- LOGIN
- MemberShip
- 2026-06-08 05:11:54
- Product
- DP·gov't "INN prescriptions for supply shortage drugs"
- by Kim JiEun Oct 02, 2025 06:11am
- A public forum was held in the National Assembly to discuss the International Nonproprietary Name (INN) prescriptions. Civic groups, pharmacists, and healthcare experts have unanimously agreed that the government should consider implementing the system if it enhances patient choice and reduces national spending. The policy debate, titled 'Introduction of a Korean Model of International Nonproprietary Name (INN) prescriptions to Expand the Public's Choice of Dispensed Medicines,' took place at the National Assembly on September 30. The event was hosted by Democratic Party of Korea Representatives In-soon Nam, Young-seok Seo, Yoon Kim, and Jong-tae Jang, as well as Cho Kuk Innovation Party Representative Sun-min Kim. The Korean Pharmaceutical Association and the Research Institute for Pharmaceutical Policy also hosted it. The panelists agreed that the INN prescriptions should not be viewed merely as a dispute over vested interests between doctors and pharmacists. The priority, they stressed, should be identifying the system that best benefits patients, who are insurance subscribers, by increasing their right to choose medication, and the government should design the system accordingly. The governing parties and the government, who are the proponents of the system's introduction, stated that they agree on the necessity of the system for drugs with unstable supply and are currently pursuing it. However, they presented the prerequisite that public consensus on the need for the system must be formed first. Taek-woo Kim, President of the Korean Medical Association, conducted a solo demonstration directly outside the National Assembly gate to protest the introduction of INN prescriptions At the same time, the debate was being held inside the National Assembly, Taek-woo Kim, President of the Korean Medical Association, conducted a solo demonstration directly outside the National Assembly gate to protest the introduction of INN prescriptions, drawing attention. "Will Enhance Patient Choice and Fiscal Savings…The Best System Helps Patients" Sung-min Park, an associate professor at Seoul National UniversityIt was emphasized at the forum that the need to discuss INN prescriptions from the perspective of patient safety and the expansion of patient choice. Sung-min Park, an associate professor at Seoul National University's Graduate School of Public Health, said, "Brand-based drug labeling increases the risk of confusion and errors during prescribing, dispensing, and administration." He added, "There is a risk of prescribing and dispensing mistakes due to confusion with similar names. Patients also face a higher risk of mistakenly taking the wrong drug." Professor Park said, "When patients with multiple illnesses receive prescriptions from various medical institutions, there is a high possibility of being prescribed the same drug ingredient redundantly," and added, "If patients are unaware of the ingredients, they are more likely to overdose, and discarded unused drugs lead to unnecessary expenditure of National Health Insurance funds." Sun-Young Oh, Director of Policy at the Korean Health and Medical WorkersSun-Young Oh, Director of Policy at the Korean Health and Medical Workers' Union, emphasized, "The expected effect of introducing INN prescriptions is granting patients, as subscribers to the National Health Insurance, the right to choose their medication." She asserted, "Patients lack specialized knowledge about medicine compared to doctors and pharmacists. Creating an environment where patients can make choices must be prioritized. Furthermore, the system must be premised on the therapeutic equivalence of generics so that patients are not anxious about their choices." Oh also said, "Encouraging patient participation in and choice over the medication they take is the desirable direction for this system," adding, "It also has the advantage of reducing dependence on brand names, which is currently very high." "Not a dispute over interests between doctors and pharmacists… Key to Completing Separation of Prescribing and Dispensing" Kwang-min Lee, Vice President of the Korean Pharmaceutical Association, argued that the current brand-name prescribing practice prevents the fundamental intent of the Separation of Prescribing and Dispensing System from being realized. Kwang-min Lee, Vice President of the Korean Pharmaceutical AssociationLee stated that despite the government implementing policies such as deleting the prescription fee item from doctors' service fees and using a market-based actual transaction price reimbursement system for reimbursable drugs, these policies are rendered ineffective by brand-name prescribing. Furthermore, Lee also pointed out that brand-name prescribing directly influences prescribing behavior and healthcare utilization, citing the tendency for self-injectable drugs, such as obesity injections, to be dispensed directly by hospitals. Lee directly criticized the link between brand-name prescribing and illegal