also be applied, the magnitude of price reductions under the tiered pricing system will become even greater.Since July 2020, under the revised generic pricing rules, a generic product must both conduct its own bioequivalence study and use a registered active pharmaceutical ingredient to qualify for the top price. For each unmet requirement, the ceiling price is reduced by 15%. If both requirements are not met, the price falls by 27.75%. Applying that 15% reduction, the 53.55% premium benchmark falls to 45.52% if one requirement is unmet and to 38.69% if both are unmet.According to the criteria introduced by the ministry after the 2020 pricing reform, “Even if a product meets both premium-price requirements, if there are already 20 or more listed identical products, starting from the 21st product, the price will be listed at 85% of the lower of the lowest price of the identical product or 38.69%.” The 38.69% rate is the result of two 15% reductions applied when a product failed to meet both of the highest-price criteria. (53.55% × 0.85 × 0.85)When 20 or more generics are listed, the 21st drug is listed at the lowest price vs 38.69%, whichever is lowerCurrently, the 21st generic drug,the first to be subject to the tiered drug pricing system, is priced at 32.86%, which is a 15% reduction from the 38.69% baseline. Compared to the highest price of 53.33%, this means the first generic drug subject to the tiered pricing system will see a 38.6% reduction. The prices of the 22nd and 23rd generic drugs will be reduced even further.The Ministry of Health and Welfare plans to increase the reduction rate applied when the highest price requirement is not met under the revised drug pricing system from 15% to 20%. If the benchmark for the highest generic drug price is set at 43%, the calculation standard will be further lowered to 34.40% for generics that fail to meet one requirement, and to 27.52% for generics that fail to meet both requirements.Under the revised drug pricing system’s tiered pricing structure, which applies the highest-price requirement, the ceiling price drops significantly starting with the 13th generic.The price of the 13th generic is calculated to drop from 27.52%, the rate applied to generics failing to meet two highest-price requirements, to 23.39%, a reduction of 15%. Comparing the same 13th generic drug, the price under the current system is KRW 53.55, whereas under the revised system, it drops to roughly half that amount. The 13th and 14th generic drugs each decrease by 38.6% from the lowest price, falling to KRW 14.36 and KRW 8.8, respectively.Under the revised drug pricing reform, the combined effects of shortening the sequence for applying the tiered pricing system, a 15% reduction upon tiered application, and a 20% price reduction for drugs that do not meet the highest-price requirement create a structure that effectively prevents late-entrant generics from entering the market.If 13 or more highest-priced generics are listed, prices will be reduced by 15% after one year.Even top-priced generics could see their prices cut a year later, depending on the number of products listed at the same time.To prevent excessive competition when the first generics enter, the ministry plans to apply pricing standards equivalent to tiered cuts to generics whose listing causes the total number of identical products to exceed 13.Even if a generic is listed within the first 12 products and initially receives the top price of 43%, if that product is among a group whose simultaneous entry causes the total to exceed 13, its price would be cut by 15% one year later.The price of generics with many listings will fall to a greater extent with the new reformFor example, if 8 generic products are listed in January and receive prices in the low-to-mid 40% range, and another 8 generics are listed in February, those products would all be treated as being listed simultaneously starting from the 9th position and could receive the top price. However, because the additional eight February listings push the total number of identical products beyond 13, those products would, one year later, fall to the 85% tiered level.Even the earliest listed generic could have its price cut by 15% one year later if 13 or more products enter simultaneously. In other words, even if it initially secures the top-price benchmark of 43%, it could later fall to 36.55% after one year. In that case, the price would be 31.75% lower than the current ceiling price.Within the industry, this is being interpreted as the government effectively intending to allow only up to 12 generics. Under restrictions on joint development, only three products may participate in one bioequivalence study. Generics beyond three such groups would face such sharp price declines that their incentive to enter the market would effectively collapse.One industry official noted, “Once the revised drug pricing system is implemented, late-entering generics will effectively be unable to turn a profit, so competition to secure the highest price by capturing the market first will inevitably intensify. Even if a company secures a leading position in the generic market, if there are many competing products, the tiered pricing mechanism will cause drug prices to fall, which could even lead to companies abandoning their plans to enter the generic market altogether.”
