- LOGIN
- MemberShip
- 2026-04-15 16:01:26
- Roche Diagnostics Korea posts record performance
- by Hwang, byoung woo | translator Alice Kang | 2026-04-15 08:04:05

Roche Diagnostics, the leader in in vitro diagnostics, has been delivering clear top-line growth every year on the back of its broad portfolio.
Even over the past five years, sales have shown a steady upward trend, and the company is credited with firmly establishing growth momentum through the expansion of companion diagnostics and the successful market penetration of digital solutions.
Channel expansion and companion diagnostics growth… 5 consecutive years of top-line expansion
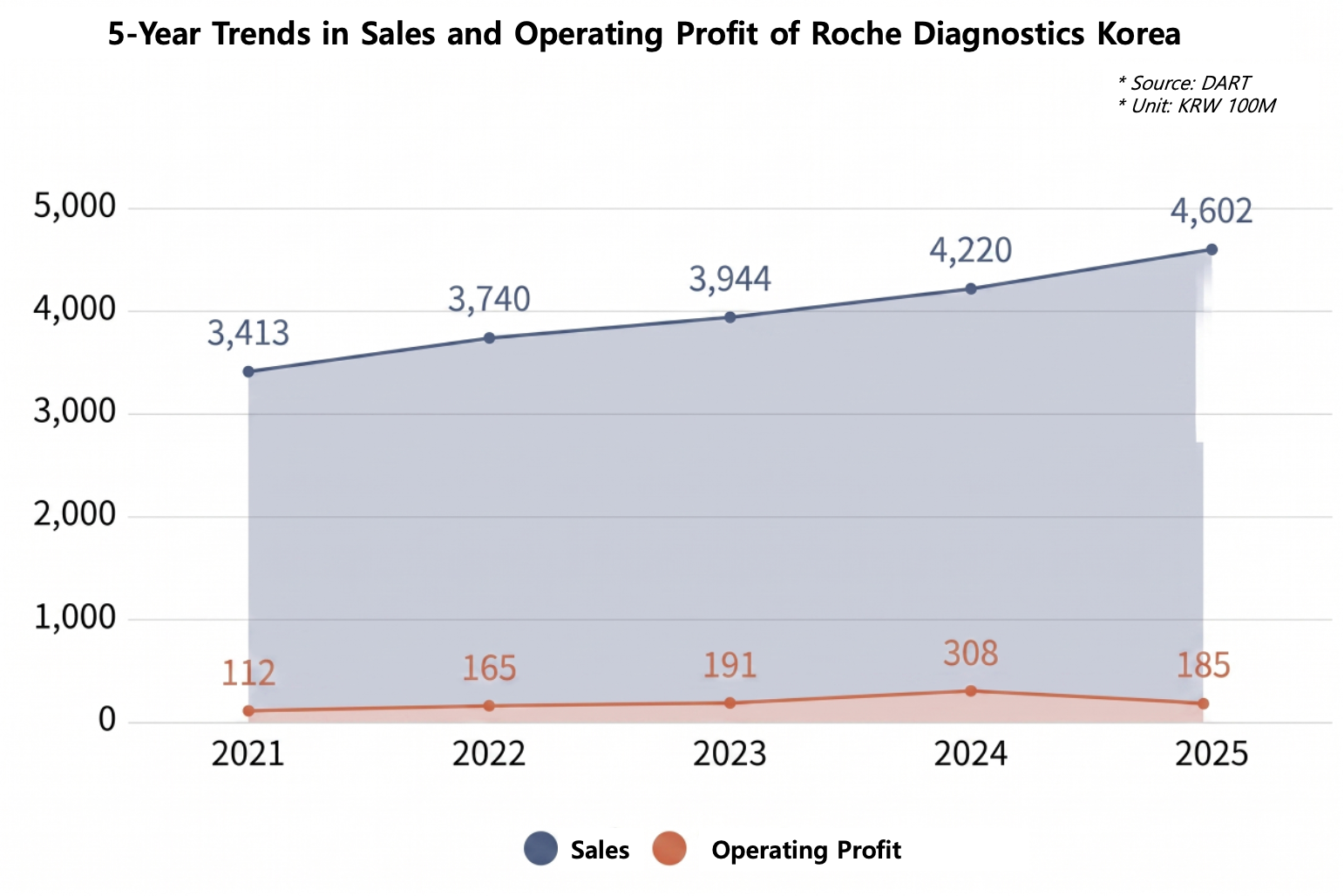
According to the audit report of Roche Diagnostics Korea, sales in 2025 reached KRW 460.3 billion, up approximately KRW 38.3 billion from KRW 422.0 billion in the previous year.
