- LOGIN
- MemberShip
- 2026-04-25 01:06:18
- Policy
- Doctors "If INN prescribing is enforced, will boycott the SPD"
- by Kang, Shin-Kook Mar 12, 2026 08:34am
- Doctors who took to the National Assembly to protest the promotion of International Nonproprietary Name (INN) prescribing legislation have said they will go as far as a total boycott of the Separation of Prescribing and Dispensing (SPD) system if the bill is enforced.On the 11th at 4:00 PM, the Korean Medical Association (KMA) Special Committee for public health promotion held a rally in front of the National Assembly Main Building in Yeouido to block the INN prescribing legislation.The doctors gathered at the National Assembly held pickets reading "Will Declare SPD System Annulment if INN Prescribing is Enforced" and demanded an immediate halt to the legislative process.At the scene, Kim Taek-woo, KMA president, emphasized, "INN prescribing is not merely about selecting a chemical component. It is a highly sophisticated professional medical act performed after considering the patient's comprehensive condition. This irrational bill, which prioritizes pharmacy inventory over patient needs when dispensing medication, must be scrapped immediately."The Korean Medical Association (KMA) Special Committee for public health promotion held a rally in front of the National Assembly Main Building in YeouidoKim warned the government and the National Assembly, saying, "Do not trade the public's health for economic logic. While pharmacist groups mislead public opinion by citing ambiguous budget savings, no budget can be more precious than the lives of our citizens."Kim further stressed, "If INN prescribing is enforced, we will regard it as a unilateral breach of the Medical-Pharmaceutical-Government Agreement and declare the total nullification of the SPD system. If our right to prescribe is violated and patient safety is threatened, we will stake everything, including my presidency, on this struggle."Following him, Kim Kyo-woong, Chairman of the KMA representative council, pointed out, "INN prescribing is not simply a systemic change. It is an act that denies the medical judgment of physicians who have precisely diagnosed a patient's condition. Furthermore, it is a highly irresponsible idea that breaks the continuity of treatment and puts patients at risk."Lee Ju-byung, Chairman of the Special Committee to Oppose INN Prescribing, asked, "The new bill states that failure to comply with mandatory INN prescribing will result in up to one year in prison or a fine of up to 10 million KRW. Is this such a serious crime? Who proposed that a doctor should face imprisonment for not prescribing by ingredient name?Lee raised his voice, saying, "Some say doctors prescribe by brand name to receive rebates," adding, "Then, are you advocating for INN prescribing just to receive rebates yourselves?"Lee added, "If the government insists on mandating INN prescribing for the sake of the public, we doctors will, in accordance with the government's will, launch a movement to prescribe only original drugs solely for the benefit of the people."Park Jong-hwan, President of the Medical Association Council for 25 clinics in Seoul, also noted, "On one hand, they say we must implement INN prescribing for public convenience, but on the other hand, they want to forcibly close large-scale pharmacies that the public can use late at night and which offer a wide range of drug choices," adding, "Isn't this an act of inconsistency?"Park stated, "The legislation proposed by Rep. Jang Jong-tae is a self-contradiction," adding, "Both bills contain the words public and patient, but the actual public and patients are nowhere to be found. They only look toward the interests and votes of specific professional interest groups."Park concluded, "I do not know whether it is a National Assembly member's job to pass bills that give all dispensing authority to pharmacists through INN prescribing and protect the interests of small pharmacy owners by regulating large pharmacies, or if this is simply for the benefit of a specific group."
