- LOGIN
- MemberShip
- 2026-05-01 13:13:43
- Policy
- Non-inferior drugs likely to receive preferential pricing
- by Lee, Tak-Sun Dec 15, 2023 05:51am
- Non-inferior drugs from Korea innovative pharmaceutical companies will likely be eligible for preferential drug pricing starting next year. Previously, non-inferior drugs were priced below the weighted average of alternative drugs. However, through improvement measures, if the patent of an alternative drug is still active, non-inferior drugs are now likely to be priced within a range between the weighted average price and the highest price of alternative drugs. According to the industry, the government and the pharmaceutical industry held a meeting for ‘Measures to Recognize the Appropriate Value of Innovative Drugs,’ and shared these updates. The improved measure states that if a drug of a Korea innovative pharmaceutical company receives approval through the Ministry of Food and Drug Safety (MFDS)'s GIFT system (Global Innovative Products on Fast Track, GIFT), it will be included and be applied the existing preferential treatment measures used during reimbursement review. The existing preferential treatment measures that will be used during reimbursement review for the GIFT drugs refer to the article, ‘1.7. Medicines in need of preferential treatment when considering its effects on public healthcare,‘ in the Health Insurance Review and Assessment Service (HIRA)’s ‘Specific evaluation criteria for new drugs and medicines subject to pricing negotiations.’ Specifically, the existing criteria contain preferential drug pricing measures for essential medicines endorsed by the WHO or national essential medicines, globally first-approved innovative drugs, and cell therapy products that satisfy the criteria. If the candidate drug’s clinical utility was similar to (or non-inferior to) an alternative, its price was set as ▲either an amount between the weighted average price and the highest price of the alternative drugs, or ▲the weighted average price of its alternative multiplied by (100/53.55), whichever was lower. The criteria has now been expanded to include drugs from Korea innovative pharmaceutical companies approved through the GIFT system. Whereas previously, non-inferior drugs from these companies were priced below the weighted average price if the alternative drug's patent had not expired, they can now be potentially priced at an amount between the weighted average price and the highest price of the alternative drugs. Since most new drugs developed domestically are non-inferior drugs from Korea innovative pharmaceutical companies, these homegrown new drugs will likely receive better drug prices in the future. Meanwhile, it has been reported that the agenda of including natural product-derived new drugs like cell therapy products as a subject to the preferential measures is set to be discussed in the future. Although this item was omitted from discussions in the meeting held on the 12th, it might be included again as an agenda for the Health Insurance Policy Deliberation Committee's subcommittee, which is scheduled to meet on the 14th. Additionally, it has been reported that for new drugs meeting the criteria for innovative drugs, there will be flexible application of the ICER (Incremental Cost-Effectiveness Ratio), and implementation of a risk-sharing plan to provide eased measures such as removing the final 3 negotiations if a drug has already received 3 three PVA negotiations during the past five years. The improvement measures are scheduled to be reported at the meeting of the Health Insurance Policy Deliberation Committee planned for the 20th, and the measures are expected to be sequentially applied from next year.
- Policy
- Will Enhertu be reimbursed using flexible ICER threshold?