- Policy
- Boehringer discontinues original meloxicam 'Mobic Capsules' in KOR
- by Lee, Tak-Sun Mar 17, 2026 09:22am
- Product photo of 'Mobic Capsules''Mobic Capsules,' the original drug of the Non-Steroidal Anti-Inflammatory Drug (NSAID) meloxicam, is set to discontinue its supply in South Korea.Boehringer Ingelheim's headquarters has decided to stop imports, with product stocks expected to be depleted by April next year. The decision to withdraw from the Korean market is interpreted as a result of low sales performance and a market saturated with numerous alternatives of the same ingredient and class.According to industry sources on the 16th, Boehringer Ingelheim Korea has notified distributors regarding the discontinuation of Mobic Capsules.Boehringer Ingelheim stated, "The company has decided to discontinue the supply after recognizing that a wide variety of alternative treatments are already available in the Korean market." The company added, "Currently imported stocks will be supplied until they are exhausted, noting that the expected depletion date may vary depending on usage volume. The projected depletion for Mobic Capsules 7.5mg is around April next year, while Mobic Capsules 15mg is expected to run out by March next year."Mobic is an NSAID containing meloxicam as its active ingredient. It is effective in alleviating inflammation and pain associated with rheumatoid arthritis, osteoarthritis, and ankylosing spondylitis by selectively inhibiting COX-2. While it tends to have fewer gastrointestinal side effects compared to traditional anti-inflammatory drugs, it carries risks of blood clots and cardiovascular issues; therefore, it should be taken at the lowest effective dose for the shortest duration possible.Currently, there are 82 meloxicam products in South Korea at the 7.5mg dose and 15 at the 15mg dose. Even with the withdrawal of the original brand, the impact of the supply disruption is expected to be minimal due to the abundance of substitute medications.Mobic's sales have also been decreasing. According to UBIST, its outpatient prescription sales last year were KRW 1.5 billion, a 2% decrease from the previous year. This is a significant gap compared to Celebrex, another drug in the same NSAID class, which recorded KRW 41.7 billion in outpatient prescriptions last year.Consequently, Boehringer Ingelheim may have decided to halt supply to focus on other pharmaceutical products, given Mobic's low profitability in the Korean market.
- Policy
- Generics companies challenge ₩100B Lixiana·Livalozet mkt
- by Lee, Tak-Sun Mar 17, 2026 09:22am
- LixianaGeneric drug development is accelerating for Lixiana (edoxaban tosylate hydrate, Daiichi Sankyo Korea) and Livalozet (pitavastatin calcium + ezetimibe, JW Pharmaceutical), both of which are approaching the end of their exclusivity periods.Because both drugs have recorded more than KRW 100 billion in prescription sales, they are emerging as major targets for generic manufacturers this year and next.In the case of Lixiana, it has been disclosed that 13 products have applied for approval this year alone.According to the Ministry of Food and Drug Safety on the 16th, under the approval-patent linkage system, the original company was notified of marketing authorization applications filed for 13 generic products containing edoxaban tosylate hydrate in 2026.The recent increase in Lixiana generic approval applications is due to the fact that its substance patent is scheduled to expire on November 10 of this year.As for the pharmaceutical composition patent, which is scheduled to expire on August 21, 2028, most generic manufacturers have already succeeded in circumventing it through passive scope-of-rights confirmation trials. Once the patent expires, there will be no obstacles to launching generic drugs on the market.Lixiana is a direct oral anticoagulant (DOAC) whose prescription volume has continued to expand as it replaces warfarin. According to UBIST, its outpatient prescription sales alone reached KRW 117.5 billion last year.Generic companies had made attempts to invalidate the substance patent due to its strong commercial value, but failed. In addition to the original product, nine follow-on companies have already obtained product approvals, and their generic versions are expected to be launched after the substance patent