- LOGIN
- MemberShip
- 2026-05-07 17:28:12
- Company
- Yuhan presents 4 studies at ASCO...builds reliability
- by Jung, Sae-Im Jun 08, 2023 05:33am
- Yuhan Corp’s EGFR-targeting anticancer drug ‘Leclaza’ has started to accumulate data for the international stage. At the ‘2023 ASCO Annual Meeting (ASCO 2023)’ that was held on the 2nd (local time), Yuhan Corp made 4 poster presentations on studies related to its Leclaza, including one long-term follow-up study. With the results of Janssen’s main MARIPOSA study expected to be released in the second half of this year, initial research results that give a glimpse of the MARIPOSA study have also been updated. Leclaza is a treatment for non-small-cell lung cancer that was approved as the 31st homegrown novel drug in January 2021. It is a 3rd generation EGFR TKI that inhibits the proliferation and growth of lung cancer cells. It is currently approved as a treatment for patients with locally advanced or metastatic NSCLC who developed resistance after being previously treated with 1st generation or 2nd generation EGFR-TKIs. The drug had raised KRW 25 billion in sales in only 2 years of release. The company is also awaiting to add a first-line indication to the drug. Supported by such domestic growth, the company seeks to further its presence with Leclaza in the global market. Janssen, which introduced Leclaza’s technology, is developing it as a combination therapy with Rybrevant, its EGFR bispecific antibody treatment. The results of the MARIPOSA Phase III study, which looks at the drug’s effect of combination therapy in the first line, are expected to be announced soon. Research data that provide a glimpse into the brain metastasis effect of Leclaza and the efficacy of Leclaza as a combination therapy were presented at this year’s ASCO. First, the long-term follow-up results of the Phase I CHRYSALIS trial, which used EGFR exon 20 mutation treatment Rybrevant with Leclaza in EGFR-mutated NSCLC, were presented as a poster. The CHRYSALIS trial was the first study to evaluate the efficacy of Rybrevant + Leclaza as a first-line treatment. Long-term follow-up of Leclaza+Rybrevan combo (Data: ASCO) After following 20 patients for 33.6 months, results showed that half of the patients were able to achieve progression-free survival. At that time point, key indicators such as overall survival, progression-free survival, and duration of response had not yet reached median values. Following the poster presentation, an oral presentation on the Cohort D data of the CHRYSALIS-2 study, which evaluated the safety of Leclaza + Rybrevant therapy or Leclaza monotherapy after treatment with Tagrisso, was made at the meeting. The presented data were the results of the additional analysis conducted on biomarkers from the data that had been announced in 2021. Subanalysis of Leclaza+Rybrevant combo according to presence of MET mutations (Data: ASCO) The effect of combination therapy was significantly higher in the MET expression group. The objective response rate (ORR) of the combination therapy in patients with MET mutations and amplification (28 patients) was 61%, which was higher than 14% in the MET-negative patient group (49 patients). The median duration of response and median progression-free survival were 10.8 months and 12.2 months in the MET-positive group, respectively, compared to 6.8 months and 4.2 months in the MET-negative patient group. A domestic phase 2 study that measured the effect of Leclaza in patients with brain metastasis after failure with existing first- and second-generation targeted anticancer drugs in EGFR-positive non-small cell lung cancer was also presented as a poster. 40 EGFR-positive patients with brain metastases after using first- and second-generation treatments were enrolled in the study to evaluate the intracranial activity of Leclaza in patients with asymptomatic or mild brain metastasis. Patients who failed with conventional treatment were divided according to the presence or absence of the T790M mutation. The primary evaluation index was intracranial objective response rate (iORR), and the secondary evaluation index was intracranial progression-free survival (iPFS). The intracranial objective response rate of 38 evaluable patients was 55.3%. 3 patients showed a complete response and 18 patients showed a partial response. There were only 5 T790M-positive patients, but 4 showed partial response, recording an intracranial response rate of 80%. Of the 33 T790M-negative patients, 3 showed complete responses and 14 partial responses, recording an objective response rate of 51.5%. The median overall progression-free survival and the progression-free survival in the T790M positive and negative groups were 15.2 months, 9.9 months, and 15.4 months, respectively. Intracranial progression-free survival was similar for T790M positive and negative patients, 15.2 months and 15.8 months, respectively. The research team said, “Leclaza showed significant intracranial activity regardless of the presence or absence of T790M mutation, and satisfied the primary evaluation index with an intracranial objective response rate of 55.3%." This indicates that patients that showed brain metastasis after treatment with targeted therapies may use lazertinib instead of topical treatment." In addition to this, studies that studied predictable biomarkers for the use of Rybrevant + Leclaza after Tagrisso were also announced at ASCO 2023.
