- LOGIN
- MemberShip
- 2026-04-24 02:52:23
- Company
- AZ launches triple-combination COPD inhaler Breztri in KOR
- by Son, Hyung Min Apr 03, 2026 08:02am
- Breztri AerosphereAstraZeneca Korea (Country President: Eldana Sauran) announced the launch of ‘Breztri Aerosphere (budesonide, glycopyrronium, formoterol),’ as a maintenance therapy for moderate-to-severe chronic obstructive pulmonary disease (COPD).Breztri Aerosphere is a single-inhaler triple-combination therapy that combines an inhaled corticosteroid (ICS), a long-acting beta-2 agonist (LABA), and a long-acting muscarinic antagonist (LAMA) in a single inhaler. It is indicated as a maintenance treatment for adult COPD patients to control symptoms and reduce exacerbations, and is administered twice daily.The clinical efficacy and safety profile of Breztri Aerosphere have been established through global Phase III trials, including ETHOS and KRONOS.The ETHOS (The Efficacy and Safety of Triple Therapy in Obstructive Lung Disease) study was a 52-week, multicenter, randomized, double-blind Phase III trial involving 8,588 patients aged 40–80 with moderate-to-very severe COPD.Results showed that Breztri Aerosphere reduced the annual rate of moderate or severe COPD exacerbations by approximately 24% compared with LAMA/LABA dual therapy, and by about 13% compared with ICS/LABA, demonstrating statistically significant results.A post-hoc analysis of ETHOS also demonstrated a significant reduction in all-cause mortality in the Breztri treatment group compared with LAMA/LABA.In another pivotal trial, KRONOS, Breztri Aerosphere demonstrated improvements in lung function.The 24-week global Phase III study enrolled 1,902 patients with moderate-to-very severe COPD. At Week 24, Breztri improved lung function by 22 mL compared with LAMA/LABA and by 74 mL compared with ICS/LABA (BFF MDI).COPD is a representative chronic respiratory disease caused by abnormalities in the airways and alveoli, such as bronchitis, bronchiolitis, and emphysema. It is characterized by chronic respiratory symptoms such as shortness of breath and coughing, and is a heterogeneous disease involving persistent and progressive airway obstruction. Compared to the general population, patients with COPD tend to develop various comorbidities at an earlier age. Factors such as smoking, aging, and chronic conditions associated with the disease itself, which include cardiovascular disease, musculoskeletal disorders, and diabetes, further increase disease burden.The 2026 Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines recommend triple therapy with ICS, LAMA, and LABA in patients receiving ICS+LABA who are currently not experiencing an exacerbation but have a high symptom burden, or who are experiencing an exacerbation and have a blood eosinophil count of 100 cells/μL or higher.The guidelines also note that using a single inhaler can improve treatment convenience and adherence compared with multiple inhalers.Ji-young Kim, Executive Vice President of the Respiratory Business Unit at AstraZeneca Korea, stated, “Breztri Aerosphere is a treatment option that has demonstrated reductions in exacerbations and improvements in lung function in global clinical trials, offering a new treatment option for COPD patients in Korea. AstraZeneca Korea will continue to strive to improve the treatment environment and disease management for patients with respiratory diseases.”Breztri Aerosphere is a pMDI (pressurized metered-dose inhaler) designed to uniformly disperse drug particles using Aerosphere delivery technology, ensuring that the medication is delivered throughout both the large and small airways.pMDIs can be used by patients with insufficient inhalation flow rates and offer the advantage of delivering a consistent dose with each actuation. Additionally, as a single inhaler capable of administering three medications simultaneously, it can help improve treatment convenience and medication adherence for patients.