expires.Since no follow-on product has secured first generic exclusivity, it appears that many products will flood the market simultaneously upon expiry of the substance patent, without restrictions on sales.LivalozetThere is also a strong possibility that a large number of generics will emerge for Livalozet, which posted KRW 117 billion in outpatient prescription sales last year. On the 13th, Nelson Korea, Pharmbio Korea, and Austin Pharmaceuticals all received approval on the same day for their bioequivalence study protocols for generic development.Generic development has already begun in earnest since 2024, with many pharmaceutical companies aiming to file applications. Applications can be submitted after the Livalozet re-examination period ends on July 27 next year.Livalozet is a dyslipidemia treatment combining pitavastatin and ezetimibe, and it has gained popularity due to its lower risk of diabetic side effects and strong lipid-lowering efficacy.In addition to the original company, JW Pharmaceutical, 5 companies have directly launched products with the same active ingredients through their own clinical trials. Ahn Gook Pharmaceutical, Boryung Pharmaceutical, Dongkwang Pharmaceutical, Hanlim Pharm, and Daewon Pharmaceutical launched related products in the second half of 2023 and have continued rapid growth. Last year, Ahn Gook Pharmaceutical’s Pevarozet recorded KRW 29.2 billion in outpatient prescription sales, while Daewon’s Tavalozet recorded KRW 18.2 billion. These companies also succeeded in launching their products early by invalidating Livalozet’s use patent.More recently, low-dose products combining pitavastatin 1 mg and ezetimibe 10 mg have also appeared. In January, Ilsung IS, Ildong, Daewoong, and Hanlim obtained approvals for related products, and this month, JW Pharmaceutical also received approval for a new product.Given recent sales trends, there is a high likelihood that generic drugs will flood the market once the re-evaluation period ends next July.
- Policy
- The key elements of government’s drug pricing reform plan
- by Jung, Heung-Jun Mar 17, 2026 09:22am
- Under the government’s new drug pricing reform, price cuts for already listed drugs are expected to begin in the third quarter, with preferential treatment for innovative companies’ new listings expected to begin next year.Innovative companies are set to receive a temporary exception from price cuts for existing drugs. However, the duration of these exceptions is scheduled to be finalized at the Health Insurance Policy Deliberation Committee meeting on the 26th.In addition, the fast-track listing–post-evaluation and adjustment track is planned for 2028, and its criteria will be established to select which products are eligible.According to industry sources on the 16th, the Ministry of Health and Welfare plans to finalize the drug pricing reform at the HIPDC plenary meeting on the 26th. Based on the discussions at the subcommittee, the direction of the reform will be fixed after some further adjustments.AI-generated image◆ Generic pricing ratio in low-to-mid 40% range…failure to meet required criteria: 85% → 80% = The pricing ratio, which had been described as “in the 40% range” last November, has recently narrowed to the low-to-mid 40% range at the HIPDC subcommittee.The industry expects it to fall somewhere between 43% and 45%. Since many had hoped for the high 40% range, the industry had expressed disappointment. However, because time remains before the HIPDC meeting, the final rate still remains to be seen.If the required conditions for generic pricing are not met, the pricing level is expected to be lowered from 85% to 80%. If a company does not submit its own bioequivalence data or fails to use APIs registered with the MFDS, the reduction rate will become greater.◆ Price cuts for already listed drugs to begin in Q3…cuts will be deferred for innovative companies, but duration undecided=The government plans to begin price cuts for already listed products in Q3. It intends to divide companies into groups based on their listing dates and implement sequential adjustments using varying calculation rates.Innovative pharmaceutical companies will be granted a deferral period under a separate pricing calculation rule. However, an exception is being discussed for ingredients with 21 or more listed products. A deferral period of 4 to 5 years has been mentioned, but it will be finalized at the HIPDC meeting on the 26th.For innovative companies, their reduction ratio under the price-volume agreement will be raised from 30% to 50%, with application expected to start at the end of this year.