business practices by pharmaceutical companies, wholesalers, and CSOs, arguing that it leads to the misuse and abuse of medications. Lee said, "If INN prescriptions become implemented, patients will be able to conveniently receive dispensing services at pharmacies near their homes or workplaces, not just those near medical institutions." He added, "Even patients using multiple institutions for multiple illnesses can receive unified dispensing and medication guidance services from their regular pharmacy, rather than relying on pharmacies near each institution. This will also have the effect of reducing unnecessary drug use." Nam Eun-kyung Lee added, "In Korea, where alarms are sounding over the worsening financial health of the National Health Insurance, a system is needed to promote the use of cost-effective medicines during the prescribing and dispensing stages," and emphasized, "Generic prescribing should not be dismissed as merely a dispute over pharmacists' and doctors' interests; it must be designed with the public at the center to ensure the stability and sustainability of the domestic public health finances." A criticism was also raised that the government needs to take a clear stance on the doctors' argument against INN prescriptions that "even drugs with the same ingredients may differ in efficacy depending on the formulation (release mechanism, dosage form, excipients, etc.)." Nam Eun-kyung pointed out, "The government must clearly draw the line on the controversy over whether generics are different drugs or identical ones. By not formalizing its position, the government is causing unnecessary social conflict." Nam added, "Provided that safety and efficacy are not compromised, the public will agree to a policy that can save NHI funds. I understand that both the Democratic Party and the government are willing to put INN prescriptions on the public agenda. I hope a forum for public discussion involving doctors, pharmacists, and the public can be arranged." "INN prescriptions limited to supply shortage drugs still under discussion"…what do the governing party and government plan? What are the opinions of the National Assembly and the government regarding the highly debated INN prescriptions? Rep. Won-joon Cho, Senior Expert Member for Health and Welfare at the Democratic Party of KoreaThe ruling Democratic Party of Korea and the administrative ministry, the Ministry of Health and Welfare, both agreed on the necessity of introducing the system limited to supply shortage drugs. However, they drew a clear line, stating that widespread adoption of the system would require prior public consensus. Rep. Won-joon Cho, Senior Expert Member for Health and Welfare at the Democratic Party of Korea, explained, "The Tylenol shortage was one of the three major crises after COVID-19. Even in the National Assembly, there was an internal discussion about the need for INN prescriptions for a certain period, given the special circumstances of the pandemic, but it was not pursued," and added, "At that time, the burden of opposition from the medical community could not be overcome. It is from this perspective that a limited INN prescriptions was included in the Democratic Party's last presidential election pledge." Rep. Cho said, "Bills related to INN prescriptions are being introduced, and the National Assembly has plans to discuss institutional improvements and legal amendments." He added, "Various policy measures are being proposed to resolve drug supply instability in addition to the limited introduction of INN prescriptions. All these policy measures must be mobilized to resolve the recurring problem of drug supply instability as much as possible." Jun-hyuk Kang, Director of the Pharmaceutical Policy Division at the Ministry of Health and WelfareFurthermore, Cho added, "However, for the system to be introduced, it must be clearly explained how it will help the public." He concluded, "I believe the system's introduction can gain momentum when public consensus on its rationality and sustainability is formed." Jun-hyuk Kang, Director of the Pharmaceutical Policy Division at the Ministry of Health and Welfare, said, "The government is viewing INN prescriptions from the perspective of the public's drug accessibility." He added, "While Korea has many pharmacies per capita, some administrative districts lack pharmacies, and the issue of drug accessibility has been raised due to unstable drug supply. INN prescriptions is one of the alternatives being discussed to resolve this." Kang said, "It is undesirable that this issue is being highlighted as a conflict between doctors and pharmacists," and added, "Doctors, pharmacists, and the public must discuss this to find a common ground." He concluded, "The government is preparing measures to simplify alternative dispensing and introduce INN prescriptions limited to supply shortage drugs. It seems necessary to conduct specific research on how to introduce INN prescriptions for supply shortage drugs. We understand that foreign countries encourage this through various methods, not necessarily mandating it. We also intend to review these aspects.