- Company
- "Finished drug firms·API suppliers suffer from high exchange rate"
- by Chon, Seung-Hyun Apr 03, 2026 08:02am
- The pharmaceutical industry is facing significant challenges conducting business amid high exchange rates and the war in the Middle East. As the KRW/USD exchange rate surpasses KRW 1,500, the cost pressure on imported Active Pharmaceutical Ingredients (APIs) is intensifying. Furthermore, with upcoming generic drug price cuts, seeking cheaper imported APIs is becoming increasingly difficult. The high exchange rate and price reductions are also major issues for API manufacturers.According to industry sources on the 2nd, the KRW/USD exchange rate in the Seoul foreign exchange market reached 1,501.5 KRW on the 1st. Compared to KRW 1,352.6 on July 2 of last year, this represents an increase of more than KRW 150 in just eight months.The KRW/USD exchange rate trend (unit: KRW, source: SMB)The exchange rate first surpassed KRW 1,500 on the 24th of last month and briefly stayed in the KRW 1,400 range for two days before surpassing KRW 1,500 again on March 29, continuing to increase. Amid concerns over a prolonged conflict stemming from the high-intensity standoff between the U.S. and Iran, the rate even surged past KRW 1,530 during intraday trading on the 31st, the highest level in 17 years since the global financial crisis in 2009.The decline in the value of the Korean won directly translates to cost-push pressure for pharmaceutical companies. Since these companies are highly dependent on imported APIs, the core raw materials for medicines, the rise in the KRW/USD exchange rate directly increases production costs.In 2024, the self-sufficiency rate for APIs was recorded at 31.4%, calculated using an average exchange rate of KRW 1,367. Self-sufficiency refers to the ratio of domestically produced products within the total market.With 69.6% of APIs used domestically being imported, the reliance on foreign raw materials is absolute. Since US dollars are used even when purchasing APIs from China and India, the largest sources of imports, the impact of the rising exchange rate is unavoidable.Recently, domestic pharmaceutical companies have also been considering switching API suppliers to reduce costs in response to the government's announced price cuts for generic drugs.Under the reformed drug pricing system discussed by the Ministry of Health and Welfare (MOHW) at the Health Insurance Policy Review Committee on the 26th of last month, the price for both off-patent drugs and generics will decrease from 53.55% to 45% of the pre-patent-expiry price of the new drug. Mathematically, this is a 16.0% cut in generic prices.Under the reformed drug pricing system discussed by the Ministry of Health and Welfare (MOHW) at the Health Insurance Policy Review Committee on the 26th of last month, the price for both off-patent drugs and generics will decrease from 53.55% to 45% of the pre-patent-expiry price of the new drug.The price reduction range increases further when "top-tier price requirements," such as conducting bioequivalence (BE) studies and using registered drug substances (DMF), are applied to existing listed generics. Under the reform, the penalty for failing to meet these requirements will expand from 15% to 20%. Since July 2020, requirements have been in place that generics can receive the 53.55% maximum price only if they meet both the direct BE study and DMF criteria. For each unmet requirement, the ceiling price drops by 15%.Applying the new 45% standard and the 20% cut for unmet requirements, generics failing one requirement drop to 36%, and those failing both drop to 28.8%. This means the price for a generic failing one requirement will be 20.9% lower than current levels, while those failing both will see a 25.6% decrease.Due to these price-reduction pressures, pharmaceutical companies are forced to seek cheaper imported products rather than relatively expensive domestic APIs to save costs.The value of API imports from China is already increasing. In 2024, Chinese API imports reached USD 816.32 million, up 110.2% from USD 388.31 million in 2014. In 2014, China was the 6th-largest source of drug imports for Korea, but it rose to 3rd place by 2024.Chinese API Import Amounts by Year (USD 1,000)In 2024, the value of Chinese APIs used in Korea was KRW 1.1159 trillion. Of the KRW 4.4007 trillion in APIs produced in Korea, only KRW 1.43 trillion was used in the domestic market. This indicates that the amount of Korean and Chinese APIs used in the local market is roughly equal. Considering that Chinese APIs are generally cheaper than domestic ones, this suggests that the actual volume of Chinese APIs used by domestic companies overwhelms that of domestic APIs.Under these circumstances, there are concerns that if generic prices drop further, more companies would refrain from using relatively expensive domestic APIs. Both finished drug manufacturers and API suppliers are structured in a way that could lead to simultaneous losses under pressure to reduce drug prices.For API manufacturers, the high exchange rate and price cuts act as major setbacks. Even for domestically produced APIs, starting materials are often imported, so they must worry about rising costs due to exchange rates. As pharmaceutical companies search for cheaper imported alternatives, the concerns of domestic API firms are further compounded.A pharmaceutical industry official stated, "If generic prices fall further, the movement to replace raw materials with cheaper alternatives to save costs will spread. As the dependency on imported APIs increases, domestic API companies find themselves in a position where they must worry about survival."Critics pointed out the ineffectiveness of the government's API preferential pricing policy. The government plans to apply price preferences to listed "essential national medicines" that use domestic APIs. This involves granting a price preference of up to 68% of the pre-patent-expiry price of new drugs for essential medicines made with domestic raw materials.However, pharmaceutical companies complain that the proportion of essential medicines within total drug sales is negligible, and even if prices are raised, there is insufficient incentive to switch to domestic APIs. To receive the preferential designation for domestic raw materials, a company must prove that all raw materials were synthesized at a domestic manufacturing site. Required documents include the ▲API Registration Certificate ▲Common Technical Document (CTD) ▲Manufacturing Instructions and Records.During a parliamentary audit last October, Rep. Baek Jonghean of the People Power Party pointed out, "The fact that not a single pharmaceutical company has applied for the price preference for essential national medicines using domestic raw materials for seven months is proof that the system exists in name only," adding, "Despite industry-wide complaints that the application criteria are too stringent, the MOHW's failure to recognize that the regulations will undermine the policy's ability to foster the domestic API industry."An industry source stated, "Due to the recent aftermath of the war in the Middle East, there are concerns over the supply instability of various raw materials, and with the added burden of costs from the high exchange rate, it is difficult to predict business plans for this year," adding, "In addition to the government's drug price reduction policy, it has become difficult to guarantee the business sustainability of both domestic finished drug and API manufacturers."