◆ New “innovation-equivalent” category to be established next year…50% preferential pricing=When newly listing drugs, innovative companies will receive a 60% pricing add-on. The period for this premium will be 1+3 years. If the product is manufactured domestically, an additional 3 years will be granted.In addition, the government is considering adding a category of companies equivalent to innovative firms, with 50% preferential pricing. These “quasi-innovative” companies would be defined based on annual sales above or below KRW 100 billion, with R&D investment ratios of at least 5% or 7% of sales, respectively.However, companies that have received administrative sanctions for rebates within the past 5 years will be excluded. The preferential period for quasi-innovative companies is expected to be the same as that for certified innovative companies.The preferential measures for innovative and quasi-innovative companies are expected to take effect starting next year.◆ Stepwise price reduction from the 13th product onward…15% reduction rate maintained=To manage multiple generic listings, the government plans to strengthen the stepwise reduction system. The stepwise reduction, which currently begins from the 21st product, is expected to shift to the 13th listed product.The reduction rate itself is expected to remain unchanged, with the price set at 85% of the previous lowest price. However, if multiple products are listed at the point when the 13th product is entered, the price of those products will be adjusted to the 85% level one year later.The strengthened stepwise reduction and multi-product listing management are expected to take effect next year.◆ 100-day fast-track listing to be institutionalized next year…selection criteria for ‘innovative new drugs’ to be established=The government plans to institutionalize fast-track listing for innovative new drugs next year. It also plans to establish post-evaluation and price-adjustment mechanisms.Selection criteria will also be established for determining which innovative new drugs will qualify for fast-track listing. The operation of a grading system is also under review.The government plans to operate a performance-based evaluation model incorporating digital healthcare elements such as hospital EMR systems and AI data, and is also reviewing the creation of a specialized agency.It appears that decisions to delist drugs will be made through post-market evaluation. The plan is to establish an evaluation and adjustment system to determine selective reimbursement coverage and appropriate drug prices.◆ Flexible drug pricing contract system to expand in Q2…will prepare measures to prevent inconvenience to patients =The expansion of the so-called ‘flexible drug pricing contract system,’ which allows for contract prices different from the listed price, is expected to be implemented in Q2.The expanded scope is expected to include listed new drugs, already listed off-patent originals, new drugs whose RSA refund period has ended, incrementally modified new drugs, and biosimilars.As the number of products subject to dual pricing increases, the government is preparing measures to prevent inconvenience to patients. One approach under review is to charge patients based on the separately contracted price rather than the listed price, thereby preventing the need for refunds.◆ Price evaluation and adjustment every 3–5 years…targeting ingredients 5 years after first generic entry=The government is establishing a system for evaluating and adjusting drug prices every 3–5 years. This involves comparing, by ingredient, ▲the number of products, ▲market structure, and ▲drug prices in major countries.The target appears to be ingredients for which 5 years have passed since the first generic entered the market. The specific operational model will be established and implemented after gathering industry feedback.The Ministry of Health and Welfare is reportedly expected to revise and supplement the reform plan containing these elements and approve it at the HIPDC meeting on the 26th.