- Company
- Novartis counterattacks against Celltrion / a patent lawsuit
- by Kim, Jin-Gu Jun 07, 2023 09:47pm
- Novartis is investigating Celltrion for patent infringement of its biosimilar Xolair, asthma, and urticaria treatment. According to the pharmaceutical industry on the 2nd, Novartis filed two claims against Celltrion on the 1st. A proactive trial to confirm the scope of rights is to ask the Intellectual Property Trial and Appeals Board to accurately determine the scope of patent validity to see if the patent holder has been infringed. This case is interpreted as a situation in which Novartis is claiming Celltrion's patent infringement. In other words, Celltrion is claiming that the Xolair biosimilar, which is being developed, has infringed on two pharmaceutical patents it owns. Xolaire's material patent has already expired, and only the formulation patent remains. In Korea, the formulation patent expires in March 2024. Celltrion is developing 'CT-P39' as a Xolair biosimilar. It is evaluated that it is close to commercialization, such as announcing the interim results of the global phase 3 clinical trial in April. It has already applied for product approval in Europe and plans to apply for approval within the year in Korea. Analysts say that if the Intellectual Property Trial and Appeals Board upholds Novartis, the original company, in an active trial to confirm the scope of rights, setbacks in Celltrion's plan to release the Xolair biosimilar in Korea are inevitable. Xolair is a biopharmaceutical developed by Genentech and Novartis. It is used to treat allergic asthma, chronic urticaria, and chronic rhinosinusitis. According to IQVIA, a pharmaceutical market research institute, Xolair's domestic sales in the first quarter were 4.6 billion won. It increased by 34% from 3.4 billion won in the first quarter of last year. Last year, it posted sales of 15.7 billion won in Korea.
- Company
- Kwangdong wins patent lawsuit against Ibrance in reverse
- by Kim, Jin-Gu Jun 07, 2023 05:38am
- Ibrance As Kwangdong Pharmaceutical succeeded in reversing the second trial of the patent lawsuit against Ibrance, it boarded the last train for generic exclusivity. As a result, Ibrance's generic for exclusivity is owned by three companies, including Daewoong Pharmaceutical and Shinpoong Pharmaceutical, which won in the first trial, and Kwangdong Pharmaceutical. According to the pharmaceutical industry on the 3rd, the Patent Court ruled in favor of the plaintiff on the 2nd in the patent trial cancellation lawsuit filed by Kwangdong Pharmaceutical against Pfizer. Kwangdong Pharmaceutical, which was defeated in the first trial, succeeded in reversing in the second trial. A total of five companies have challenged the Ibrance patent. These are Kwangdong Pharmaceutical, Shinpoong Pharmaceutical, Daewoong Pharmaceutical, Boryung, and Samyang Holdings. They requested a passive trial to confirm the scope of rights to the Ibrance crystalline patent, which expires in February 2034. Their plan was to avoid crystalline patents and release generics early in time for substance patents to expire in 2027. In the first trial, the decision was mixed. The Intellectual Property Trial and Appeal Board rendered a victory decision for Daewoong Pharmaceutical and Shinpoong Pharmaceutical, and a defeat decision for Kwangdong Pharmaceutical, Boryung, and Samyang Holdings. With the first trial victory, Daewoong Pharmaceutical and Shinpoong Pharmaceutical acquired Ibrance generic for exclusivity. Kwangdong Pharmaceutical, Boryung, and Samyang Holdings, which lost in the first trial, appealed to the Patent Court. Among the three companies, the ruling of Kwangdong Pharmaceutical came out first. Kwangdong Pharmaceutical barely succeeded in obtaining generic exclusivity through a second trial victory. This is because the period for acquiring generic for exclusivity was about to expire. According to the current regulations, there are three requirements to receive generic for exclusivity. A patent trial must be requested for the first time, this trial or subsequent litigation must be won, and a generic license must be applied for the first time. At this time, one proviso clause is attached to the requirement that the judgment and lawsuit be won. The point is that a generic company must apply for generic exclusivity to the Ministry of Food and Drug Safety and obtain a winning trial decision or ruling within nine months from the date the original company is notified of this fact. Kwangdong Pharmaceutical applied for a generic for the exclusivity of Ibrance on August 29 last year. In early September, it is expected that this fact will be notified to Pfizer. In other words, if Kwangdong Pharmaceutical did not win the case by early this month, even generic for exclusivity would be useless. As Kwangdong Pharmaceutical's second trial proceeded quickly, it succeeded in winning the case in early June, and generic exclusivity was eventually made possible. Kwangdong Pharmaceutical is the only company that has even obtained product approval among patent challengers. Kwangdong Pharmaceutical received approval for Alency as a generic version of Ibrance in March of this year. On the other hand, it is analyzed that Boryung and Samyang Holdings, which appealed in the second trial along with Kwangdong Pharm, are highly unlikely to receive generic for exclusivity. The hearing date for Boryung and Samyang Holdings was set for July. Ibrance is a hormone receptor (HR) positive or human epidermal growth factor receptor 2 (HER2) negative treatment for advanced and metastatic breast cancer. According to IQVIA, a pharmaceutical market research institute, Ibrance posted sales of 56.2 billion won in Korea last year. In the first quarter of this year, it was 12.6 billion won, down 13% from the same period last year.