- Company
- AstraZeneca Korea appoints Ohad Goldberg as new Country President
- by Son, Hyung Min Apr 03, 2026 08:02am
- Ohad Goldberg, new Country President of AstraZeneca KoreaAstraZeneca Korea announced that it has appointed Ohad Goldberg, Country President of AstraZeneca Israel, as the new Country President of AstraZeneca Korea, effective May 1, 2026.As the new Country President, Goldberg will oversee the company’s business operations in Korea. He plans to expand patient access to AstraZeneca’s medicines and continue to enhance AstraZeneca’s standing as a top-tier partner dedicated to strengthening the domestic life sciences ecosystem for the benefit of Korean patients and society.During his tenure as Country President of AstraZeneca Israel, Goldberg led significant organizational growth and strengthened partnerships across the healthcare ecosystem. With over 20 years of international leadership experience across life sciences, biotech, and ag-tech, he possesses proven capabilities in commercial operations, market access, and external affairs.He has led AstraZeneca Israel’s external partnerships with government, healthcare stakeholders, academia, and innovation platforms. He also served as Chairman of the Board for AION Labs and as a board member of Pharma Israel, representing AstraZeneca in multiple leadership roles.Ohad Goldberg, the new Country Manager of AstraZeneca Korea, said, “It is a great honor to take on this role at a pivotal time for healthcare reform in Korea. Together with our talented team, I look forward to contributing to the advancement of healthcare policy and to creating sustainable, long-term value for Korean patients and society by expanding equitable access to breakthrough medicines.”Goldberg has held senior leadership roles at AstraZeneca, including Global Launch Leader for Respiratory Biologics, Market Access Director, and Respiratory Business Unit Director for Israel, and senior roles in commercial strategy across Europe.
- Company
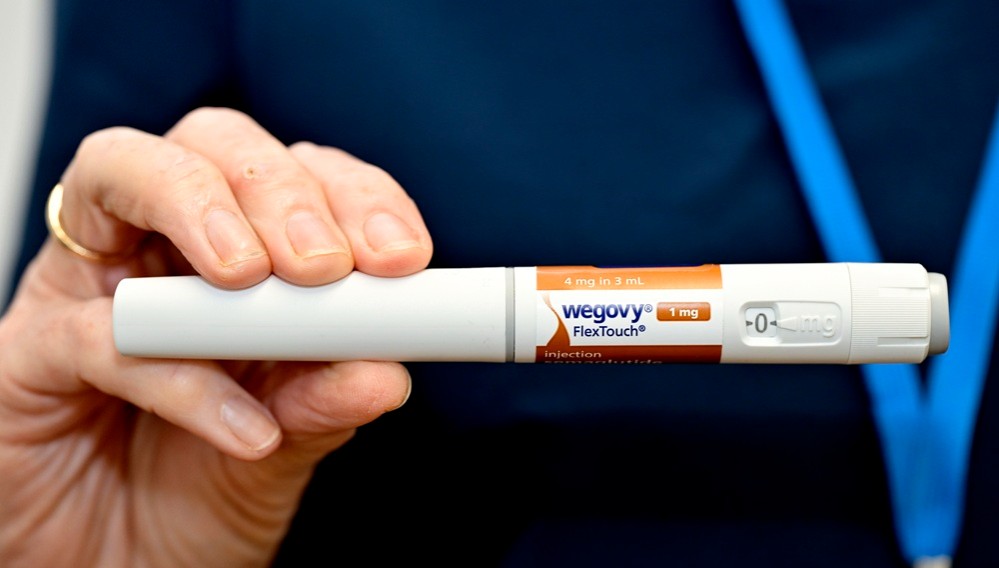
- Novo Nordisk hits record sales in Korea… led by Wegovy
- by Son, Hyung Min Apr 03, 2026 08:02am
- Novo Nordisk achieved record domestic sales, driven by its obesity treatment 'Wegovy.' As obesity treatments become a primary growth driver, the company’s earnings structure is shifting away from its previous focus on diabetes and hemophilia.According to the Financial Supervisory Service on the 3rd, Novo Nordisk’s sales increased from KRW 308.5 billion in 2024 to KRW 613.6 billion last year, up 85.6% year-on-year. Operating profit rose 77.1% over the same period, from KRW 13.7 billion to KRW 24.2 billion.Novo Nordisk’s performance shows a clear distinction before and after the launch of Wegovy (semaglutide).Prior to the arrival of Wegovy, the company maintained steady growth based on insulin products, hemophilia treatments, and the once-daily obesity drug ‘Saxenda (liraglutide),’ although growth rates were relatively limited.However, its performance surged with the launch of Wegovy in Korea in 2024. With the launch of Wegovy, Novo Nordisk recorded KRW 374.7 billion in sales that year, representing a 62.7% increase from the previous year.According to the market research institution IQVIA, Wegovy generated KRW 467 billion in sales last year, accounting for over 70% of total revenue, and established itself as Novo Nordisk’s core revenue source just one year after its launch. This represents a rare case where a single product drives the growth of a local affiliate.Quarterly trends also highlight rapid growth. Wegovy posted KRW 60.3 billion in Q4 2024, and sales rose to KRW 133.8 billion in Q2 2025, surpassing KRW 100 billion in quarterly sales. It recorded KRW 137 billion and KRW 116.7 billion in Q3 and Q4, respectively, quickly dominating the market.This shift has directly translated into actual market demand.Previously, obesity treatment relied mainly on diet and exercise, with limited use of adjunctive drugs. Injectable treatments like Saxenda existed, but the daily dosing burden and adherence issues constrained market expansion.In contrast, Wegovy demonstrated significant weight loss with once-weekly dosing, greatly improving convenience. Clinical results showing over 15% weight reduction served as a catalyst for shifting obesity treatment from a selective management option to an aggressive treatment option.In Korea, demand surged immediately after launch, leading to supply shortages, with clinics and hospitals reporting a spike in prescription inquiries. This is interpreted not as a temporary trend but as a release of latent demand.As a result, inventories increased sharply. Novo Nordisk’s inventory rose from KRW 80.8 billion in 2024 to KRW 348.2 billion last year, a 331% increase.This reflects the company’s supply expansion strategy to