- by Lee, Tak-Sun Dec 14, 2023 05:47am
- With the government reportedly planning to flexibly apply the Incremental Cost-Effective Ratio (ICER) threshold, whether the breast cancer treatment Enhertu (fam-trastuzumab deruxtecan, Daiichi Sankyo) will finally find its way to reimbursement is gaining attention in Korea. The drug’s reimbursement review is currently stuck in HIRA’s pharmacoeconomic evaluation stage. The drug was unable to pass the stage due to the longer period of administration, which rather increased drug cost, despite its significantly better effect than existing drugs. Therefore, a more flexible application of the ICER threshold during pharmacoeconomic evaluation is expected to allow Enhertu’s passage. According to the industry on the 13th, a plan to flexibly apply the ICER threshold for innovative new drugs in consideration of their innovative value was shared at the public-private consultative body meeting for 'Measures to recognize the appropriate value of innovative new drugs’ that was held on the 12th. The current ICER threshold is flexibly used to evaluate the cost-effectiveness of drugs in consideration of the severity of the disease, impact on quality of life, and innovativeness of a new drug. As the criteria used to evaluate the economic feasibility of a new drug with an improved effect, it determines the additional cost spent by a new drug compared to its comparator for the improved efficacy or each unit of use. If a drug’s ICER threshold is less than or equal to a certain level, the drug is interpreted as being cost-effective to its comparator. In general, the level is set at around KRW 50 million for anti-cancer and rare disease drugs and around KRW 30 million for other general drugs. Enhertu’s price had reportedly exceeded the ICER threshold of KRW 50 million. The industry believes that if the threshold is flexibly applied according to the characteristics of each drug, it could serve as a breakthrough for Enhertu’s reimbursement which is stuck in the pharmacoeconomic evaluation process. However, concerns have been raised about the strict standards set on which innovative new drugs may receive flexible application of the ICER threshold. To be eligible for flexible application of the ICER threshold, a new drug must meet all of the following criteria: ▲ has a new mechanism of action or substance, ▲ has no alternative treatments (including drugs) available, ▲ has shown clinically significant improvement, such as a significant extension in survival, ▲ received a Breakthrough Therapy Designation (BTD) from the U.S. FDA or approved as a Priority Medicines (PRIME) by the European Medicines Agency (EMA), and ▲ is a treatment for a rare disease or an anti-cancer drug. Enhertu must also meet all of the criteria above to receive preferential treatment in its pharmacoeconomic evaluation process. The industry believes Enhertu can meet the criteria for innovative new drugs and receive preferential treatment. Although Enhertu’s reimbursement passed the Health Insurance Review and Assessment Service (HIRA) Cancer Disease Deliberation Committee’s review after redeliberation in May and set reimbursement standards, it has been unable to pass the Drug Reimbursement Evaluation Committee evaluation ever since. The agenda is said to be stuck in the pharmacoeconomic evaluation stage.
- Policy
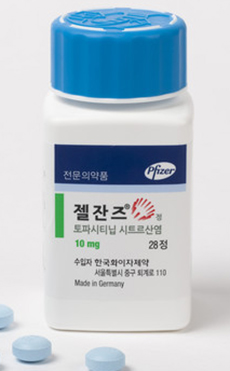
- Only CKD and Jeil have high-dose Xeljanz generics approved
- by Lee, Hye-Kyung Dec 13, 2023 05:38am
- Pfizer Jeil Pharm received approval for its ‘Topazan Tab 10mg,’ a high-dose generic version of the rheumatoid arthritis treatment ‘Xeljanz (tofacitinib citrate).’ With the Ministry of Food and Drug Safety’s approval of Topazan Tab 10mg on November 11 in addition to the Topazan Tab 5mg that was approved on November 27, Jeil Pharm now owns both low- and high-dose generic versions of Xeljanz. Following Boryung, which received approval for ‘Boryung Tofacitinib TAb 5mg (tofacitinib aspartate) as the first Xeljanz generic in August 2020, 58 generics have been approved to date, but Chong Kun Dang is the only company that received approval for both the 5mg and 10mg doses. Reasons include safety concerns and reduced indications associated with the higher dosage formulation, and even the outpatient prescription amount of the original shows this clear difference in demand between the low and high doses. The low dose is approved for five indications including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, ulcerative colitis, polyarticular juvenile idiopathic arthritis (pJIA), and juvenile psoriatic arthritis, whereas the indication for the high dose formulation is limited to ulcerative colitis. In addition, post-marketing surveillance studies of Xeljanz have shown that the drug is associated with an increased risk of blood clots in ulcerative colitis. The MFDS also added a warning to the drug’s label in 2020, cautioning healthcare providers to evaluate the risk of thrombosis in patients with ulcerative colitis and to cautiously use Xeljanz 10mg in patients with risk factors for thrombosis. With persisting safety concerns over the higher dose, according to the market research institution IQIVA, out of the KRW 15.9 billion sales of Xeljanz in 2021, the 5mg dose accounted for about KRW 13 billion, while the 10mg dose accounted for only a mere KRW 2.1 billion. Meanwhile, Xeljanz owns a substance patent that expires in November 2025 and a crystalline patent that expires in November 2027. 20 companies, led by Chong Kun Dang, have tried to overcome it by filing an invalidation trial, and 16 companies including Boryung Pharmaceutical, have tried to avoid the patents by filing a passive scope of patent rights trial. As a result, generic companies succeeded in avoiding Xeljanz’s patent in January 2018, and then won the invalidation trial in November 2019, thereby removing the crystalline patent from the green list in March 2020. As a result, Xeljanz generics that are being granted marketing authorization will be launched after November 2025, when the substance patent expires.