- Company
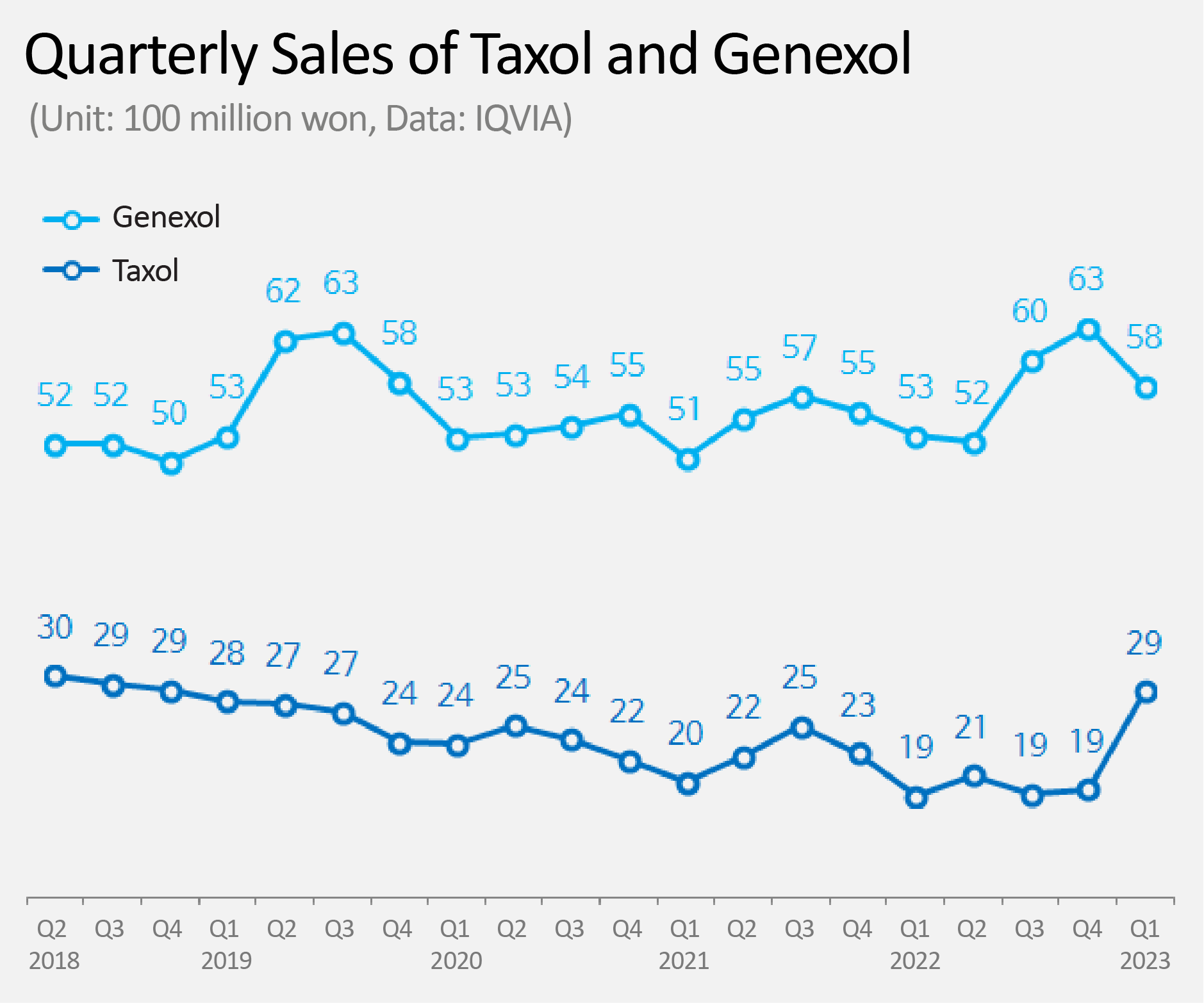
- Sales of anti-cancer drug 'Taxol' increases by 53%
- by Kim, Jin-Gu Jun 07, 2023 05:38am
- Product Photos of Taxol (LT) and Genexol (RT) Sales of the original Paclitaxel anti-cancer drug 'Taxol' increased 53% in one year. This change has seemed to have occurred as Boryung took over domestic sales from the beginning of this year. According to IQVIA, a pharmaceutical market research institute, on the 2nd, Taxol's sales in the first quarter was KRW 2.9 billion. This is a 53% increase from KRW 1.9 billion in the first quarter of last year. Taxol is a paclitaxel-based cytotoxic anti-cancer drug, and is widely used in various types of cancer such as ovarian cancer, breast cancer, lung cancer, and stomach cancer. Despite having been nearly 30 years since it was approved in Korea in 1996, and is still widely used. Until last year, sales of Taxol had been on a steady decline. The downhill started in the second quarter of 2018 from KRW 3 billion, declining to KRW 1.9 billion in quarterly sales last year. The pharmaceutical industry cites Boryung as the reason for Taxol's successful rebound. Boryung has been in charge of sales of Taxol since the first quarter of this year. It is analyzed that Boryung brought rebound in sales of Taxol through its already-established solid sales grounds in the field of anti-cancer drugs. Quarterly Sales of Taxol and Genexol (Unit: 100m million won, Data: IQVIA) Boryung's past actions are quite interesting. From 2008 to 2015, Boryung jointly sold Taxol with BMS for 8 years. From 2016 up to last year, Boryung jointly sold Genexol, a competing product of Taxol, with Samyang Holdings (formerly Samyang Biopharmaceuticals Corp.). From this year onwards, Boryung has taken charge of the sales of Taxol again. While Boryung was in charge of sales of Genexol, Genexol surpassed its original product Taxol and rose to no.1 in the market. For Boryung, a situation unfolded in which it now has to compete with the product that it had put on the top of the market. Genexol, which was previously sold by Boryung, is now being sold by HK inno.N from this year. Genexol's first quarter sales were KRW 5.8 billion, which is a 9% increase from the first quarter of last year, KRW 5.3 billion. HK inno.N has jointly sold Genexol from 2001, when Samyang Holdings first developed the product as a generic Paclitaxel drug, to 2013. The joint sales agreement reunited the two companies after 10 years. The pharmaceutical industry's attention is focused on whether Boryung, which has again taken charge of the sales of Taxol, will be able to catch up with Genexol by maintaining the same upward trend as in the first quarter. For HK inno.N and Samyang Holdings, on the other hand, the key is how firmly they maintain the market share of Genexol.