Looking at revenue over the past 5 years, the company has shown a steady upward trend: ▲KRW 341.3 billion in 2021, ▲ KRW 374.0 billion in 2022, ▲ KRW 394.4 billion in 2023, and ▲ KRW 422.0 billion in 2024.
The expansion of sales and distribution channels is cited as the primary driver of this revenue growth.
Analysts note that as access to testing expanded during the COVID-19 pandemic, the company shifted from a structure centered on large hospitals to one that includes medium-sized hospitals and testing centers, broadening its customer base and increasing the number of equipment installations.
Given the nature of the in vitro diagnostics business, where reagent and consumables sales recur after equipment installation, this channel expansion is highly likely to provide a foundation for mid- to long-term growth.
The expansion of companion diagnostics is also cited as a factor driving revenue growth.
The Roche Group has adopted Personalized Healthcare (PHC) as a core strategy, and the role of companion diagnostics, which identifies patient groups that stand to benefit most from the specific targets of innovative new drugs, has become more important than ever.
As specific biomarker-based therapies continue to increase, demand for pathology and molecular diagnostic testing is rising, and with more therapies gaining reimbursement along with market launch, this is being evaluated as a driver of stable sales growth.
However, the company’s operating profit left something to be desired. While operating profit jumped significantly from KRW 19.1 billion in 2023 to KRW 30.8 billion in 2024, it fell to KRW 18.5 billion last year, returning to the level of two years ago.
Nevertheless, the decline in profitability is interpreted as a result of the base effect from prior price adjustments rather than an actual business slowdown.
Roche Diagnostics Korea has entered into an Advance Pricing Agreement (APA) with its parent company, Roche Diagnostics International Ltd., and has reflected prior-year transfer pricing adjustments through a term test.
In 2024, about KRW 18.3 billion in transfer pricing adjustments covering 2022 through 2024 was deducted from cost of sales, significantly boosting operating profit. In contrast, in 2025, as the term of the APA expired, there was no corresponding reduction in the cost of sales.
As a result, the company’s 2025 operating profit of KRW 18.6 billion is interpreted as a sign of sustained fundamental strength, as it represents profitability generated solely through actual business operations without the benefit of transfer pricing adjustments.
From digital adoption to Alzheimer’s diagnostics… new businesses move into full swing
As Roche Diagnostics Korea continues to strengthen both top-line growth and underlying business quality, the company’s new growth area of building a ‘digital ecosystem’ is also yielding tangible results.
Roche Diagnostics Korea launched a separate Digital Insights business unit and is driving the digital transformation of diagnostic laboratories into Smart Labs, led by NAVIFY, its cloud-based data integration platform.
As a new business area, the digital segment currently accounts for a relatively small portion of total revenue. In particular, the Korean healthcare environment tends to be highly conservative toward adopting new digital systems, with data interoperability relatively low, and institutional support, including reimbursement, is still not fully in place.
Nevertheless, the newly launched Digital Insights Business Unit is reported to have successfully established itself in the market by achieving the key performance indicators (KPIs) set for the Korean market last year.
Muhwan Yun, Head of Digital Insights at Roche Diagnostics Korea, explained, “Healthcare digitalization is now an unavoidable new normal, and we have entered the era of artificial intelligence transformation. NAVIFY is a truly Smart Lab tool that reduces the time healthcare professionals spend on administrative tasks, allowing them to focus solely on patient care.”
The biggest reason the industry is looking positively at Roche Diagnostics Korea’s future is its ‘Blood diagnostic portfolio for Alzheimer’s,’ for which a new approval is strongly anticipated as early as the end of this year.
Recently, Roche’s blood-based biomarker test for Alzheimer’s disease, ‘Elecsys pTau181,’ which Roche developed jointly with Eli Lilly, achieved full approval from the U.S. Food and Drug Administration (FDA).
This is a groundbreaking innovation capable of detecting the accumulation of amyloid plaques and tau proteins at an early stage using just a single drop of blood, without the need for invasive cerebrospinal fluid tests or expensive PET imaging.
If this Alzheimer’s blood diagnostic device secures domestic approval, it is expected to serve as a game-changer that could elevate Roche Diagnostics Korea’s performance next year to a new level in the domestic market, where demand for early dementia diagnosis is surging.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.