- Policy
- KIDS ‘Oral VEGFR-TKI use may increase risk of AAD’
- by Lee, Hye-Kyung Dec 12, 2023 05:38am
- The Korea Institute of Drug Safety & Risk Management (President: Jeong Wyan Oh, KIDS) announced that the use of Vascular Endothelial Growth Factor Receptor-Tyrosine Kinase Inhibitor (VEGFR-TKI) was associated with an increased risk of aneurysm and artery dissection occurrence. The findings were published in the internationally recognized American Medical Association (AMA) journal, JAMA Network Open (IF=13.8) on November 29. Recently, the U.S. FDA identified an association between VEGF-TKI inhibitors such as sorafenib and pazopanib and the development of aneurysms and arterial dissection through an analysis of the FDA Adverse Event Reporting System (FAERS) data and reflected the results in the indication of identified drugs. Korea’s Ministry of Food and Safety also reflected the association in the indications of affected drugs after conducting a safety information review. The MFDS conducted a study to investigate the risk of aneurysm and arterial dissection associated with the use of VEGFR-TKIs in the Korean population, after deeming that prior epidemiologic studies were lacking in Korea. The study collected information on 127,710 cancer patients aged 40 years or older who were prescribed VEGFR-TKIs or capecitabine based on the National Health Insurance Service’s insurance claims data (2007-2020) and followed them for 1 year after use. Among patients receiving VEGFR-TKIs, the incidence of AAD within 1 year of treatment initiation was 6.0 per 1000 person-years (1,000 subjects observed for 1 year). The incidence of AAD in those receiving capecitabine was 4.1 per 1000 person-years, showing that the risk of AAD occurrence was significantly higher by 1.48 times (95% CI, 1.08-2.02) among patients prescribed VEGFR-TKIs than those receiving capecitabin. The incidence and risk of AAD were significantly higher among patients treated with VEGFR-TKIs than those treated with capecitabine in females (2.08 times), older adults aged 65 years or older (1.42 times), and patients with dyslipidemia (1.58). KIDS President Oh said, “The study holds significance as that it demonstrated the possibility of an association between aneurysm and arterial dissection and the use of VEGFR-TKIs in cancer patients in Korea. This is expected to not only increase the treatment effect but also reduce socioeconomic losses and contribute to building a safer drug use environment."
- Policy
- MOHW agrees to review current prior authorization system
- by Lee, Jeong-Hwan Dec 12, 2023 05:38am
- Director Chang-hyun Oh Medical professionals are raising concerns about significant disparities in approval rates of the 'rare diseases drug prior authorization system,' which differ by patients and by diseases, and the ambiguity of the criteria. The government appears to be failing to provide a direction for system improvement and address these concerns. However, the government also agreed that significant disparities do exsit in the approval rates of prior authorization by diseases and by medical institutions. Thus, it has expressed plans to improve the approval rate through case-by-case feedback. On 11th, Oh Chang-hyun, Director of Pharmaceutical Benefits at the Ministry of Health and Welfare, spoke at the National Assembly roundtable discussion for the institutional improvement of the rare disease drug prior authorization system, saying, “We’re considering various systemic improvements to enhance patient access to high-cost drugs, and investigating why approval rates vary for the same drug in different diseases." The low approval rate of Soliris (eculizumab) for aHUS (atypical hemolytic uremic syndrome) sparked this issue of disparity in the reimbursement approval rate following prior authorization review. aHUS is a severe genetic rare disease. Up to 79% of patients die, require dialysis or experience permanent renal damage within 3 years of diagnosis. The prior authorization system allows reimbursement eligibility decisions to be made for each individual patient prior to treatment so that high-priced rare diseases treatments can be provided to patients in need. Individual cases are determined by an expert prior authorization subcommittee. The review started in 1992 and was expanded to include