- Product
- ‘Will promote phased implementation of INN prescriptions’
- by Kim JiEun Oct 01, 2025 06:10am
- Research Director Dae-Jin Kim, Korea Institute for Pharmaceutical Policy Affairs A study has shown that introducing an International Nonproprietary Name (INN) prescription system could save up to KRW 9 trillion annually in drug costs and related social expenses. At a policy forum on INN prescriptions held at the National Assembly on the 30th, Dae-jin Kim, Director of the Korea Institute for Pharmaceutical Policy Affairs (KIPPA), gave a presentation on “Plans for Introducing a Korean INN Prescription Model.” The forum was hosted by National Assembly members In-soon Nam, Young-seok Seo, Jong-tae Jang, Yoon Kim (Democratic Party of Korea), and Sun-min Kim (Rebuilding Korea Party), and organized by the Korean Pharmaceutical Association and KIPPA. Director Kim first pointed out that the excessive number of generic drug items in the domestic pharmaceutical market is increasing management costs and leading to waste in the National Health Insurance finances. As of January this year, 27.7% of all items had 61 or more products containing the same active ingredient. This directly leads to costs associated with stockouts, unused inventory, recalls, and disposal. He also criticized the structure where a non-differentiated generic industry is being sustained by health insurance finances. He further argued that the high proportion of expensive generics limits cost-saving effects, while the fact that most generic products are marketed under brand names reduces patients’ awareness of the active ingredient, creating communication difficulties between patients and healthcare professionals. This, in turn, heightens concerns about patient safety incidents. Criticism was also raised regarding restrictions on patient choice. Director Kim stated that the low generic substitution rate limits patients' opportunities to choose among medicines with the same ingredients and equivalent effects. Director Kim also presented the results of a survey on public awareness of INN prescribing. In this survey of 3,000 adult men and women residing in Korea, 18.3% reported experiencing drug shortages in the past year. Furthermore, in a survey on acceptance of the INN prescription system, 83.8% of respondents gave positive responses. Based on this domestic pharmaceutical situation and public perception, Director Kim proposed a Korean-style INN prescription model. He explained that its fundamental purpose is ‘to reduce national medical expenses and health insurance premiums while strengthening patient safety and rights’. Director Kim clarified that the definition of INN prescribing in this study refers to prescriptions that list only the ‘active ingredient code + INN + dosage form + strength’ without mentioning brand names. The application targets will be implemented in phases: Phase 1 covers ▲ingredient groups with unstable supply ▲ingredient groups frequently substituted during dispensing ▲the top 100 ingredient groups by claim volume. Phase 2 expands to 5 major efficacy groups (gastrointestinal, diabetes, hypertension, hyperlipidemia drugs, antibiotics, etc.), and Phase 3 extends to all pharmaceuticals. Prerequisites for the system were also outlined: ▲ Reducing domestic drug prices for identical ingredient groups to below the average price of overseas A8 countries ▲ Operating a National Health Insurance Service-recommended drug list and introducing differential co-insurance policies ▲ Improving the coninsurance system. Based on the research findings, Director Kim estimated the maximum annual economic effect of introducing this INN prescription system model at KRW 9.3641 trillion. This figure combines savings of KRW 7.9 trillion in drug costs and KRW 1.4741 trillion in reduced social costs. Director Kim stated, “It is necessary to prioritize applying this system to drug groups with high social demand and cost-effectiveness, then gradually expand the system thereafter. Above all, ensuring patients' right to know and their choice of medication is crucial. Measures to enhance patient access to information, such as utilizing mobile applications or electronic medication guides, can be considered.” He continued, “If fiscal efficiency measures like drug price reductions, differential copayments, and promoting generic competition are implemented alongside the system, they can contribute to reducing national health insurance expenditures. The INN prescription system, implemented in a Korean-style format, will be the core alternative. It strengthens patient safety and rights, reduces national medical expenses and health insurance premiums, and simultaneously addresses chronic issues in the domestic pharmaceutical market, such as the use of high-priced generics and excessive product variety.” Meanwhile, before the forum, the hosting lawmakers In-soon Nam, Young-seok Seo, and Jong-tae Jang each made remarks on the purpose and necessity of introducing the system. Rep. In-soon Nam said, “Public interest in INN prescriptions has grown since the COVID-19 pandemic prompted drug supply instability. I believe the introduction of INN prescriptions can be discussed not only as an alternative at the production and distribution stages related to supply instability issues, but also as an alternative at the prescription stage. Rep. Young-seok Seo said, “We must strive to ensure the system is implemented, considering whether it is necessary for the public and whether it should be introduced to improve the healthcare system. Given the current state of the global pharmaceutical market, supply instability is likely to worsen in the future. INN prescribing is expected to become a tool ensuring a stable drug supply and delivery to patients. I hope the pharmacy community can effectively persuade the public of this.” Korean Pharmaceutical Association President Young-hee Kwon emphasized, “INN prescriptions allow patients to know the ingredients of their medications and make rational choices, thus ensuring their right to choose and realizing the right to health. At the national level, it is also an alternative to reduce health insurance expenditure, secure sustainability, and expand coverage. From the perspective of public health and socioeconomics, it is an essential system demanded by the times.”