- Policy
- Approval of HER2-bispecific Ab 'zanidatamab' in KOR imminent
- by Lee, Tak-Sun Mar 16, 2026 09:25am
- BeOne Medicines' 'zanidatamab (product name: Ziihera 300mg),' a HER2-targeted bispecific antibody, is likely to be approved soon.Zanidatamab is the first HER2-targeted therapy for biliary tract cancer (BTC) approved by the U.S. FDA. Once it is introduced to the Korean market, this drug is expected to improve treatment outcomes for local patients significantly.According to industry sources on the 12th, the Ministry of Food and Drug Safety (MFDS) recently completed the safety and efficacy evaluation for zanidatamab. Once a "suitable" decision is made from this review, only the final administrative granting of the license remains. Furthermore, under the "Approval-Reimbursement Linkage System," the company can apply for insurance coverage through the Health Insurance Review and Assessment Service (HIRA) immediately following the completion of the safety/efficacy review.In November 2024, the U.S. FDA granted accelerated approval of zanidatamab for patients with previously treated HER2-positive advanced or metastatic BTC. There has been a high unmet medical need in HER2-positive BTC patients.Treatment options for HER2-positive BTC have been extremely limited. BTC is known for its poor prognosis, with a 5-year survival rate of less than 5% once metastasized. In the pivotal HERIZON-BTC-01 clinical trial, the zanidatamab group showed an objective response rate (ORR) of approximately 52%, and a median duration of response (DOR) of approximately 14.9 months.Following its FDA approval, launch in Korea was expedited after the drug was selected for the Global Innovative products Fast Track (GIFT) program in December 2024. In Korea, the specific indication was submitted for approval for the treatment of adult patients with previously treated, unresectable, locally advanced, or metastatic HER2-positive (IHC 3+) biliary tract cancer.Unlike conventional monoclonal antibodies, zanidatamab is a bispecific antibody that binds simultaneously to two distinct sites on the HER2 (Human Epidermal Growth Factor Receptor 2) protein. This dual-binding mechanism allows for more potent inhibition of cancer cell growth.Beyond BTC, clinical development is currently expanding into gastric and gastroesophageal junction (GEJ) adenocarcinomas.BeOne Medicines underwent a global corporate rebranding last year. Beigene Korea was originally a China-based oncology developer; the company later changed its name to BeOne Medicines and moved its legal registration to Switzerland. In Korea, the company already supplies innovative oncology drugs, including Brukinsa (zanubrutinib) and Tevimbra (tislelizumab).Ziihera is a new drug originally developed by Jazz Pharmaceuticals. BeOne Medicines holds the commercialization rights for the Asia-Pacific region.
- Policy
- Bill proposed to apply 5% coinsurance for follow-up tests
- by Lee, Jeong-Hwan Mar 16, 2026 09:25am
- An amendment to the National Health Insurance Act is being proposed to reduce the patient coinsurance rate to 5% for follow-up examinations for patients with cancer, rare diseases, and severe or intractable illnesses.A bill to amend the Patient Safety Act has also been introduced in the National Assembly. It imposes an obligation on doctors to fully explain the details and circumstances of medical accidents to patients, while stipulating that expressions of consolation, empathy, or regret made by doctors during this process cannot be used as evidence of the doctor’s liability or to their disadvantage in medical malpractice lawsuits.On the 13th, Rep. Kyo-heung Kim of the Democratic Party and Representative Cheol-soo Ahn of the People Power Party each introduced a bill to amend the National Health Insurance Act and the Patient Safety Act, respectively, containing these provisions.Bill to reduce patient coinsurance for follow-up tests for serious diseases to 5%Representative Kim’s bill proposes reducing the patient coinsurance rate to 5% for follow-up examinations for cancer, rare diseases, and severe or intractable diseases that have a high risk of recurrence.Under the current law, insured individuals and their dependents are required to bear part of the cost of medical services when receiving healthcare benefits.For certain diseases with high treatment costs, a special calculation system is applied in which the patient’s share of medical costs is reduced to 5–10% of the total medical benefit costs for a specified period.Rep. Kim pointed out that while cancer, rare diseases, and severe or intractable diseases carry a high risk of recurrence even after treatment, making continuous monitoring and follow-up examinations essential, patients face high testing costs once the special calculation exception ends after the disease is cured.He argued that this creates a problem where patients are unable to receive proper follow-up examinations due to such a financial burden.To address