meet surging Wegovy demand. As supply shortages persisted, with repeated sell-outs following the initial launch, the company made a proactive effort to secure inventory and improve its distribution capabilities.In particular, since consistent and stable administration is crucial for obesity treatments, securing inventory is evaluated not merely as an increase in costs but as a key operational indicator supporting revenue growth.Beyond GLP-1 to next-generation mechanisms… Novo Nordisk expands its metabolic pipelineObesity drug ‘Wegovy’Novo Nordisk is globally expanding semaglutide’s indications beyond obesity and diabetes into broader metabolic diseases.Semaglutide has demonstrated reductions in major adverse cardiovascular events (MACE), extending its therapeutic scope beyond diabetes and obesity.By accumulating clinical evidence that encompasses not only diabetes patients but also high-risk cardiovascular groups, it has been clearly demonstrated that GLP-1 agonists can contribute to improving long-term outcomes beyond weight loss.Furthermore, with the addition of chronic kidney disease (CKD) indications, the company is increasing its presence in the renal disease sector previously pioneered by SGLT-2 inhibitors.Recently, semaglutide gained accelerated approval for MASH, further expanding into liver disease.Their mechanistic strengths of weight loss, improvement in insulin resistance, and suppression of inflammation have led to reduced hepatic fat accumulation and improved fibrosis, positioning them as a new alternative in the MASH field, where treatment options have previously been limited.In addition, the company is preparing new drugs with novel mechanisms of action. CagriSema is a combination drug containing 2.4 mg of semaglutide, the active ingredient in Wegovy, and 2.4 mg of the long-acting amylin analog cagrilintide. Cagrilintide mimics the action of amylin, a hormone that naturally suppresses appetite, and is being developed as a once-weekly dose due to its longer duration of action compared to existing treatments.Additionally, the triple agonist ‘UBT251 (GLP-1/GIP/GCG), currently being codeveloped with a Chinese partner, is also emerging as a next-generation growth driver. This is a multi-target drug in the same class as Eli Lilly’s retatrutide. Results of a 24-week Phase II clinical trial recently disclosed in China showed that UBT251 demonstrated a maximum weight loss of 19.7%.If these drugs are introduced to the domestic market, Novo Nordisk’s growth trajectory is expected to accelerate further.
- Company
- Imfinzi shifts gastric cancer treatment paradigm
- by Son, Hyung Min Apr 02, 2026 08:46am
- With ‘Imfinzi’ receiving approval as a perioperative treatment for gastric cancer, there are signs that the immunotherapy-plus-chemotherapy strategy, which has long been established as the standard of care overseas, is set to gain full-scale traction in Korea as well.On March 31, AstraZeneca Korea held a press conference at the Four Seasons Hotel in Seoul to share the significance of the expanded indication for Imfinzi (durvalumab) in gastric cancer and its clinical data.On the 23rd, Imfinzi was approved as a perioperative treatment for patients with resectable gastric or gastroesophageal junction adenocarcinoma. The regimen involves combination therapy with FLOT chemotherapy (5-fluorouracil, leucovorin, oxaliplatin, and docetaxel) before surgery, followed by Imfinzi monotherapy as maintenance after surgery.Do-Youn Oh, Professor of Hematology and Oncology at Seoul National University HospitalWith this approval, Imfinzi has become the first immuno-oncology drug approved in Korea for use in the perioperative treatment setting for gastric cancer.Due to advanced screening systems and surgical techniques, the 5-year survival rate for gastric cancer patients in East Asia has been in the 75–80% range with postoperative adjuvant chemotherapy alone.However, approximately 30–40% of stage III patients still experience recurrence, indicating persistent unmet needs.Against this backdrop, perioperative treatment strategies involving chemotherapy before and after surgery have emerged as an alternative.The goal of perioperative therapy is to eliminate micrometastases early and continuously suppress systemic disease thereafter.In the U.S. and Europe, FLOT-based perioperative treatment has already become the standard. The addition of Imfinzi to this regimen has demonstrated significant clinical efficacy, supporting a shift in treatment patterns.Do-Youn Oh, Professor of Hematology and Oncology at Seoul National University Hospital, said, “Perioperative strategies to improve resection rates are already standard overseas. The clinical benefits of combining immunotherapy with chemotherapy are clear.”The Phase III MATTERHORN study was the basis for