- Product
- Generic substitutions and INN-based prescriptions on track
- by Kim JiEun Sep 04, 2025 06:12am
- The prolonged instability in drug supply has fueled momentum in the pharmacy community, with both streamlined generic substitutions and limited mandatory international nonproprietary name prescriptions gaining traction in the National Assembly. On September 2, Democratic Party of Korea lawmaker Jong-Tae Jang introduced amendments to the Medical Service Act and the Pharmaceutical Affairs Act. The proposed bill would create a legal definition for supply-unstable medicines and mandate ingredient name-based prescriptions for such drugs on a limited basis. This follows the passage of the so-called “Generic Substitution Activation Act,” recently approved by the National Assembly’s Health and Welfare Committee, adding yet another long-pursued initiative of the pharmacy sector to the legislative agenda. The shift began with revisions to the enforcement regulations of the Pharmaceutical Affairs Act, which simplified the notification process for generic substitutions. Despite strong opposition from physicians, the Ministry of Health and Welfare pushed through the revision, allowing the HIRA (Health Insurance Review and Assessment Service) online portal as one of the means for post-notifications. Even with doctors voicing concerns of “infringement of prescribing rights,” the government and the National Assembly have continued to push for regulatory revisions and legislation due to the persistent issue of drug shortages. The issue, which first emerged during the early COVID-19 period, have remained unresolved even 5 years after the pandemic subsided. Critics argue the government has failed to present a clear solution. As a result, consensus between the government and the National Assembly on the need to address drug shortages has directly driven attempts at systemic reform. During the last presidential election, the Democratic Party of Korea pledged to implement limited INN-based prescriptions for essential medicines as part of its project to secure a stable drug supply. This confirmed the Lee Jae Myung administration’s commitment to addressing the shortage issue. In its May policy platform, the Democratic Party of Korea included “promoting generic substitutions and limited ingredient-name prescriptions for essential drugs with unstable supply” under its measures to ensure stable access to essential medicines. The pharmacist community believes the streamlining generic substitution law will pass smoothly, given that regulatory changes have already been enacted. However, they remain cautious on mandatory INN-based prescriptions, anticipating fierce resistance from physicians. The Korean Medical Association is likely to intensify its opposition and pressure the government. A pharmacy association representative commented, “We welcome the fact that bills addressing drug shortages are being introduced and advanced with a sense of urgency. For years, responses to shortages have been nothing more than post-hoc measures. Now is the time for preventive and systemic solutions. Although past attempts at generic substitution and INN-based prescriptions repeatedly collapsed due to opposition from the medical community, we are hopeful that with shortages now recognized as a societal issue, this time will be different.”
- Product
- Mounjaro arrives at pharmacies..₩310,000–350,000
- by Kang, Hye-Kyung Aug 21, 2025 06:07am
- Mounjaro (tirzepatide), which had been attracting considerable attention even before its arrival in Korea, has now been made available to pharmacies. With prescriptions also beginning on the 20th, pharmacies are bustling with activity. According to local pharmacies, two dosage forms, 2.5mg and 5mg, have begun arriving at pharmacies through wholesalers. Mounjaro Prefilled Pen 2.5mg, 5mg stocked at pharmacies A local pharmacist stated, “The 2.5mg and 5mg doses arrived this morning. Although no prescriptions have been made yet, we are receiving calls asking if the product has arrived. It seems that consumer interest is quite high.” The two main concerns for pharmacies are pricing and medication guidance. Mounjaro is priced in the ₩300,000 range, with the price lowest in Jongno at ₩290,000 ◆Prices vary between pharmacies, with the lowest price at KRW 290,000 = Since Mounjaro is a non-reimbursed drug, the price varies even between pharmacies. For this reason, pharmacies are struggling to set the initial price. According to telemedicine platforms, prices for 2.5mg in-hospital prescriptions at clinics range from KRW 320,000 to KRW 350,000 Pharmacies are pricing the medication between KRW 310,000 to KRW 350,000. While the number of pharmacies stocking Mounjaro is still limited, prices seem to be forming in the early to mid three hundred thousand won range. However, in Jongno, the price is set at KRW 290,000, which is the lowest price. ◆Medication guidance for Mounjaro?