medications in 2007. The approval rate for Soliris, a treatment for aHUS (atypical hemolytic uremic syndrome), is currently below 30%. In contrast, the approval rate for the same drug for Paroxysmal Nocturnal Hemoglobinuria (PNH) is around 90%. Medical professionals and patients have consistently raised concerns that the disparities in prior authorization rates for the same medication by different diseases and by medical institutions, are causing difficulties in patient treatment. In response, Shin Hyun-Young, a member of the Democratic Party of Korea, hosted a symposium to discuss improvements to the prior authorization system, gathering opinions from government agencies and experts. Oh Chang-hyun, Director of Pharmaceutical Benefits at Ministry of Health and Welfare, who attended the symposium said that he is aware of the concern regarding the disparity in pre-approval rates, and is committed to finding solutions to the issue. Director Oh revealed that out of the 18 new drugs approved this year (2023), only two are subject to the prior authorization system. He explained, "The decision was not due to financial reasons, but because of the high cost, uncertainties, and misuse concerns. The Health Insurance Review and Assessment Service (HIRA) is working on an improvement plan." "The prior authorization rate for most drugs is above 80%, but in the case of Soliris, its approval rate last year for PNH was 90.7%, while being below 50% for aHUS," said Director Oh, adding, “There is a need to scrutinize why there are disparities in approval rates by different diseases and to strictly enforce compliance requirements for medical institutions seeking approval.” Director Oh stated, “Approval rates would improve if we conduct a statistical analysis on the reasons behind the many rejections that were made, and providing this feedback to individual medical institutions and doctors." Oh stressed, “The reliability of the data will go up during the process, and the Ministry is considering transitioning drugs with a long-term stable approval rate from prior approval to post approval.” Likewise, the MOHW acknowledged the problem regarding disparities in the prior authorization system rates, but hadn't yet established a specific and finalized improvement plan. However, improvements are expected soon, as the Ministry is working to address the issue quickly in collaboration with HIRA. Panelist Choi Seungwon, Director of Doctors News, suggested that the MOHW should create and reinforce a channel for clear communication and discussion with medical professionals and patients about the reasons for rejections. Director Choi said, “Patients should not suffer due to systemic uncertainties. The prior authorization system should be based on the principle that it’s better that nine guilty ones escape, rather than let one innocent suffer,” he stressed, “It’s the doctor’s duty to their patients to provide an explanation. The government should be able to explain how it will communicate its decisions in cases where medical professionals and patients find rejection decisions unacceptable." "A system that specifies which person and method of inquiry should be used to inquire about the government’s disapproval decision, is necessary and must be included as part of the prior authorization system," said Director Choi Seungwon, adding, “We ask Director Oh to preprare this."
- Policy
- Roche’s DLBCL treatment Columvi approved in Korea
- by Lee, Hye-Kyung Dec 11, 2023 05:02am
- Commissioner Oh Yu-Kyoung of the Ministry of Food and Drug Safety (MFDS) announced on the 7th that MFDS has approved Roche Korea's rare disease drug 'Columvi (glofitamab)' for the treatment of diffuse large B cell lymphoma (DLBCL). The drug is a CD20/CD3 bispecific monoclonal antibody that binds to CD3 on the surface of T cells and CD20 on B cells. Columvi provides a new treatment opportunity for DLBCL patients who had relapsed or were refractory after two or more lines of systemic therapies. Columvi (Glofitamab) binds to the CD3 and activates the T cells, it simultaneously binds to CD20, to redirecting B cells closer to activated T cells and inducing cytosis. The MFDS stated that based on their regulatory science expertise, they will continue to promptly supply treatments that have proven safety and effectiveness.