Imfinzi’s expanded indication. The trial was conducted on patients with stage II-III advanced gastric cancer who were candidates for curative surgery. While stage I gastric cancer has a high cure rate with surgery alone, stages II–III represent locally advanced disease with a higher risk of recurrence.In this study, Imfinzi-based perioperative therapy showed a statistically significant improvement in overall survival (OS).The efficacy of Imfinzi was consistently observed in Asian patients as well.In an Asian subgroup analysis presented at ESMO Asia 2025, the Imfinzi plus FLOT combination demonstrated improvements in event-free survival (EFS), 3-year OS, and pathological complete response (pCR) compared to placebo plus FLOT.At 24 months, the EFS rate was 72.1% in the Imfinzi group versus 64.2% in the placebo group. Median EFS was not reached in either group, suggesting potential widening of the gap with longer follow-up. OS also showed a similar improvement trend to that observed in global studies.The improvement in pCR was particularly notable. In the Asian patient population, the pCR rate in the Imfinzi combination group was 18.9%, more than three times higher than the 5.6% in the placebo group.Safety was also confirmed to be manageable compared to standard FLOT therapy. There were no significant differences between the two groups in Grade 3 or higher adverse events or treatment discontinuation rates, indicating that new safety concerns arising from the addition of an immunotherapy were limited.On the 31st, AstraZeneca Korea held a press conference at the Four Seasons Hotel Seoul to explain changes in treatment strategies following the expansion of Imfinzi’s indication for gastric cancer.Despite surgery remaining the cornerstone of gastric cancer treatment, there is growing recognition that surgery alone may not be sufficient for a cure. The MATTERHORN study suggests that combining immunotherapy and chemotherapy before surgery, followed by surgery and maintenance therapy, can improve long-term outcomes.Professor Oh emphasized, “The proportion of patients completing postoperative Imfinzi adjuvant therapy was around 50%, which exceeded expectations. For patient groups at high risk of micrometastasis, it is important to determine treatment strategies by comprehensively considering various factors such as extensive lymph node involvement, T4 stage, and aggressive histological subtype.”She added, “Clear criteria for determining which patients should undergo surgery first or receive neoadjuvant chemotherapy have not yet been established. Further discussion and accumulation of evidence are necessary to establish treatment strategies tailored to patient characteristics.”
- Company
- Oral CSU drug ‘Rhapsido’ nears approval in KOR
- by Eo, Yun-Ho Apr 02, 2026 08:46am
- The oral urticaria drug ‘Rhapsido’ is nearing commercialization in Korea.According to industry sources, Novartis Korea’s oral BTK inhibitor Rhapsido (remibrutinib) is expected to receive marketing authorization from the Ministry of Food and Drug Safety next month (May).Rhapsido is an oral targeted therapy that inhibits Bruton’s tyrosine kinase (BTK), a key pathway in the pathophysiology of CSU, thereby blocking the release of histamine and inflammatory mediators.The drug was approved in the United States last September for the treatment of adult CSU patients whose symptoms persist despite second-generation H1 antihistamines.CSU is a disease characterized by severe symptoms and unpredictable exacerbations, making diagnosis and management difficult. It is known to arise from immune dysregulation. In CSU patients, the immune system can be activated via allergic (IgE) or autoimmune (IgG) pathways.This leads to specific immune cells activating the BTK protein. Once activated, BTK triggers the release of histamine and other pro-inflammatory mediators, causing red, swollen, and itchy hives.The most notable feature of Rhapsido is that it is an oral drug (taken twice daily). Until now, treatment options for patients unresponsive to first-line antihistamines have been largely limited to the injectable biologic Xolair (omalizumab). The arrival of Rapsido opens a new option, an oral targeted therapy.The drug demonstrated efficacy in the Phase III REMIX-1 and REMIX-2 studies. Results showed Rhapsido demonstrated superiority over placebo in improving itch severity (ISS7), hive severity (HSS7), and total urticaria activity score (UAS7) starting from Week 2. Approximately one-third of patients achieved complete remission (defined as zero itch and zero hives) by Week 12.Beyond CSU, Novartis is also expanding clinical development of Rhapsido across a range of immune-mediated diseases, including chronic inducible urticaria (CIndU), hidradenitis suppurativa (HS), food allergy, and multiple sclerosis.