= Mounjaro comes in a package containing four single-use pre-filled pens. The outer case states, “Mounjaro is used once a week. Read the instructions carefully before using the medication and keep the instructions with the medication.” Mounjaro was approved as ▲an adjunct to diet and exercise therapy for improving blood glucose control in adult patients with type 2 diabetes; ▲an adjunct to low-calorie diet and exercise therapy for chronic weight management in adult patients; and ▲as an adjunct to a low-calorie diet and exercise therapy for the treatment of moderate-to-severe obstructive sleep apnea (OSA) in obese adult patients with an initial body mass index (BMI) of 30 kg/m2 or higher. When administered for weight management ▲Obese patients with an initial body mass index (BMI) of 30 kg/m2 or higher ▲Overweight patients with an initial body mass index (BMI) of 27 kg/m² or higher but less than 30 kg/m² and one or more weight-related comorbidities such as hypertension, dyslipidemia, type 2 diabetes, obstructive sleep apnea, or cardiovascular disease, may use the treatment. The recommended starting dose is 2.5 mg subcutaneous injection once weekly, increased to 5 mg subcutaneous injection once weekly after 4 weeks, and maintained at that dose. The maximum dose is 15 mg subcutaneous injection once weekly. If a dose is missed, administer the missed dose as soon as possible within 4 days (96 hours) of the missed dose. If more than 4 days have passed, skip the missed dose and administer the next dose on the scheduled day. Mounjaro can be administered at any time of the day, regardless of meals, and should be administered as a subcutaneous injection in the abdomen, thigh, or upper arm. It is recommended to rotate (alternate) the injection site with each dose. The recommended that the injections are refrigerated at 2–8°C. If necessary, each single-use pen can be stored at a temperature not exceeding 30°C for up to 21 days without refrigeration. Do not freeze. If frozen, the medication should not be used and should be stored in its original container to protect from light. The most commonly reported adverse reactions include gastrointestinal disorders such as nausea (very common), diarrhea (very common), and vomiting (common). Generally, the severity of these reactions was mild or moderate, and occurred more frequently during dose escalation, and decreased over time. Meanwhile, Lilly Korea announced that it plans to first release 2.5 mg and 5 mg doses in Korea, but will also supply higher doses of 7.5 mg, 10 mg, 12.5 mg, and 15 mg in accordance with patient demand. Meanwhile, the Ministry of Food and Drug Safety sent a notice to medical organizations and other relevant parties to prevent misuse of the drug following the release of Mounjaro. The MFDS emphasized, “Please prescribe and dispense the drug appropriately in accordance with the approved efficacy, dosage, and precautions for use. To prevent side effects and misuse, please provide accurate information about the drug (efficacy, dosage, and precautions for use) to the patients and instruct them on how to take the drug.”
- Product
- Mounjaro set to hit shelves next week... pricing a dilemma
- by Jung, Heung-Jun Aug 13, 2025 06:07am
- With the imminent arrival of the diabetes and obesity treatment Mounjaro (tirzepatide) in Korea, pharmacies are struggling to set an appropriate price for the item. Due to the nature of high-priced non-reimbursable drugs, there are regional price differences, and price competition is expected in the future, so pharmacies are showing a cautious stance on their initial sales price. Recently, some distributors notified pharmacies of Mounjaro ‘s supply schedule for the 20th and surveyed the pharmacies’ willingness to handle the product to anticipate demand. Additionally, telemedicine platforms are guiding affiliated clinics and pharmacies to input their planned selling prices. Like Wegovy, Mounjaro’s supply price was disclosed before its domestic launch. However, unlike Wegovy, the supply price varies by dosage. While there may be minor differences depending on the distributor, the prices are currently set at KRW 278,000 for 2.5mg and KRW 369,300 for 5mg. The prices for the upcoming 7.5mg and 10mg doses are reported to be KRW 521,300. Pharmacists are expected to monitor the prices set by nearby clinics and pharmacies before determining their own sales prices. Pharmacist A from Seoul stated, “Based on the 2.5mg dosage, the price is expected to range from the mid-300,000 KRW to the 400,000 KRW bracket. Although distribution is scheduled for next week, we have not yet decided whether to carry the product, as we are unsure whether hospitals will issue outpatient prescriptions.” Pharmacist A added, “Although both products are used for obesity treatment, their mechanisms of action are different, so it is not appropriate to simply compare their dosage and price.” In the case of Wegovy, the selling price, which was in the range of KRW 500,000 to KRW 600,000 when it was first launched in Korea, gradually decreased, and the price was adjusted. In the case of obesity treatments, price information is quickly shared through telemedicine platforms and communities, so even among non-reimbursable drugs, the sales price can fluctuate greatly. Some pharmacists are hesitant to handle the drug because the supply price is known and there is a risk that it could become dead stock. Pharmacist B from Seoul stated, “It has a competitor, so it's difficult to predict how much demand there will be for Mounjaro, so we're waiting to see how demand develops. We also have remaining stock of Saxenda and Wegovy. Price competition is intense, so expanding our inventory can be a burden.”