- Policy
- ‘Pricing reform should not be finalized without reporting to NA’
- by Lee, Jeong-Hwan Mar 11, 2026 08:29am
- Rep. Sunmin Kim of the Rebuilding Korea Party criticized the Ministry of Health and Welfare, saying it should not move forward with approving and implementing a drug pricing reform plan— which primarily involves lowering generic drug prices— by having it approved by the Health Insurance Policy Review Committee without reporting to the National Assembly.Kim effectively put the brakes on the ministry after it attempted to proceed with the March schedule for subcommittee and full meetings of the Health Insurance Policy Deliberation Committee (HIPDC) without including any mention of the drug pricing reform plan in its 2026 annual policy briefing to the National Assembly, despite the issue being of the utmost concern to the pharmaceutical industry.National Assembly Health and Welfare Committee Joomin Park of the Democratic Party of Korea also agreed with Kim on the need for a separate briefing on the reform plan. Addressing Health and Welfare Minister Eun Kyeong Jeong, Park said, “Because this is a very important issue, it would be appropriate to provide an additional briefing at the committee’s plenary meeting after concluding your discussions on the reform plan.”At the full committee meeting on the 10th, Minister Jeong responded affirmatively to Rep. Kim's procedural remarks and Chairman Park's request for an additional briefing on the drug pricing system reform plan, saying the ministry would comply.Consequently, it is highly likely that the MOHW will provide a separate briefing on the direction of the drug pricing system reform plan at the full committee meeting of the National Assembly's Welfare Committee this month (March), after subcommittee discussions and before the full committee vote.Currently, the ministry plans to hold a one-point HIPDC subcommittee meeting on March 11, followed by further discussion at another subcommittee meeting on March 18, before bringing the reform plan to the HIPDC plenary meeting on March 26 for approval. This plan includes lowering the pricing calculation rate for already listed generic drugs from the current 53.55% to the 40% range.The pharmaceutical industry has criticized the proposal as a mechanical, across-the-board “lawnmower-style” price cut, presenting 48% as their absolute bottom line for the generic drug calculation rate.With the gap between the MOHW and the pharmaceutical industry over the reform plan showing no signs of narrowing, Rep. Kim raised the issue through a procedural remark as the MOHW attempted to pass the plan without reporting it to the National Assembly.Rep. Kim stated, “After reviewing the MOHW's briefing materials, I question what the most pressing issue in current healthcare policy actually is. From what I understand, the drug pricing system reform is the topic most intensely discussed in the media and policy circles recently.”Kim continued, “I hear that tomorrow and next week, the HIPDC subcommittee will finalize the generic drug price reduction ratio. Following HIPDC deliberations at the end of March, discussions are proceeding with the goal of implementation next January. Yet, such an important policy report was not included among today’s key briefing items.”Kim added, “At this rate, there are concerns that the government could proceed with the drug pricing reform without reporting it to the relevant standing committee of the National Assembly. Therefore, I ask the Chairman to ensure that the MOHW clearly reports on the drug pricing system reform currently being pursued during today's briefing.”When Chair Park asked Minister Jeong whether she could provide an immediate briefing on the reform plan, Jeong replied, “We plan to hold about two more discussions in the HIPDC subcommittee to further coordinate opinions and gather more diverse input from the industry. As the proposal has not yet been finalized, we will review the progress and provide either a written report or a separate briefing.”In response, Park demanded that the MOHW prepare to provide an additional briefing at the committee’s plenary session, given the critical nature of the drug pricing system reform plan being pursued by the MOHW.Park stated, “Since additional processes and procedures remain (regarding the reform plan), individual briefings are fine, but because this is an extremely important issue, it would be better to provide an additional report at the committee’s plenary meeting once the discussions are complete.
- Policy
- MOHW agrees to relax convenience store medicine policy
- by Lee, Jeong-Hwan Mar 11, 2026 08:28am
- The Ministry of Health and Welfare (MOHW) has cast a vote in favor of an amendment to the Pharmaceutical Affairs Act that legislates the '20-item limit' on the number of safe household medicine items, currently prescribed by the Pharmaceutical Affairs Act, to a Presidential Decree (Enforcement Decree) to allow for lower-level legislation.The Ministry also expressed support for a provision that eases the '24-hour operation' requirement, which is a mandatory registration criterion for sales outlets, in eup, myeon, and dong where there are no pharmacies or safe household medicine sales outlets.In addition, the Ministry presented a specific proposal to delay enforcement by one year from implementation date to prepare for various matters related to institutional improvement, such as the establishment of subordinate statutes.The bill currently under discussion is facing significant backlash from the pharmacist community, as it could lead to the complete removal of regulations on the number of safe household medicine items in convenience stores and to the expansion of the sale of general medicines outside pharmacies. However, with the competent MOHW expressing support, the likelihood of its passage has increased.In addition to the MOHW, the Korean Society of Oriental Pharmacy, the Korea Alliance of Patients Organizations, the Consumers Union of Korea, and the Korea Association of Convenience Store Industry also supported the legislation. The