- Company
- Pfizer's ADC Mylotarg can be prescribed by Big 5
- by Eo, Yun-Ho Jun 07, 2023 05:38am
- Pfizer's new ADC drug, Mylotarg, has settled in a Big 5 general hospital. According to the related industry, Pfizer Korea's acute myeloid leukemia (AML) Mylotarg is a drug for medical institutions such as SMC, SNUH, Seoul St. Mary's Hospital, AMC, Shinchon Severance Hospital, as well as advanced general hospitals such as the National Cancer Center and Hwasun Chonnam National University Hospital. passed the committee. Mylotarg is an antibody-drug conjugate that can be used for the first-line treatment of adult patients with newly diagnosed CD33-positive AML. However, Mylotarg has not yet received insurance benefits. This drug was presented to HIRA in May of last year, but it was judged that the reimbursement standard was not set. Mylotarg, which was approved in Korea in December 2021, is an ADC composed of a CD33-targeting monoclonal antibody and a cytotoxic drug, calicheamicin, and acts on cells expressing the CD33 antigen, which is seen in 90% of all AML patients. This blocks cancer cell growth and induces apoptosis. The approval of Mylotarg was based on a clinical study conducted on 271 newly diagnosed AML patients with no treatment experience before the age of 50 to 70 years. The clinical trial was an open-label, randomized, multicenter phase 3 clinical trial for ALFA-0701. The existing anticancer chemotherapy, daunorubicin or cytarabine combination therapy, and Mylotarg, daunorubicin, and Cytarabine combination therapy were compared and evaluated. As a result, the Milotac + Daunorubicin + Cytarabine combination administration group had a median event-free survival (EFS) of 17.3 months, which was about 7.8 months longer than the Daunorubicin + Cytarabine combination administration group's 9.5 months. seemed It reduced the risk of induction failure, relapse, or death by approximately 44%. The median relapse-free survival (RFS) was 28.0 months for the Milotac + Daunorubicin + Cytarabine combination group and 11.4 months for the Daunorubicin + Cytarabine combination group, showing a significant difference of about 16.6 months. The median overall survival was 27.5 months for the Mylotarg + Daunorubicin + Cytarabine combination group and 21.8 months for the Daunorubicin + Cytarabine combination group, showing no statistically significant difference.