- Product
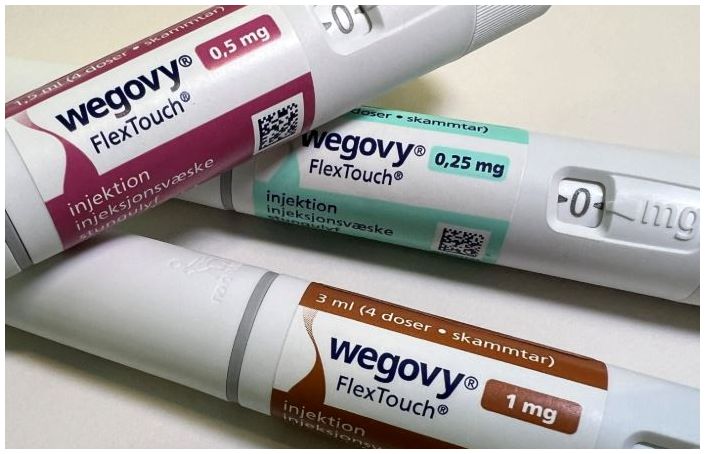
- Wegovy supply price cut by 40%... to counter Mounjaro
- by Kang, Hye-Kyung Aug 13, 2025 06:06am
- The supply price for Wegovy will be reduced by approximately 40% starting on the 14th. Refund measures are also expected to be implemented for pharmacies with inventory. Novo Nordisk notified pharmacies and other relevant parties of the changes through Julic and other channels. According to Dailypharm's coverage, the prices for four dosage forms—0.25mg, 0.5mg, 1.0mg, and 1.7mg—will be reduced, excluding the highest dosage of 2.4mg. The previous single price system will be replaced with a tiered pricing system based on dosage, with the 0.25mg starting dosage set to receive a price cut of around 40%. Once the price reductions are implemented, the purchase prices for pharmacies and clinics will be around KRW 220,000. However, to prevent confusion in medical and pharmacy settings, the specific price reduction rates will not be disclosed. Refunds for existing inventory held by pharmacies will also be processed according to established procedures. Details regarding refund procedures and amounts per pharmacy will be communicated sequentially through Zuellig Pharma sales representatives and the suppliers. Some have speculated that the price reduction policy for Wegovy may be in response to the upcoming launch of Mounjaro this month. Mounjaro, which is expected to be launched around mid-month, will apply tiered pricing across six dosage forms, with the starting dosage being approximately KRW 100,000 cheaper than Wegovy. However, Novo Nordisk stated that it had been reviewing a tiered pricing policy by dosage for a long time after the launch and had decided to switch from a uniform single price system to a tiered pricing system by dosage. A Novo Nordisk representative said, “We decided to lower the shipping price and notify customers to minimize confusion among distributors, pharmacies, and patients who have been prescribed the drug.” They added, “In supplying innovative treatments like Wegovy, we prioritize treatment continuity and accessibility for Korean obesity patients as our top principle, and this principle also applies when determining the shipping price of the treatment. We will continue to strive to improve the obesity treatment environment in Korea through a patient-focused approach.”
- Product
- Platforms promote Wegovy's lowest price is ₩390,000
- by Kang, Hye-Kyung Jul 30, 2025 06:10am
- Non-face-to-face treatment platforms are facing backlash from pharmacies after launching activities promoting the lowest prices for non-reimbursable drugs. Doctornow, a leading non-face-to-face medical consultation platform, is being criticized for encouraging price competition for non-reimbursable drugs and causing confusion among consumers. A local pharmacist stated, “Recently, Doctornw has posted promotional content on its social media accounts, such as ‘How to buy diet injections for KRW 200,000 less,’ 'The real way to buy semaglutide diet injections for KRW 390,000,‘ and 'Hair loss medication consultation fee + medication cost: KRW 9,060.’ I believe this is likely to cause distrust and confusion among consumers toward pharmacies.” For example, in a video titled ‘How to buy diet injections for KRW 200,000 less,’ it says, 'Wow! I used to buy weight-loss injections for KRW 600,000, but I was paying KRW 200,000 more. If you don't know the latest way to buy diet injections cheaply, you're losing out 100%. If you know this method, you can get a prescription for KRW 200,000 less, at an unbelievable price!“ The video states that all you need is to access Doctornow. The video explains that you can find the lowest price at a pharmacy near you by clicking ”Find a Pharmacy,“ then clicking ”Get a Prescription" to select a nearby and affordable doctor, going to the hospital for a consultation, and then getting the injection at the lowest-priced pharmacy. However, the video has been criticized for causing consumer distrust and confusion about pharmacies. This pharmacist said, "Doctornow claims that the real price for semaglutide diet injections is KRW 390,000. It suggests that pharmacies are taking a middleman margin. However, the purchase price of the semaglutide diet injection, Wegovy, far exceeds the 390,000 won suggested by Doctornow.” When credit card transaction fees are added to the purchase price, it is practically impossible to meet the criteria for ”buying cheaply and well" suggested by Doctornow. When Dailpharm searched for the lowest price of Wegovy on Doctornow, the price varied depending on the region, but was usually over KRW 430,000. It was found that there are pharmacies in the Gyeonggi area that sell the drug for KRW 394,000 and KRW 395,000, but in the Gyeongsangbuk-do, Chungcheongbuk-do, and Gyeongsangnam-do areas, the price of the non-reimbursable injectable was set around KRW 430,000 to KRW 450,000. Doctornow also presented KRW 9,060 as the lowest hair loss medication consultation fees available. Another pharmacist said, “There are differences between pharmacies when it comes to the price of non-reimbursable drugs. However, I think it is problematic under the Pharmaceutical Affairs Act for non-face-to-face consultation platforms to arbitrarily set the lowest price and misrepresent it as the ‘standard’. Shouldn't sanctions be imposed on the platform for such excessive promotional activities?” Pharmacists also voiced concerns about the institutionalization of telemedicine, as the majority of non-face-to-face prescriptions made are for non-reimbursable drugs for cosmetic purposes. According to a survey conducted by the Guro District Pharmaceutical Association and the Jungnang, Gwangjin, and Gangdong District Pharmaceutical Associations among their members, 46% of pharmacists stated that they had accepted non-face-to-face prescriptions in the past three months. When