Korean Pharmaceutical Association was the only organization to oppose the bill.On the 10th, the amendment to the Pharmaceutical Affairs Act, proposed by Representative Han Ji-ah of the People Power Party, is planned to be tabled at the plenary session of the National Assembly's Health and Welfare Committee.Rep. Han Ji-ah's stands that fixing the number of safe household medicine items in the Pharmaceutical Affairs Act is hindering the ability to respond flexibly and administratively to changes in the pharmaceutical market and environment, as well as to public demand.This is the reason for introducing a bill that provides a delegation provision, allowing the number of safe household medicine items to be determined by Presidential Decree.Rep. Han's proposal also includes a provision to establish the basis for the installation and operation of a Pharmaceutical Policy Deliberation Committee under the Minister of Health and Welfare.Easing the 20-item upper limit on safe household medicine items... Ministry supports, Pharmaceutical Association opposesThe Ministry supported the provision delegating the regulation of the 20-item limit for convenience store medicines, which is currently fixed in the Pharmaceutical Affairs Act, to a Presidential Decree.The MOHW also submitted a supportive opinion on relaxing the 24-hour operation requirement and the registration criterion for sales outlets on a limited basis in eup, myeon, and dong areas where there are no pharmacies or safe household medicine sales outlets, to improve accessibility to safe household medicines.However, the MOHW stated that the enforcement date should be adjusted to 1 year after promulgation to prepare for various matters related to institutional improvement, such as the establishment of subordinate statutes.The Korean Society of Oriental Pharmacy also agreed with the amendment to reorganize the safe household medicine system.The Korea Alliance of Patients Organizations and the Consumers Union also supported it. The Consumers Union also expressed the view that it is appropriate to determine the regulation on the number of items through an Enforcement Rule, an Ordinance of the Ministry of Health and Welfare, rather than a Presidential Decree.The Korea Association of Convenience Store Industry supported the bill while suggesting a modification that would require the MOHW to designate the number of household medicine items after receiving advice from a committee.The Korean Pharmaceutical Association strongly opposed. They pointed out that although various systems to supplement medically underserved areas, such as public late-night pharmacies, health clinics, and the designation of special locations, are already in place, the management and operation of safe household medicine sales outlets and special locations remain insufficient.The Korean Pharmaceutical Association argues that, rather than indiscriminately expanding the convenience store medicine system, the priority should be to evaluate whether the management systems of existing programs are functioning properly and to reorganize the system to ensure safe use.The Korean Pharmaceutical Association stated, "To resolve the pharmacy accessibility of residents in medically underserved areas such as farming and fishing villages, special locations can already be designated according to the Pharmaceutical Affairs Act, and a system is in place to allow the purchase of medicines through designated special locations. Currently, medicine is also accessible through 1,895 health clinics nationwide," and emphasized, "In particular, considering the increase in cases of acetaminophen poisoning, the lack of management of sales outlets and the increase in cases of non-compliance with requirements, and the trend of strengthening regulations abroad, we actively oppose the expansion of safe household medicine items at this point as it is a policy that directly contradicts national health."Establishment of Pharmaceutical Policy Deliberation Committee...Ministry of Health and Welfare, Ministry of the Interior and Safety, etc., all opposedThe MOHW and the Ministry of the Interior and Safety opposed the provision establishing a Pharmaceutical Policy Deliberation Committee, arguing that it would create an environment in which pharmaceutical-related matters, such as medicines, could be discussed regularly.The logic is that, since the Central Pharmaceutical Affairs Council exists and is established, installed, and operated by the MFDS, the establishment of an additional body is unnecessary.The Ministry of the Interior and Safety stated, "The Pharmaceutical Policy Deliberation Committee is an advisory committee under the MOHW, but considering the purpose of the Act on the Establishment and Operation of Committees Under Administrative Agencies, it is necessary to consider plans to expand the functions of the Central Pharmaceutical Affairs Council or utilize the policy advisory committees within the MOHW rather than establishing a separate committee."The Korean Pharmaceutical Association and the Korean Medical Association also submitted opposing views.The Korean Pharmaceutical Association opposed it, stating, "The role of the Pharmaceutical Policy Deliberation Committee in the bill is managed by the Pharmaceutical Policy Division of the MOHW," adding, "The policy planning and drafting are performed by the Ministry's Division of Pharmaceutical Policy, while the Pharmaceutical Policy Deliberation Committee performs policy deliberation and advice. In practice, this would lead to discussing the same matter twice, causing inefficiency and making the responsibility and subject of policy decisions unclear."The Korea Medical Association said, "Since the Central Pharmaceutical Affairs Council (CPAC) is already running, establishing a separate Pharmaceutical Policy Deliberation Committee would be a redundant installation of a committee performing similar functions, which is a waste of administrative power," adding, "There are concerns that it could lead to role conflicts between committees and inefficiency in the policy-making process."