- Company
- 'Saxenda' continues to dominate weight-loss market
- by Kim, Jin-Gu Jun 07, 2023 05:38am
- Product Photos of Saxenda (LT) and Qsymia (RT) 'Saxenda (Liraglutide)' is further strengthening its dominance in the obesity treatment market. In the first quarter, it recorded sales of KRW 15.9 billion, 53% increase from the previous year, more than doubling the gap with the runner-up product, 'Qsymia (Phentermine+Topiramate).' However, it remains to be seen how much longer Saxenda's dominance will last, as release of promising products such as 'Wegovy (Semaglutide)' and 'Mounjaro (Tirzepatide)' are imminent in the obesity treatment market. The pharmaceutical industry predicts that the two products which have proven their marketability in the global market will be released in Korea as early as this year. Saxenda grows 53% in 1 year... more than doubles the gap with Qsymia According to IQVIA, a pharmaceutical market research institute, on the 5th, Novo Nordisk's Saxenda recorded sales of KRW 15.9 billion in the first quarter. This is a 53% increase in 1 year, compared to KRW 10.4 billion in the first quarter of 2022. Saxenda is the world's first obesity treatment approved as a glucagon-like peptide-1 (GLP-1) analog. It has the same ingredients as the type-2 diabetes treatment 'Victoza,' but with different usage and dosage. Saxenda has grown rapidly since its domestic release in the second quarter of 2018. In 2019, the second year after its release, Saxenda dominated the obesity treatment market with sales of KRW 42.6 billion. Saxenda, unlike existing obesity treatments, is not a psychotropic drug, and therefore gained explosive popularity because it is relatively safe and can be taken over a long period. The sales of Saxenda have somewhat slowed down due to the impact of COVID-19 in 2020 and 2021. However, as outdoor activities have become revitalized again from last year, demand for obesity treatments regained its place, and Saxenda's sales soared up to KRW 58.9 billion. Quarterly Sales of Saxenda and Qsymia (Unit: 100 million won, Data: IQVIA) Qsymia, the runner-up product in the market, marked sales of KRW 7.7 billion in the first quarter. Compared to KRW 6.3 billion in the first quarter of last year, sales has increased by 21%. It is analyzed that Qsymia was also affected by the recovery of the obesity treatment market. Qsymia is a combination drug of 'Phentermine' and 'Topiramate,' which Alvogen Korea secured domestic sales right from the US pharmaceutical company Vivus in 2017. Alvogen Korea started domestic sales with Chong Kun Dang at the end of 2019. In addition to the advantage that the content of psychotropic ingredient is relatively low even though it is an oral drug, Chong Kun Dang's sales power generated synergy and quickly penetrated the market despite being a latecomer. However, the gap with Saxenda has widened. Although Qsymia chased Saxenda's sales up to about 90% with KRW 5.9 billion in the first quarter of 2021, the gap is widening again as the obesity treatment market is recovering. In the first quarter of this year, the gap between to two products widened by 2.4 times. Some in the pharmaceutical industry predicts that the gap between the two products will widen further in the future. This is because obesity treatments containing Phentermine and Phendimetrazine, including Qsymia, were included in the 'list of narcotics and drugs of concern for misuse and abuse' as the non-face-to-face treatment pilot project was implemented. The government urged caution in prescibing psychotropic drugs such as Qsymia through non-face-to-face treatment. Saxenda has not been included in the list. In addition, treatments such as Daewoong Pharmaceutical's Dietamin, Korea Prime Pharm's Phendimen, Huons's Hutermin, etc. recorded sales of more than KRW 1 billion in the first quarter. In case of Phendimen, its sales, which recorded just KRW 300 million in the first quarter of last year, increased about 6 times to KRW 1.8 billion in one year, showing remarkable growth. Wegovy and Mounjaro's impending release... Saxenda's domination coming to an end It remains to be seen how much longer Saxenda's dominance will last. Two mega-sized products that have proven their competitiveness in the global market are waiting to be released. In the pharmaceutical industry, the prevailing view is that Saxenda's dominance will come to an end with the advent of next-generation products such as Wegovy and Mounjaro. Product Photos of Wegovy (LT) and Mounjaro (RT) In April, the Ministry of Food and Drug Safety approved Novo Nordisk's Wegovy. Wegovy is a GLP-1 analog, just like Saxenda. Novo Nordisk improved its Saxenda, which was administered daily, to weekly administration. In the US market, in which Wegovy was released earlier, demand for the product soared, with shortages occurring. In particular, due to its popularity, shortage of Ozempic, a diabetes treatment with the same ingredients and usage, has also occurred. Even now, there is still a lack of supply of Wegovy in the US. Due to the circumstances, the official release of Wegovy is being delayed in Korea even after product approval. The pharmaceutical industry predicts that domestic supply will be possible at the end of this year or early next year. The domestic release of Eli Lilly's Mounjaro, which is considered a strong competitor of Wegovy, is also imminent. The Ministry of Food and Drug Safety recently completed a safety and efficacy review on Mounjaro. Completing the review means that the product approval process will soon begin. Mounjaro is a GLP-1 analog, just like Saxenda and Wegovy. After obtaining approval as a type-2 diabetes treatment, Lilly is trying to expand its indications for obesity. In the case of the Mounjaro, in addition to the mechanism acting on the GLP-1 analog, its mechanism also acts on the glucose-dependent insulinotropic polypeptide (GIP). Due to this, Mounjaro's weight loss effect was better than that of Wegovy in each drugs' clinical trials. Lilly also entered a phase 3 clinical trial comparing the effects of Mounjaro and Wegovy on a one-to-one basis.