asked about the proportion of non-face-to-face prescriptions for cosmetic purposes, such as hair loss, weight loss, and eye drops, among the prescriptions received, over 80% of respondents answered that they accounted for the majority of non-face-to-face prescriptions. The associations stated that the majority of pharmacists who accepted non-face-to-face prescriptions in the last three months responded that they were for cosmetic purposes. We are concerned about the reality that non-face-to-face medical consultations are being used as a means to purchase non-reimbursable drugs for cosmetic purposes, such as hair loss, diet, and eye drops, which deviates from the original purpose. We believe that non-face-to-face prescriptions for cosmetic purposes should also be excluded or restricted by the system, similar to psychotropic drugs.” Meanwhile, during her confirmation hearing as nominee for Minister of Health and Welfare, Eun-kyung Jeong stated, “The institutionalization of telemedicine should be aimed at ensuring the stability of medical care and improving primary care, rather than expanding platform profits. During discussions with the National Assembly, appropriate regulatory measures should be discussed to address concerns that platforms will effectively take over clinics and pharmacies or promote profit expansion.”
- Product
- KPDS ‘Gov’t action needed to counter price hike pressure’
- by Jul 08, 2025 06:35am
- The Korean Pharmacists for Democratic Society (President: Kyung-Lim Jeon, KPDS) urged the government to take decisive action against Trump and multinational pharmaceutical companies' pressure to raise drug prices. In a statement released on the 4th, the KPDS stated: “Trump signed an executive order on May 12 demanding a reduction in drug prices within the United States, claiming that ‘the United States accounts for less than 5% of the world's population but generates three-quarters of the global pharmaceutical industry's profits.” This is intended to support U.S. pharmaceutical companies by expanding their access to foreign markets, enabling them to lower drug prices within the United States while increasing profits in other countries.” However, ironically, the U.S. pharmaceutical industry has expressed opposition to President Trump's “most-favored-nation (MFN)” policy for the U.S., citing concerns that its double standard could lead to reduced investment and lower new drug development. The KPDS also pointed out that the U.S. pharmaceutical industry has long opposed policies that view health as a basic social security for citizens and seek to establish a foundation for the health of individuals and society as a whole through a national health insurance system and drug price control policies. Rather, the companies demanded a society where pharmaceutical companies can sell drugs at monopolistic prices that guarantee maximum profits, packaging the anti-market system of patent monopolies as “fair trade.” They have also sought to extend patent periods or operate various monopolistic systems, such as through the new drug product exclusivity system, and have exploited loopholes in the patent system. The KPDS stated, “US President Trump's executive order to lower drug prices acted as a catalyst, turning into a storm of drug price hikes for South Korea and many other countries. The Korean government must recognize this change in the trend and respond proactively.” The KPDS proposed 3 countermeasures. The first is to improve the drug pricing system being promoted by the government to reflect the innovative value of treatments. The currently promoted system is not a system for patients, but rather a capitulation to the demands of multinational pharmaceutical companies to raise drug prices. Efforts to increase bargaining power to counter the rapidly rising prices of new drugs should be prioritized. They also urged the government to seek international cooperation to counter the pharmaceutical companies' unreasonable demands for price hikes. KPDS stated, "The Korean pharmaceutical market accounts for only about 1% of the global market. The Korean government's bargaining power is so weak that multinational pharmaceutical companies can easily abandon the Korean market if they so desire. Recently, Europe has been countering the high prices of new drugs by forming alliances with neighboring countries to negotiate drug prices with companies. Korea must also build alliances with neighboring countries in various ways to strengthen its bargaining power with multinational pharmaceutical companies in drug price negotiations." In addition, they proposed the need to establish policies that would substantially reduce drug costs. The fundamental reason for the high price of drugs in the United States is that the government has excessively overestimated the value of drugs under the pretext of fostering domestic pharmaceutical companies and has made no effort to control drug prices. KPDS added, “The reason why rebates by Korean pharmaceutical companies continue is that the drug pricing policies that are lenient to domestic companies provide incentives for companies to sell drugs even if they have to pay extra to doctors. Amid rapid aging, South Korea's drug pricing policy needs to be readjusted, and it is urgent to establish new policies to resolve drug pricing issues, such as the 2006 ‘Drug Price Rationalization Plan.’” "From the perspective of reasonable corporate profits, drug prices in South Korea are not low. There is a growing trend of high-priced new drugs receiving economic evaluation waivers during review, and generic drug prices are among the highest in the world. From the U.S. perspective, demanding that all countries pay high drug prices on the grounds that South Korea's drug prices are low is not justifiable. Instead, the U.S. should review and reform its overly protective pharmaceutical patent monopoly system.