this, accordingly, Representative Kim introduced a bill that reduces the patient cost-sharing rate to 5% when cancer patients, rare disease patients, or patients with severe or intractable diseases undergo follow-up examinations for their respective conditions.Bill preventing doctors’ expressions of sympathy after medical accidents from being used as evidence in lawsuitsRepresentative Cheol-soo Ahn introduced a bill requiring heads of healthcare institutions or healthcare professionals to make efforts to sufficiently explain the details and circumstances of patient safety incidents. The bill also stipulates that expressions of consolation, empathy, or regret made by doctors during this process cannot be used as evidence regarding the doctor’s liability in medical malpractice lawsuits or other legal proceedings.Under the current law, there is no separate provision requiring doctors to explain the details or circumstances of patient safety incidents.In particular, doctors sometimes avoid providing explanations or maintain a defensive stance regarding patient safety incidents, as expressions of humanitarian regret or explanations could later be used as evidence of the healthcare institution’s or healthcare professional’s liability in future trials related to the incident.Representative Ahn expressed concern that the information gap and lack of communication between doctors and patients deepen distrust among patients and their guardians toward healthcare institutions, turning simple incidents into complex legal disputes.He also noted that this situation creates excessive psychological pressure and litigation burdens on healthcare professionals, contributing to instability in the medical environment.The proposed bill would therefore require doctors to provide detailed explanations about medical accidents, while explicitly stating that expressions of sympathy, empathy, or regret cannot be used as evidence of liability in medical lawsuits.Rep. Ahn explained, “This bill aims to restore trust between healthcare professionals and patients and minimize unnecessary social costs, such as those arising from litigation.”
- Policy
- Pace of expanding low-priced purchase incentive has been adjusted
- by Jung, Heung-Jun Mar 16, 2026 09:25am
- The government is expected to adjust the schedule of its implementation policy, considering the potential side effects of expanding low-priced purchase incentives for medical institutions.While the original plan was to increase the incentive for low-priced purchases from 20% to 50%, officials are now reviewing a plan to lower the target to 35%.According to industry sources on the 16th, the government's fundamental policy direction of transitioning market price reductions into a 'market-linked, transaction-based price system' remains unchanged.However, given the potential negative consequences of a rapid expansion of incentives, the Health Insurance Policy Deliberation Committee is expected to lower the planned increase rate at its meeting this month.The low-price purchase incentive is a system that provides a portion of the savings as a grant to medical institutions when they purchase medicines at a transaction price below the maximum reimbursement limit. Under the current standards notified by the Ministry of Health and Welfare (MOHW), approximately 20% to 30% of the price difference is paid to these institutions.The reform plan released last November included a proposal to expand the incentive payout rate to a maximum of 50%. It is intended to lower actual transaction prices through market competition without relying on government-mandated price cuts.Since the announcement, there have been persistent concerns about overheated price competition and chaos in the distribution order. Critics have pointed out that the side effects of damaging the industry ecosystem could outweigh the benefits of reduced pharmaceutical expenditures.During a recent National Assembly debate on the drug pricing system, specific concerns were raised regarding the potential for rebates following the expansion of these incentives.Hong Seok-hwan, the Policy Director of the Korean Confederation of Trade Unions, criticized the move by stating that rather than the government directly removing the price bubble, the policy induces pharmaceutical companies and hospitals to enter into side agreements at prices lower than the officially notified price, effectively legalizing rebates by guaranteeing the price difference as profit.The government is adjusting the rate of increase in incentives downward to address these concerns. The proposed 35% rate is currently under discussion, and the final payout rate will be decided at this month's Health Insurance Policy Deliberation Committee meeting.Meanwhile, this expansion of low-price purchase incentives applies only to private tertiary general hospitals, general hospitals, clinics, and pharmacies. National and public hospitals are expected to maintain the current 20% rate.