- Company
- Interleukin inhibitors Cosentyx·Tremfya busy extending reim
- by Eo, Yun-Ho Jun 05, 2023 05:37am
- News of reimbursement extensions granted for interleukin inhibitors is continuing in Korea. According to industry sources, the scope of reimbursement for Novartis Korea's IL-17A inhibitor 'Cosentyx UnoReady Pen' 300mg/2mL, and Janssen Korea's IL-23 inhibitor ‘Tremfya (guselkumab)' have been extended since last month, and the 1st of this month, respectively. As with the other doses of Cosentyx, its 300mg dose can also be prescribed to patients with moderate-to-severe plaque psoriasis, active and psoriatic arthritis (PsA), adults with active ankylosing spondylitis (AS), adults with severe ankylosing spondylitis, etc. In Korea, Cosentyx is reimbursed for patients with chronic severe plaque psoriasis that lasts for more than 6 months, and ▲10% of the total skin area has been affected, ▲has a PASI 10 or higher, ▲has shown no response to methotrexate (MTX) or cyclosporine when administered for more than 3 months or cannot continue treatment due to side effects, or ▲has shown no response to PUVA or UVB therapy when treated for more than 3 months or cannot continue treatment due to side effects. The reimbursement approval was made based on the MATURE trial. The trial results showed that 95.1%/75.6%/43.9% of patients that received Cosentyx 30mg reached PASI 75/90/100 each, and demonstrated better efficacy over its placebo. In the case of Tremfya, the drug will be granted reimbursement for palmoplantar pustulosis from this month. Under the new reimbursement standards, the drug may be used with insurance benefits from June 1st in patients aged 18 and older with moderate-to-severe palmoplantar pustulosis, ▲whose PPPASI is 12 or higher and has shown no response to therapeutic doses of acitretin, methotrexate, or cyclosporin when administered for more than 3 months or cannot continue treatment due to side effects, or ▲who has shown no response to phototherapy when treated for more than 3 months or cannot continue treatment due to side effects. Palmoplantar pustulosis is characterized by pustular blisters accompanied by erythema on the skin of the palms and soles that turn into brown scales, and then become dry, thick, and cracked. When the condition persists, the patient’s nails may be deformed and even fall out. The condition interferes with the patient's daily life because the condition causes severe itching and pain. As it is difficult to distinguish palmoplantar pustulosis from other skin diseases such as fungal infections, accurate differential analysis between the two is necessary. Cosentyx 300mg was approved on November 1st, 2022 to treat ▲plaque psoriasis patients and ▲psoriatic arthritis (PsA) patients who have accompanying moderate-to-severe plaque psoriasis or have not adequately responded to an anti-TNAα treatment before. Meanwhile, Tremfya was approved as a treatment for adult patients with plague psoriasis in April 2018 and as a treatment for adult patients with palmoplantar pustulosis in May 2019, and the indications applied for insurance benefit in September 2018 and May 2021 respectively.
- Company
- Forxiga will maintain its price for 10 months in KOR
- by Jung, Sae-Im Jun 02, 2023 05:38am
- The prices of AstraZeneca’s SGLT-2 inhibitor ‘Forxiga (dapagliflozin)’ and combination drug ‘Xigduo’ will stay as is until February next year. On the 1st, the Seoul Administrative Court’s 1st Administrative Division accepted the suspension of execution filed by AstraZeneca against the Ministry of Health and Welfare to cancel the drug price cut disposition for Forxiga and Xigduo. With the court’s ruling, Forxiga and Xigduo will be able to maintain their price until February 29, 2024. The litigation date for the merits of the lawsuit has not been set yet. The SGLT-2 inhibitors Forxiga and Xigduo are blockbuster diabetes treatments that contain dapagliflozin which has been raising KRW 90 billion in outpatient prescriptions a year. The two products were subject to price cuts due to the entry of many dapagliflozin generics in April. With the listing of their generics, the government had announced that it will make an ex officio adjustment to the prices of Forxiga and Xigduo, reducing it by 30% as of May 1st. In objection, AstraZeneca filed an administrative lawsuit and at the same time applied for suspension of execution. As Forxiga and Xigduo also have indications for chronic heart failure and chronic kidney disease that for which the patent had not expired yet, the company argued that the listing of generics that only have the diabetes indication cannot serve as a basis for lowering the price of the original drug. The company also claimed that the damages incurred from the non-acceptance of the suspension of execution will be irrecoverable. In April, the court temporarily suspended the enforcement of drug price cuts for the two products and extended the stay of execution by 2 more after examining the suspension of execution on the 16th, until a decision was made on whether to accept the claim. As the court ruled to suspend execution on the 1st, AstraZeneca Korea will now able to continue its main lawsuit while maintaining the current price for Forxiga and Xigduo. During the 10-month term the suspension of execution is applied, AstraZeneca Korea will be able to prevent a loss of about KRW 23 billion (based on UBIST) that could have risen due to drug price cuts. If the main lawsuit is not ruled upon by February 29, 2024, the suspension of the execution period may also be extended further.