- Product
- 'Govn’t must take strong action against pharma rebates'
- by Kang, Hye-Kyung Jun 26, 2025 06:08am
- The Korean Pharmacists for Democratic Society (President Gyeong-rim Jeon, KPDS) has urged the government to take strong action against pharmaceutical company rebates. KPDS issued a statement regarding rebates made by a major domestic pharmaceutical company that JTBC reported. In the statement, KPDS said, “This is not an individual incident, but clear evidence of widespread illegal rebates rampant throughout the pharmaceutical industry. “Medicines prescribed by doctors are not ordinary commodities, but special goods that directly affect patients' lives and health. The Pharmaceutical Affairs Act and Medical Service Act fundamentally prohibit pharmaceutical companies from providing economic benefits to medical professionals.” However, according to the JTBC report, the pharmaceutical company in question blatantly violated legal restrictions. The KPDS criticized, “What is even more outrageous is the government authorities' incompetent response to these clear illegal acts. Using a shortage of personnel as an excuse to avoid addressing the issue raises suspicions that there may be an intent to cover up the incident.” They went on to say, “The explanation that this was a legitimate new drug promotion activity is a clear lie, and the government must immediately implement strong measures to eradicate illegal rebates.” In other words, the KPDS believes the MOHW and the police must reinvestigate the case and severely punish those involved. The KPDS proposed the following measures: ▲reinvestigation through the formation of a special investigation team; ▲a comprehensive investigation by the MOHW into the provision of financial benefits such as support for academic conferences, lecture fees, and consulting fees; and ▲strong administrative measures such as drug price reductions when illegal rebates are detected. They emphasized, “We can no longer tolerate the illegal rebate cartel that threatens the health insurance budget and public health. The government must use this incident as an opportunity to demonstrate its firm commitment to eradicating illegal rebates in the pharmaceutical industry.”
- Product
- Patient organization proposes 6 policies as election pledges
- by Kang, Hye-Kyung May 14, 2025 06:10am
- A patient group has proposed six policies, including a “rapid reimbursement for new drugs and post-marketing adjustment system.” The Korea Alliance of Patients Organization submitted a 'Statement regarding Six Major Patient Policies' to political parties on the first day of the official campaign for the 21st presidential election on the 12th, urging them to reflect the proposals in their election pledges. The six proposed policies are: ▲Enactment of a Framework Act for patients to promote their treatment journey and protect their rights ▲Establishment of a Patient Policy Division within the Ministry of Health and Welfare ▲Establishment of an integrated support platform for patients fighting illness ▲Introduction of a system for rapid reimbursement and post-marketing adjustments for new drugs directly related to life ▲Institutionalization of a nursing care system ▲Promotion of a national responsibility system for essential organ transplant costs. KAPO stated, “The medical crisis that has lasted over a year and three months, triggered by the government's policy to increase medical school quotas. The resulting conflict with the medical community has caused immense suffering and harm to us patients. The conclusion of this medical-political conflict has left only a visual lesson: the government cannot defeat doctors by using patients' lives as a tool. Now, we patients and patient groups demand a government that will protect patients' lives and rights no matter what medical crisis arises.” They called for a government that will not hesitate to push forward policies and legislation for patients' treatment and rights even in the face of opposition from the medical community, and for a presidential candidate who will create a “patient-centered healthcare environment” where patients are no longer objects or targets but as “subjects” that actively participate for their treatment and rights. They stated, “We have conveyed patients' voices to presidential candidates as 6 major patient policies and hope they will be adopted as campaign pledges.” KAPO was established on February 4, 2010, under the slogan “Listen to Patients,” and currently includes the Korean Leukemia Patients Organization, the Korean GIST Patients Association, the Korea Kidney Cancer Association, All. Can Korea, the Korea Congential Heart Disease Patient Group, the Korea Psoriasis Association, the Korean Society of Type 1 Diabetes, the Korea Neuroendocrine Tumor Society, the Korean PROS Patient Organization, and the Korean Parkinson's Hope Association, among others, with over 92,000 patient participants.