- Policy
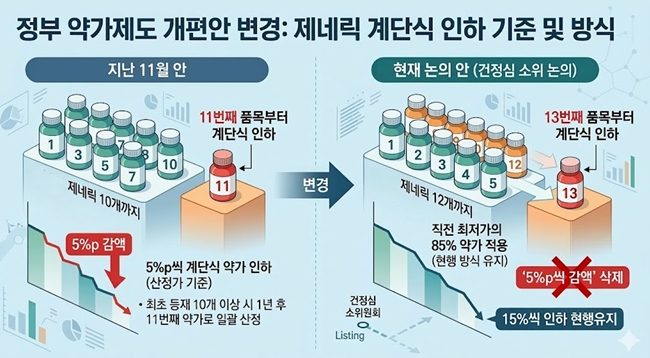
- Stepwise price reduction applied from 13th listed product
- by Jung, Heung-Jun Mar 16, 2026 09:25am
- The government, which had planned to implement stepwise drug price reductions of 5 percentage points starting from the 11th listed product, has decided to revise both the criteria for listing items and the reduction method.The new direction under discussion is to apply stepwise reductions starting from the 13th listed product, while maintaining the current method of applying 85% of the previous lowest price as the new price.According to industry sources on the 16th, the government’s drug pricing system reform proposal discussed at the recent Health Insurance Policy Deliberation Committee (HIPDC) subcommittee differs from the proposal announced in November last year.The stepwise drug price reduction reform plan is expected to undergo partial changes. AI-generated imageThe reform plan from last November included a method whereby “the price of the first generic would be reduced by 5 percentage points starting from the 11th product listing of the same formulation.” This represented a significant strengthening of the current product reduction system, which is applied starting from the 21st listed product.Additionally, the plan stipulated that if there were 10 or more initially listed generics, the price would be uniformly set at the 11th product’s level one year later.At this HIPDC subcommittee meeting, a proposal to apply the stepwise reduction starting from the 13th product was discussed. While the method of reducing the price by 5 percentage points each time would be removed, discussions are focused on maintaining the approach of applying a price set at 85% of the lowest price for subsequent listings. This effectively means maintaining the current reduction rate of 15% applied from the 21st product onward.Products listed at the point where stepwise reductions begin will have their prices adjusted to 85% of the lowest price after one year.For example, if there were 10 products this month and the number exceeds 13 with next month’s listings, the price for the newly listed products will be set at 85% one year later. However, if additional items are listed the following month, their prices would be further reduced by an additional 85% on top of the 85% already applied.Although the threshold number of products used as the basis for stepwise reductions has been slightly relaxed, the system still effectively results in an additional 15% reduction following the initial 15% reduction, depending on the listing timing.However, the details currently under discussion are not yet finalized. There is a possibility that the application method or specific figures could change at the HIPDC meeting on the 26th.Additionally, since the generic drug pricing rate is not clearly defined as being in the “mid-to-high 40% range,” the exact drug price calculations under stepwise reductions will likely be possible only after the HIPDC decision is finalized.