- Company
- BMS and Gilead compete tightly in HBV drug market in KOR
- by Nho, Byung Chul Jun 02, 2023 05:38am
- BMS’s Baraclude and Gilead Science’s Viread are fiercely competing in the KRW 200 billion original hepatitis B treatment market. Based on drug distribution results, sales of Baraclude and Viread, the No.1 and No.2 products in the HBV market in 2022, were tallied at KRW 69.5 billion and KRW 62.8 billion, respectively. Just last year, Baraclude’s sales slightly surpassed that of Viread, however, in 2018, 2019, and 2021, Viread (KRW 116.7 billion, KRW 83 billion, KRW 63.1 billion) posted higher sales than Baraclude (KRW 70 billion, KRW 69.8 billion, KRW 62.1 billion). However, Gilead Sciences' steep rise in sales of its total corporate product lineup of HBV products is noteworthy. The company also owns Vemlidy, which posted sales of KRW 34.9 billion last year. When combining sales of Viread and Vemlidy, the two drugs occupy 55.9% of the total market share with KRW 97.7 billion, far ahead of Baraclude’s 39.8%. The 4th to 8th in line, GSK’s Zeffix, Ildong Pharamceutical’s Besivo, GSK’s Hepsera, Novartis’s Sebivo, and Bukwang Pharmacuetical’s Levovir made sales of KRW 3 billion, KRW 1.7 billion, KRW 1 billion, KRW 1 billion, and KRW 600 million, respectively, occupying 0.4% to 1.8% share of the market. Sales performance of all of the lower-ranking product groups, excluding Besivo, had been on a range-bound downward slope for the past 5 years. Sales of Besivo, a new homegrown drug introduced in Korea, on the other hand, have been growing steadily. In 2018, 2019, 2020, 2021, and 2022, the drug sold KRW 400 million, KRW 900 million, KRW 1.2 billion, KRW 1.5 billion, and KRW 1.7 billion, and is expected to settle as a new growth engine for Ildong Pharmaceutical in the future. Meanwhile, Baraclude (entecavir) 1mg and 0.5mg is indicated for the treatment of chronic hepatitis B virus (HBV) infection in adults (aged 16 or older) and pediatric patients aged 2 years or older with evidence of active viral replication, persistently elevated serum alanine aminotransferase (ALT) levels and histological evidence of active inflammation and/or fibrosis. Viread and Vemlidy Tab (tenofovir disoproxil fumarate) are indicated for the ‘treatment of chronic hepatitis B and HIV 1 infection in adults and pediatric patients 12 years or older,’ and ‘treatment of chronic hepatitis B in adults,’ respectively. Zeffix (lamivudine), Besivo (Besifovir dipivoxil), Hepsera (adefovir dipivoxil), Sebivo (telbivudine), and Levovir (clevudine) are indicated for the treatment of chronic hepatitis B infection. Baraclude is a guanosine nucleoside analog with specific activity against HBV DNA polymerase that inhibits the synthesis of the HBV DNA positive strand. It has demonstrated a long-term treatment effect and safety with a high virus suppression effect and low resistance. Viread’s mechanism of action is similar to adefovir, a nucleotide analog, but has a much stronger antiviral effect, and has achieved 0% resistance until recently. Vemlidy is an HBV treatment that Gilead released as an upgraded version of ‘Viread (tenofovir).’ It contains the same active ingredient, tenofovir, but comes in a 1/10 dose, demonstrating improved safety with comparable efficacy to the existing product. Meanwhile, GSK’s adefovir dipiboxil hepatitis B treatment Hepsera Tab., which currently maintains an insurance listed price of KRW 3,839, is set to be deleted from the reimbursement list on August 1st, and expected to be withdrawn from the Korean market then.