- Policy
- Contaminated COVID vaccines, national petition for special investigation
- by Lee, Jeong-Hwan Mar 10, 2026 08:55am
- Minister of Health and Welfare Jeong Eun KyeongFollowing confirmation by disease control authorities of negligence in the management of foreign substances in COVID-19 vaccines, a national petition has been filed requesting the appointment of a Special Prosecutor to investigate the allegations.With multiple opposition lawmakers raising accountability issues against Minister of Health and Welfare Jeong Eun Kyeong, attention has been drawn to the progress of the petition.On the 6th, a petition was field on the National Assembly's e-People service stating, "We request the introduction of a Special Prosecuter to clearly investigate the foreign substance issues reported during the COVID-19 vaccine management process and the appropriateness of the administrative response."The petitioner demanded a Special Prosecutor investigation into the entire process of reporting and responding to foreign substances in COVID-19 vaccines. The request includes verifying the legality of reporting, recall, and quality control procedures; determining accountability; and conducting a full-scale investigation into potential external pressure or conflict-of-interest involvement.Furthermore, the petitioner urged the National Assembly to disclose the full results of the investigation, improve systems to prevent recurrence, and restore the public's right to know and trust in public health.According to the 'Diagnostics and Analysis of COVID-19 Response Status' report released by the Board of Audit and Inspection of Korea (BAI) on the 23rd of last month, the Korea Disease Control and Prevention Agency (KDCA) failed to notify the Ministry of Food and Drug Safety (MFDS) of foreign substance reports. Instead, they handled the issue by informing only the manufacturers and did not suspend vaccination with the same batch numbers despite the risk.Specifically, between March 2021 and October 2024, the KDCA received 1,285 reports of foreign substances in COVID-19 vaccines from medical institutions but bypassed the MFDS, opting to receive investigation results directly from manufacturers.While the majority of these cases (835, 65%) involved rubber stopper fragments due to usage errors, 127 cases (9.9%) involved hazardous foreign substances, including mold, hair, and silicon dioxide.Notably, because no suspension of administration was ordered for the vaccines containing hazardous substances, approximately 14.2 million doses from those specific batch numbers continued to be administered even after the reports were made.Based on the BAI announcement, the petitioner argued, "There is a need for an independent verification of potential deficiencies in the management and reporting systems, and whether the actions of the KDCA and MFDS complied with relevant laws and manuals," adding, "An objective investigation is also required regarding whether external pressure or conflict-on-interest interests influenced policy decisions during the reporting and management process."The petitioner further asserted, "It must be clarified whether the administrative agency's judgments were made based on public health principles or influenced by specific organizations or companies. Given that this matter directly affects national health, it is difficult to restore trust solely through internal executive branch investigations. The facts and accountability must be clarified through an investigation independent of political interests."Separate from the national petition, the opposition party is holding the Minister of Health and Welfare accountable for the contaminated COVID-19 vaccines.The People Power Party (PPP), citing the BAI report that contaminated vaccines were administered due to management failure during the pandemic, requested that Rep. Choo Mi-ae (Democratic Party), Chairperson of the Legislation and Judiciary Committee, cooperate in holding an emergency inquiry.Shin Dong-wook, Senior Supreme Council Member of the PPP, pointed out during a Supreme Council meeting on the 5th, "We requested the Democratic Party to hold an emergency inquiry into the BAI at the Legislation and Judiciary Committee, but Chairperson Choo is refusing, stating without reason that the committee cannot be convened."Rep. Na Kyung-won, the designated opposition lead for the Legislation and Judiciary Committee, also posted on Facebook yesterday, "According to KDCA data, there were 485,576 reports of adverse reactions, 2,802 deaths, and 1,285 reports of foreign substances. However, vaccinations were not stopped." Rep. Na added, "I will demand an emergency meeting of the Legislation and Judiciary Committee, the immediate withdrawal of government appeals [in vaccine lawsuits], the resignation of former Commissioner Jeong, and a parliamentary investigation."Rep. Kim Mi-ae, the PPP executive secretary of the Health and Welfare Committee, also stated at a PP meeting, "The status of COVID-19 vaccine management is a total failure that seriously threatens national health. It is a clinical experiment conducted by the state on innocent citizens." Rep. Kim urged Minister Jeong, who was the KDCA Commissioner at the time, to "take immediate responsibility, resign, and fully cooperate with the investigation."