- Policy
- Debate over fast-track listing of new drugs continues
- by Jung, Heung-Jun Mar 12, 2026 08:35am
- Debate continues over the proposed reform of the drug pricing system aimed at shortening the reimbursement listing period for new drugs. On one side are calls to strengthen treatment access, while on the other are concerns that the reform could sharply increase the burden on Korea’s national health insurance budget.While working bodies like HIRA are preparing to strengthen post-listing control, concerns persist about the effectiveness and potential side effects of expedited listing.The Korean Pharmacists for Democratic Society, which previously called for reconsideration of the fast-track listing initiative together with the Citizens' Coalition for Economic Justice (CCEJ) last month, plans to hold a briefing session next week to point out issues in the proposed new drug pricing reform.AI-generated imageOn the 10th, the Korea Alliance of Patient Organizations urged the government to promptly implement the fast-track listing and post-listing evaluation system, arguing that delays in national health insurance reimbursement after regulatory approval for anticancer drugs and rare disease treatments are reducing treatment accessibility.The government recently announced that starting in 2026, it will launch a pilot program for a “fast-track listing–post-evaluation system,” which would shorten the reimbursement listing period for rare disease treatments from the current maximum of 240 days to within 100 days. The alliance called for the swift implementation of the measures.KAPO emphasized, “Rather than blocking entry at the early stage, it is important to establish a sophisticated post-listing evaluation and management system based on treatment performance.”Until recently, attention surrounding the drug pricing reform had focused largely on price cuts for already-listed drugs, while the direction of the fast-track listing reform for new drugs received relatively less attention. The pharmaceutical industry, which has little reason to oppose faster market entry, had been cautiously waiting for details of the implementation plan.However, the issue gained prominence in February when groups including CCEJ, the Korean Association for Serious Diseases, and the Korean Pharmacists for Democratic Society urged reexamination of the fast-track listing policy. They also called for a public hearing on issues including raising the ICER threshold and introducing flexible drug pricing agreements.Criticism calling for broader social discussion is expected to continue this month. The Korean Pharmacists for Democratic Society is expected to hold a briefing on March 18 on the proposed new drug pricing reform and express opposition to the fast-track listing policy.An official from the organization said, “Although there are measures to evaluate outcomes and provide refunds if necessary, there are no clear post-management mechanisms, such as price reductions or market withdrawal, after post-evaluation. As the financial burden on the National Health Insurance is expected to surge, preparing countermeasures must be prioritized.”The official continued, “There are also doubts about whether strengthened post-listing management measures are appropriate for Korea. The briefing will address the impact of the overall new drug pricing reform, including fast-track listing.”Critics also argue that the reform of the new drug pricing system, like the proposed price cuts for generics, lacked sufficient prior discussion.A pharmaceutical industry official said, “As with the generic drug pricing reform, this situation arose because sufficient discussion was not conducted before the reform plan was announced. If there had been even a process to coordinate the implementation measures, the current backlash would not have occurred.”
- Policy
- ‘Active INN prescribing needed for drugs with supply shortages’
- by Jung, Heung-Jun Mar 12, 2026 08:35am
- Han-sook Kim, Director of Health Insurance Policy Division, MOHWThe government said it is reviewing various measures while expressing support for introducing INN (international nonproprietary name) prescribing for drugs with unstable supply.It also stated that it will continue consultations to minimize the impact of the drug pricing reform plan on the field and will further refine the system even after implementation.On the 11th, Han-sook Kim, Director of the Health Insurance Policy Division at MOHW, stated at the National Assembly's drug pricing system discussion forum, " There are many concerns surrounding INN prescribing due to conflicting stakeholder interests. From the perspective of preventing supply instability, it is necessary to actively utilize INN prescribing. We are reviewing various aspects, including the criteria for supply instability and the safety, efficacy, and effectiveness of INN prescribing.”When asked about the direction of the drug pricing reform, Kim said the government would consider the policy’s impact on the field and remain open to consultations and improvements.Director Kim explained, ”The government is also paying close attention to the sustainability of the National Health Insurance finances. We are making multifaceted efforts. Improving unreasonable structures is crucial, and advancing the drug pricing system reforms is part of that effort.“Kim added, “This is an agenda with diverse perspectives. The government cannot clearly predict the impact the reform will have on the field once implemented. The government cannot clearly predict the impact on the field when drug pricing system reforms are implemented. Even after the reform is implemented, we will strengthen monitoring and make improvements if problems arise.”Kim dismissed concerns that the drug pricing reform plan concentrates insurance spending on multinational pharmaceutical companies, and said that it was a misunderstanding.Kim said, “The drug pricing reform could be misunderstood as a policy favorable to the global pharmaceutical industry. But promising companies and technologies must receive investment. If cost savings are generated through the reform, it will be used to properly reward companies investing in R&D.”