- Company
- K-Pharma Bio ASCO launch table
- by Hwang, Jin-joon Jun 01, 2023 05:43am
- Pipeline clinical results of major domestic pharmaceutical bio companies such as Yuhan Corp. were disclosed at ASCO. According to the industry on the 31st, the abstract of a paper scheduled to be presented at ASCO, which will be held in Chicago, USA from June 2nd to 6th (local time), was recently released. ASCO is a major international academic conference that celebrates its 59th this year. About 400 biopharmaceutical companies from all over the world participate every year. There are more than 2,900 paper abstracts published this year. Most of the research results are made public in the form of posters. At this ASCO, Yuhan Corp. and GC Cell's US affiliates Artiva, Genexine, AbClon, and Abion disclosed research results of major pipelines under development. Four studies related to Yuhan Corporation's third-generation lung cancer drug Lazertinib were announced. Lazertinib is an oral third-generation EGFR tyrosine kinase inhibitor (TKI) drug with high selectivity for EGFR T790M-resistant mutations. Some details of clinical trials using Amivantama and Lazertinib in combination with EGFR mutation advanced non-small cell lung cancer (NSCLC) with no treatment experience were disclosed. These are the results of circulating tumor nucleic acid (ctNDA) liquid biopsy using plasma samples and long-term follow-up of 20 patients as of November 15 last year. Median follow-up and treatment periods were 33.6 and 33.5 months, respectively. In terms of efficacy, PFS and OS could not be estimated. At 12 months, 85% (n = 17) of the patients showed the expected PFS. At 24 months and 36 months, it was 65% (13 people) and 51% (10 people), respectively. 50% of patients are continuing treatment with PFS. In phase 2 clinical trial for NSCLC patients with brain metastasis, Lazertinib showed an intracranial objective response (iORR) in 22 out of 38 patients. Treatment-related side effects were identified in 85% (n = 27) of patients. The most common side effects were skin rash and paresthesia. Grade 3 or higher adverse events were reported in 10% (3 patients). Grade 3 or higher side effects are side effects that could potentially endanger the patient's life if not treated. Professor Min-hee Hong of Yonsei Cancer Center explained, "This study means that using Lazertinib instead of topical treatment can be a potential treatment strategy for NSCLC patients with brain metastases." The results of a study to identify biomarkers for the treatment of Amivantamab and Lazertinib in a tumor environment after administration of Osimertinib, a major third-generation lung cancer drug, were also disclosed. "The MET mutation identified by tissue immunohistochemical analysis (IHC) can be a predictive biomarker for the response to Amivantamab/Lazertinib administration after Osimertinib administration," said Professor Benjamin Besse and others at the Gustave Roussy Cancer Center in France. Analysis using tumor nucleic acid (ctDNA) did not identify some groups." Clinical results were also announced comparing the incidence of venous thromboembolism (VTE) side effects in EGFR mutant NSCLC patients with the combined administration of Amivantamab and Lasertinib and single administration of each drug. VTE is one of the common side effects of lung cancer patients. In a study of 540 patients, the incidence of VTE was higher with combination therapy than with each monotherapy. Artiva, a US affiliate of GC Cell, announced the first human administration data for 'AB-101', a CAR-NK cell therapy. These are the results of a phase 1/2 clinical trial of monotherapy of AB-101 and the combination of AB-101 and the anticancer drug 'Rituximab (product name: Rituxan)' in patients with relapsed/refractory B-cell non-Hodgkin's lymphoma. AB-101 responded in 3 out of 4 patients who had previously failed CD19-targeted CAR-T treatment. In the AB-101 monotherapy group, ORR was 27% (3/11 patients). Genexine disclosed the results of phase 2 clinical trial led by head and neck cancer researchers on the triple combination therapy of 'GX-188E', a DNA vaccine for the treatment of cervical cancer, 'GX-I7', and 'Keytruda', candidates for immuno-anticancer drugs for lymphopenia. This clinical trial was led by Professor Kim Hye-ryeon of the Department of Oncology at Severance Hospital. The study was conducted on 11 patients with head and neck cancer who were about to undergo surgery. After surgery, 63.6% (7 patients) showed MPR and 36.3% (4 patients) showed pCR. AbClon disclosed the efficacy and safety of the CAR-T treatment 'AT101' confirmed in phase 1 clinical trial. In this phase 1 of 18 patients with relapsed or refractory B-cell non-Hodgkin's lymphoma, an ORR of 66.7% (4/6 patients) was confirmed in subject group 1. AT101 showed an ORR of 100% (3/3 patients) in phase 1 clinical trial subject group 2. There was no cytokine release syndrome of grade 3 or higher in each subject group. Abion presented the results of the pilot expansion cohort analysis of NSCLC's new drug candidate 'ABN401'. The pilot expansion cohort is a clinical trial designed to predict the direction of phase 2 clinical trial after the phase 1 clinical trial. Patients with c-MET mutation NSCLC were selected and administered the same dose of ABN401 as in the ongoing phase 2 clinical trial. Through data analysis, the efficacy, safety, and tolerability of ABN401 were confirmed. No adverse events of grade 3 or higher were observed.