- Company
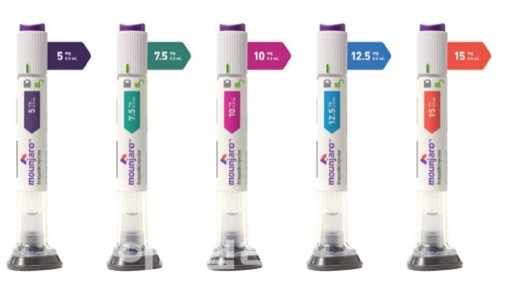
- Lilly Korea sales, 194%↑ from a year earlier…'Mounjaro' effect
- by Son, Hyung Min Apr 02, 2026 08:46am
- Eli Lilly Korea's sales structure in South Korea is undergoing a rapid, substantial changes. Following the launch of the GLP-1 class blockbuster 'Mounjaro,' the company’s financial performance has surged, completely restructuring its growth model, which previously centered on its core products.According to the Financial Supervisory Service, Eli Lilly Korea’s sales last year reached KRW 482.1 billion, a 193.6% increase compared to the previous year. During the same period, operating profit rose by 259.2%, jumping from KRW 10.3 billion in 2024 to KRW 37.1 billion last year. Eli Lilly Korea's sales performance by year (unit: KRW 100 million)Previously, the company's sales relied on oncology drugs such as ‘Verzenio (abemaciclib)’ and ‘Cyramza (ramucirumab),’ as well as the SGLT-2 inhibitor ‘Jardiance (empagliflozin)’ and the biologic ‘Taltz (ixekizumab).’ Due to a lack of new blockbuster entries, sales had stalled below KRW 200 billion from 2021 to 2024.However, this structure changed abruptly with the emergence of ‘Mounjaro (tirzepatide).’ Mounjaro secured outstanding growth momentum by rapidly expanding beyond diabetes into the obesity treatment market.Mounjaro acts on both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. Through this dual action, it stimulates insulin secretion, improves insulin resistance, and decreases glucagon secretion, thereby lowering both fasting and postprandial blood glucose levels.In patients with diabetes and obesity, the "incretin effect" is typically diminished, primarily due to reduced GLP-1 secretion and impaired GIP action. As GLP-1 and GIP are key hormones responsible for approximately two-thirds of the postprandial insulin response, the dual-axis mechanism of Mounjaro stands out as a significant clinical advantage.Mounjaro's first indication was secured in June 2023 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. In August 2024, the indication was expanded for chronic weight management. By expanding its scope to include patients with obesity or overweight patients with weight-related comorbidities, Mounjaro successfully became positioned as an obesity treatment.Released in the domestic market last August, Mounjaro quickly secured position in the market. According to market research firm IQVIA, Mounjaro recorded KRW 28.4 billion in sales in Q3 of last year, which then surged to KRW 187.1 billion in Q4, easily surpassing KRW 100 billion in quarterly sales for a single item. During the same period, it outpaced its competitor 'Wegovy (semaglutide),' rapidly increasing its market dominance.A similar trend is observed in the global market. As of Q2 of last year, Mounjaro's global sales exceeded those of Wegovy, marking a significant turning point in the battle for leadership in the obesity treatment market.Active Development of Multi-Mechanism GLP-1 SuccessorsLilly is also continuously strengthening its GLP-1-based portfolio. Currently, the oral GLP-1 agonist 'orforglipron' is undergoing regulatory approval processes in more than 40 countries, with a New Drug Application (NDA) for type 2 diabetes expected in the U.S. by the end of this year.Unlike Mounjaro, orforglipron is a single GLP-1 mechanism but distinguishes itself as an oral medication. Notably, it does not require fasting after administration and, as a small-molecule-based drug, has lower production costs, suggesting high market scalability. In clinical trials, it has shown superior results compared to competitors in both HbA1c reduction and weight loss.Lilly's diabetes and obesity treatment 'Mounjaro'Next-generation pipelines are also under development. For instance, 'retatrutide,' a triple agonist that simultaneously targets GLP-1, GIP, and glucagon (GCG). Currently, no triple-action obesity drug has been approved, and retatrutide, which is in Phase 3 clinical trials, is considered the closest to commercialization.According to recently released Phase 3 study results, retatrutide met primary endpoints by demonstrating significant improvements in HbA1c and weight loss compared with placebo. Lilly is considering strategies to expand retatrutide's indications beyond obesity to various chronic diseases, including diabetes and liver disease.Additionally, 'eloralintide,' which targets both GLP-1 and amylin receptors, has entered global Phase 3 trials. This drug mimics the action of the amylin hormone secreted by the pancreas to act directly on the brain, thereby increasing satiety and suppressing food intake.