- Policy
- Pfizer conducts trial for its RSV drug sisunatovir in KOR
- by Lee, Hye-Kyung Dec 11, 2023 05:02am
- Pfizer will be conducting a clinical trial for its Respiratory Syncytial Virus (RSV) treatment PF-07923568 (sisunatovir) on infants and children in Korea. On the 7th, the Ministry of Food and Drug Safety approved Pfizer Korea's ‘Interventional, phase 1B, randomized, double-blinded, sponsor-open, placebo-controlled, multicenter, dose-finding study to evaluate the safety, tolerability, and pharmacokinetics of sisunatovir in pediatric participants up to 60 months of age with respiratory syncytial virus (RSV) lower respiratory tract infection (LRTI).’ The study will be conducted at Seoul National University Bundang Hospital, Severance Hospital, Seoul National University Hospital, and Korea University Anam Hospital. As a type of common cold, RSV is an acute respiratory infection caused by the infection of the respiratory syncytial virus. It is highly contagious, affecting nearly all children by the age of 2. Most children and adults recover within a week or two without treatment, but it requires attention as it can progress to severe cases in high-risk groups such as infants, those immunocompromised, and the elderly. According to the U.S. Centers for Disease Control and Prevention, RSV causes up to 120,000 hospitalizations and up to 10,000 deaths in the U.S. each year among those aged 65 and older. Currently, AstraZeneca's ‘Synagis’ is the only licensed antibody preventive treatment for RSV that can be administered to pediatric patients. Pfizer acquired sisunatovir, a novel candidate substance for RSV when it acquired the UK-based antiviral drug developer ReViral last year. Pfizer paid up to $525 million to ReViral, including an upfront payment and future development milestone payments. Pfizer expects ReViral’s programs to generate annual sales of $1.5 billion or more. Meanwhile, other large multinational pharmaceutical companies have also jumped into the RSV prophylaxis game, and are making tangible results. Just this year, the US FDA approved GSK’s ‘Arexvy,’ Pfizer’s ‘Abrysvo,’ and AstraZeneca and Sanofi’s pediatric RSV treatment ‘Beyfortus.’ In Korea, companies such as SK Bioscience and EUGiologics have been working to develop RSV vaccines.
- Policy
- First Depakote generic is being reviewed for reimb in KOR
- by Lee, Tak-Sun Dec 08, 2023 05:55am
- The first generic of Abbot Korea’s epilepsy drug ‘Depakote ER Tab’ is undergoing a reimbursement process in Korea. The drug is ‘Divalpro ER Tab 500mg,’ developed by Korea Pharma. The drug was first approved in Korea on November 8th. It is the first generic drug to contain divalproex sodium, the main active ingredient of Depakote ER. Depakote ER was approved in Korea in 2002. It was approved in two dosage forms, and the drug posted sales of KRW 10 billion in 2022 according to IQVIA. With no patents listed on the MFDS’s green list, there were no obstacles to the approval or release of its generic versions. However, due to difficulties in bioequivalence testing, only a few companies, including Korea Pharma, WhanIn Pharma, and Mirae Pharm, had been reportedly developing its generics. In the field, Korea Pharma became the first to receive marketing authorization and seize the first generic status in Korea, making a head start in the development race. The company applied for reimbursement immediately after receiving approval. As a result, HIRA has been reviewing whether Korea Pharma’s generic drug satisfies the reimbursement conditions as part of its drug pricing calculation process. The insurance ceiling price of the original drug, Depakote ER 500mg, is currently KRW 391. If Divalpro ER satisfies both requirements – directly completing bioequivalence tests and using a substance registered in the DMF – its price will be set at 53.55% of the price the original drug received before patent expiry. In Korea, generic drugs usually take 3 months from reimbursement to listing, so it is expected that the drug will be listed for reimbursement in February next year, after which the company will conduct full-scale marketing activities. The introduction of reimbursed generics means that the prescribing options will be expanded in the field. The drug is indicated as monotherapy and adjunctive therapy in complex partial seizures in adults and pediatric patients down to the age of 10 years, and in simple and complex absence seizures.