- Company
- Global pharmas cautiously welcome pricing reform
- by Son, Hyung Min Apr 01, 2026 08:15am
- “Domestic firms are crying, multinational firms are smiling.” This is the prevailing assessment of Korea’s latest drug pricing reform.With the reform centered on lowering prices of generic drugs now finalized, Korea’s drug pricing structure is approaching a turning point. While the government has proposed reallocating savings toward rewarding innovative drugs and improving patient access, the industry is also voicing cautious views that outcomes will depend on the system’s effectiveness and execution.According to industry sources, the Ministry of Health and Welfare finalized the drug pricing reform plan on the 26th through the Health Insurance Policy Deliberation Committee (HIPDC). The reform focuses on lowering the pricing benchmark for generic and off-patent drugs from the current 53.55% to around 45%.This reform is regarded as significant not simply as a price cut, but as a structural reorganization aimed at strengthening incentives for innovative drugs using the savings generated.The key lies in resource reallocation. The strategy is to use the funds secured by adjusting the generic-centric pricing structure to lower barriers to reimbursement for new drugs through measures such as ▲faster reimbursement listing of treatments for rare and severe diseases, ▲introduction of flexible drug pricing contracts, and ▲raising the ICER (incremental cost-effectiveness ratio) threshold.This is interpreted as a response to long-standing criticism that Korea’s pricing system has focused excessively on cost containment, limiting access to innovative therapies.The Ministry of Health and Welfare also defined the reform as a structural transformation of the pricing system.The Ministry stated, “By advancing the drug pricing system to the level of major countries, we can enhance public access to treatment and coverage while r reducing drug expenditure burdens. Establishing a compensation system for research and development and efforts to ensure the stable supply of essential medicines will serve as a momentum for the pharmaceutical and biotech industries to take the leap forward.”Global pharmas express “cautious optimism”…System design is keyGlobal pharmaceutical companies are generally welcoming the reform. Given that patient access and reimbursement rates for new drugs in Korea have lagged behind major countries, there is an expectation that savings from generic price cuts could improve access to innovative therapies.The Korea Research-based Pharmaceutical Industry Association (KRPIA), which is primarily composed of multinational pharmaceutical companies, also offered a positive assessment.The association stated, “he policy reflects a commitment to reflecting the value of innovative new drugs and enhancing patient access. It is crucial whether the reform’s intent is actually realized through system design and implementation,” thereby emphasizing the importance of policy implementation.This expectation is also supported by data. According to PhRMA’s ‘2023 Global Access to New Medicines Report,’ among the 460 new drugs covered by health insurance worldwide from 2012 to 2021, South Korea’s coverage rate was 22%, falling below the G20 (28%) and OECD (29%) averages.For innovative cancer drugs, the rate was 23%, and for rare disease treatments, it was just 12%, both significantly lower than the G20 and OECD averages, respectively.However, global pharmaceutical companies are also expressing conditional caution. While the entry environment may improve, there are concerns that requirements for demonstrating value during reimbursement listing have become more stringent.An official from a global pharmaceutical company noted, “It is positive in terms of improving access to new drugs and strengthening clinical value-based evaluation. However, the strengthened post-listing price control raises concerns about predictability.”Another official from a global pharmaceutical company agreed with the direction but raised questions about its implementation.The official said, “The changes in government perception toward rare and severe diseases are significant. Even drugs under the pilot approval-evaluation-negotiation linkage program are facing reimbursement delays, so concrete execution plans are urgently needed.”Regarding the increase in the ICER threshold, the official emphasized, “The magnitude of the increase is more important than the direction itself. If implementation is delayed under the pretext of policy research, it will just become another waiting period for patients.”Institutional issues surrounding global pharmaceutical companies also remain unresolved. Industry feedback regarding the revision of certification criteria for innovative pharmaceutical companies was, “only the name has changed, with limited substantive improvement.”Apart from the fact that bonus points are awarded for certain factors such as attracting foreign capital, joint research, and open innovation, the assessment is that the industry’s long-standing demand for the inclusion of achievements in attracting headquarter-level R&D investment has not been sufficiently reflected. Furthermore, the fact that indicators, such as the scale of pharmaceutical exports, which are difficult for multinational companies to meet, remain unchanged, poses disadvantages.While some companies are already expanding cooperation with the government and joint research with domestic firms with certification in mind, the extent to which these efforts will be reflected in evaluations remains uncertain.An industry official stated, “To foster the domestic biotech ecosystem, collaboration with global pharma is essential. Since the role of domestic branches is crucial in attracting headquarter-level R&D investment, these characteristics need to be reflected in the design of the system.”Another industry official noted, “The new system is already affecting future pipeline processes. Multinational companies have been strengthening preparations to obtain the Innovative Pharmaceutical Company certification,” suggesting that this reform is bringing about changes in actual business strategies in practice.