- Policy
- HIRA’s reimb reevaluation results to be released on the 7th
- by Lee, Tak-Sun Dec 07, 2023 05:47am
- The final results of the 2023 reimbursement adequacy reevaluations will be released on the 7th. On the day, the Health Insurance Review and Assessment Service’s Drug Reimbursement Evolution Committee will deliberate on the final reevaluation results that reflect the appeals made by pharmaceutical companies on the first results. In general, the pharmaceutical industry is not expecting the results to differ greatly from the first results, given the lack of appeals that were filed. However, in the case of hyaluronic acid eye drops, the committee discussed revising the reimbursement standards regarding the use amount after releasing the initial results, so industry eyes are on whether DREC will make a final decision on this on the 7th. The initial results announced at the end of DREC’s September 6 meeting concluded that rebamipid and levosulfiride were reimbursable. However, limaprostalpha-dex for improving ischemic symptoms of Berger's disease, such as ulcers, arterial pain, and coldness, was not considered to be adequate for reimbursement. Also, loxoprofen’s use to reduce fever and pain related to acute upper respiratory tract infection was not considered to be adequate for reimbursement. In the case of hyaluronic acid eye drops, which attracted the most attention, the committee deemed reimbursement inadequate for its use for exogenous diseases caused by surgery, drugs, trauma, or contact lens wear. Although its use for endogenous diseases such as Sjögren's syndrome, mucocutaneous ocular syndrome, and dry eye syndrome were deemed adequate, the committee determined that its reimbursement standards such as the number of prescriptions per patient visit and the total number of prescriptions per patient per year would need to be established for its appropriate use. The industry generally expressed pleasure about the initial results because the indications that were deemed non-reimbursable were not the drugs’ primary indications. However, as antipyretic and analgesic indications for loxoprofen have been heavily used during the COVID-19 pandemic, there was some pushback regarding its non-reimbursement in the medical community. In the case of the hyaluronic acid eye drops, there were fewer appeals from pharmaceutical companies because s its use for exogenous diseases is relatively low. The approved use for endogenous diseases accounts for the absolute majority of hyaluronic acid eye drop prescriptions, with over an 80% share. However, the industry is keeping a keen eye on restrictions that may be made on its use volume. Although there has been an opinion that its annual use should be restricted to 60 eyedrops (4 boxes), it has been reported that the committee had difficulty coming up with a unified plan due to strong opposition from the medical community and the pharmaceutical industry. Nevertheless, the industry prospects are that HIRA will report the results of its discussion to DREC to make a final decision. However, it is unlikely that DREC will come to an easy conclusion, therefore the reimbursement standards for restricting the volume is likely to be discussed beyond the year, according to industry analysis. If revisions to the reimbursement standards are postponed for this reason, it is expected that sodium hyaluronate eye drops’ reimbursement standards for exogenous diseases will first be removed and be granted reimbursement use only for endogenous diseases from January next year. An industry official said, "In the case of sodium hyaluronate eye drops, restricting its usage may greatly affect performance. We are waiting to see what decision DREC will make on this tomorrow."
- Policy
- Bill proposed to prioritize domestic drugs and vaccines
- by Lee, Jeong-Hwan Dec 06, 2023 06:00am
- A bill has been presented to prioritize purchases and use of domestic drugs and vaccines when Korea's national and local governments conduct drug stockpiling and National Immunization Programs (NIP). The bill also contains a provision to strengthen Korea's pharmaceutical sovereignty, including a plan to promote self-sufficiency of drugs in the Comprehensive Plan for Pharmaceutical Industry Development and Support. On April 24, Rep. Young-hee Choi, a member of the People Power Party presented a bill as representative to partially amend the Special Act on Fostering and Supporting the Pharmaceutical Industry. The current law requires the Minister of Health and Welfare to establish a Comprehensive Plan for Pharmaceutical Industry Development and Support every 5 years, which includes mid-to-long-term goals for fostering the pharmaceutical industry, to create a basis for industry development and strengthen its international competitiveness. However, the need to strengthen Korea’s drug sovereignty to enable domestic companies to develop and produce vaccines and essential medicines independently to overcome public health crises such as COVID-19 has been constantly raised since the COVID-19 outbreak. As competition among countries to foster the pharmaceutical industry is intensifying, the argument is that Korea also needs a policy to firmly establish its pharmaceutical sovereignty and rise to become a pharmaceutical powerhouse. For this, the revision presented by Rep. Choi includes amendments to provide grounds for the national government and local governments to prioritize the purchase of domestically produced drugs when conducting drug stockpiling or for Korea’s National Immunization Program (NIP), and to include a plan that can promote self-sufficiency of drugs in the contents of the Comprehensive Plan for Pharmaceutical Industry Development and Support. Rep. Choi said, "The Yoon Suk-yeol government has set the goal of making Korea one of the Top 6 global pharmaceutical and bio-pharma powerhouses by 2027. In the post-COVID-19 era, where the world is pursuing a “My nation first” policy for medicines, we hope that Korea will be able to achieve pharmaceutical sovereignty and emerge as a pharmaceutical and bio-pharma powerhouse by increasing the self-sufficiency rate of medicines.”