- Company
- NMOSD drug ‘Uplizna’ fails reimbursement again in KOR
- by Eo, Yun-Ho Apr 01, 2026 08:15am
- Uplizna, a new drug for neuromyelitis optica spectrum disorder (NMOSD), has failed to secure reimbursement listing in its second attempt.According to Dailypharm coverage, Mitsubishi Tanabe Pharma Korea’s pricing negotiations with the National Health Insurance Service (NHIS) for Uplizna (inebilizumab), a treatment used to treat adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are positive for anti-Aquaporin-4 (AQP4) antibodies, have ultimately collapsed.Although both sides made efforts to continue discussions by extending the negotiation period, it is understood that disagreements arose over adjustments to the expenditure cap.Consequently, it is expected that Uplizna, which had been gaining expectation as a new treatment option for NMOSD, will face significant challenges in establishing a practical prescription environment in the domestic market for the time being.Uplizna had also previously halted its listing process last October at the pricing negotiation stage due to supply-related issues.At the time, the company had accepted the “below the evaluated amount” condition set by the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee and entered price negotiations. However, the parties failed to reach an agreement within the 60-day negotiation period. Subsequently, HIRA attempted to enter into extended negotiations, but the talks could not begin as the pharmaceutical company was unable to ensure domestic supply.Supply-related issues for Uplizna remain unresolved. The drug was originally developed by Amgen, while Tanabe holds commercialization rights for Korea and other Asian countries through a licensing agreement.It remains to be seen what steps Uplizna will take next after failing its second attempt to secure reimbursement coverage in Korea.NMOSD occurs when AQP4 autoantibodies, a disease-specific biomarker produced by B cells, bind to AQP4, a target antigen present on glial cells in the central nervous system, and activate the immune responses, causing nerve damage.Uplizna is an anti-CD19 human monoclonal antibody that selectively binds to CD19, a B-cell-specific surface antigen, depleting B cells that produce AQP4 antibodies, thereby preventing disease relapse.The safety and efficacy of Uplizna were demonstrated in the N-MOmentum study, which evaluated the use of Uplizna monthly in 230 patients without the use of concomitant immunosuppressive agents.Study results showed that 89% of patients treated with Uplizna did not experience a relapse during 197 days of follow-up, resulting in a 77.3% reduction in the risk of relapse compared to placebo. Safety evaluations of Uplizna also showed comparable rates of adverse events to the placebo group.
- Company
- Patient & Consumer Alliance says "Structural reform needed for drug pricing reform"
- by Son, Hyung Min Mar 31, 2026 08:45am
- Patient & Consumer Alliance for Healthcare Rights (PCA) provided both positive evaluations and concerns over structural limitations regarding the recent drug pricing system reform. The drug price cut is a significant change; However, they argue that a breakthrough reform remains difficult as long as the market remains centered on rebate-driven structures.On the 27th, the PCA stated as such after the Health Insurance Policy Review Committee approved the "Measures to Improve the National Health Insurance Drug Pricing System" the previous day.The Patient & Consumer Alliance for Healthcare Rights (PCA) was officially launched on the 24th joined by four public citizens and patient organizations, including the Consumers Union of Korea, the Korea Alliance of Patient Organizations, Citizens' Movement for Consumers, and the Korean Organization for Rare Diseases, with the goal of shifting the current "government-led and provider-centric" medical structure toward a patient- and consumer-centered one (photo= Korean Organization for Rare Diseases).Previously, the Ministry of Health and Welfare (MOHW) finalized a plan to lower the drug price calculation rate for generics and off-patent drugs from the current 53.55% to 45%. The PCA evaluated this measure as "meaningful progress, marking the first reform of the generic drug pricing structure in 14 years."However, the PCA stated that price reductions alone are insufficient to eradicate the practice of rebates.The PCA stated, "Drug prices are not commensurate with rebates and that rebate practices will persist as long as the unfair competitive structure across licensing and distribution remains intact."They also raised concerns about the tiered price adjustment structure and the newly introduced "New Innovative Pharmaceutical Company" system included in the reform.According to the PCA, "The grace period of up to 10 years delays market restructuring," adding, "New Innovative Pharmaceutical Company" system lacks clear criteria and evaluation methods, potentially allowing companies without innovative capabilities to be incorporated into the support system.Furthermore, the PCA identified potential issues with the government's policies supporting the pharmaceutical and biotech industries. They argued that if this continues, with savings from price cuts being reinvested in industrial support, it could result in using taxpayer money to sustain companies with uncertain competitiveness.The PCA further stated, "The scale and performance of investments into pharmaceutical R&D over the 26 years since the separation of prescribing and dispensing have never been fully disclosed to the public," adding, "Discussions on additional support should be provided only after independent performance evaluations are made public."Issues were raised regarding the sales structures of certain companies within the Korean pharmaceutical market.The PCA pointed out, "There may be so-called bogus pharmaceutical companies that maintain market presence through rebate-driven sales via Contract Sales Organizations (CSOs) without possessing production facilities or research capabilities," and argued that "These companies undermine the competitive foundation for legitimate companies."The PCA stated, "If this structure persists even after lowering drug prices, companies relying on rebates, rather than those with true competitiveness, will maintain their market status," and adding, "The key is the normalization of the competitive order rather than just price reduction."The PCA stressed that government support is necessary to improve the pharmaceutical market structure. They called for the immediate launch of a 'Pharmaceutical Market Fair Trade Task Force (TF)' involving the Ministry of Health and Welfare, the Ministry of Food and Drug Safety, and the Fair Trade Commission to promote: ▲Eradicating rebates and strengthening CSO management ▲Establishing exit criteria for pharmaceutical companies without production capabilities ▲Disclosing performance results of financial support for pharmaceutical R&D.The PCA stated, "Drug price reduction is only the beginning, and as long as the rebate structure remains, patients may not receive benefits," adding, "The government must prioritize creating a foundation for fair competition before returning financial savings to the industry."The PCA concluded, "We hope this pricing reform serves as a starting point for reform leading to a fair pharmaceutical market," and stated, "The alliance would continuously monitor the implementation process." The PCA emphasized that government support is necessary to improve the pharmaceutical market structure.