- Policy
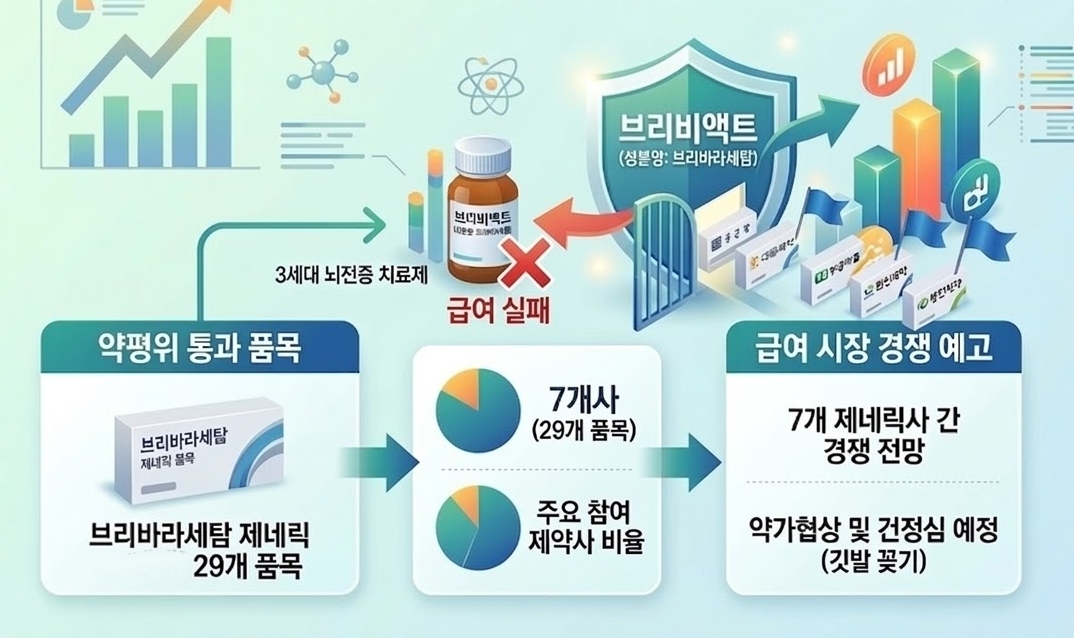
- Will Briviact become the next Vimpat? Generics near early listing
- by Jung, Heung-Jun Mar 10, 2026 08:55am
- Attention is focusing on whether generics will be listed first in the reimbursement market, in which the third-generation epilepsy treatment Briviact (brivaracetam) failed to enter.If 7 generic manufacturers, including Chong Kun Dang, Daewoong Pharmaceutical, and Samjin Pharmaceutical, secure reimbursement listing, competition among generic companies is expected in the reimbursed market without the original drug.Generic companies targeting Briviact’s vacancy are on the verge of securing reimbursement listing. AI-generated image.On the 5th, the Drug Reimbursement Evaluation Committee recognized the reimbursement adequacy of 29 epilepsy treatment products containing brivaracetam from 7 domestic pharmaceutical companies. Chong Kun Dang (Briveta Tab), Daewoong Pharmaceutical (Brivatop Tab), Bukwang Pharmaceutical (Bukwang Brifil Tab), Whanin Pharm (Briva Tab), Samjin Pharmaceutical (Bricetam Tab), Myung In Pharmaceutical (Buripam Tab), and Hyundai Pharmaceutical (Brilact Tab) are expected to proceed with listing procedures after price negotiations and review by the Health Insurance Policy Deliberation Committee. The original product is UCB Korea's Brivact, which received domestic approval in 2019. However, it failed to secure reimbursement listing when attempting to enter the insurance market and reportedly did not submit post-marketing surveillance (PMS) data last year. With its patent expiring last February, a series of generic approvals followed. Having passed the DREC hurdle, the generic versions are now on the verge of entering the reimbursement system.If price negotiations are finalized, a unique scenario could unfold where generics secure reimbursement listing before the original drug.Should generics gain early listing, it would mirror the case of UCB Pharma's other epilepsy treatment, ‘Vimpat (lacosamide)’.Vimpat, which was approved in Korea in 2010, ultimately failed to obtain reimbursement listing due to pricing issues, while generic versions of companies such as SK Chemicals’ Vimsk entered the reimbursement market first.According to the market research institution UBIST, Vimsk recorded sales of KRW 4.5 billion last year, marking 32% growth from KRW 3.4 billion the previous year. Its prescription performance has steadily increased over 4 years, from KRW 2.3 billion in 2021.The 7 companies preparing to list brivaracetam generics are also expected to attempt market entry using the same formula.The ultimate question is whether they can clear the drug price hurdle that the original failed to overcome.
- Policy
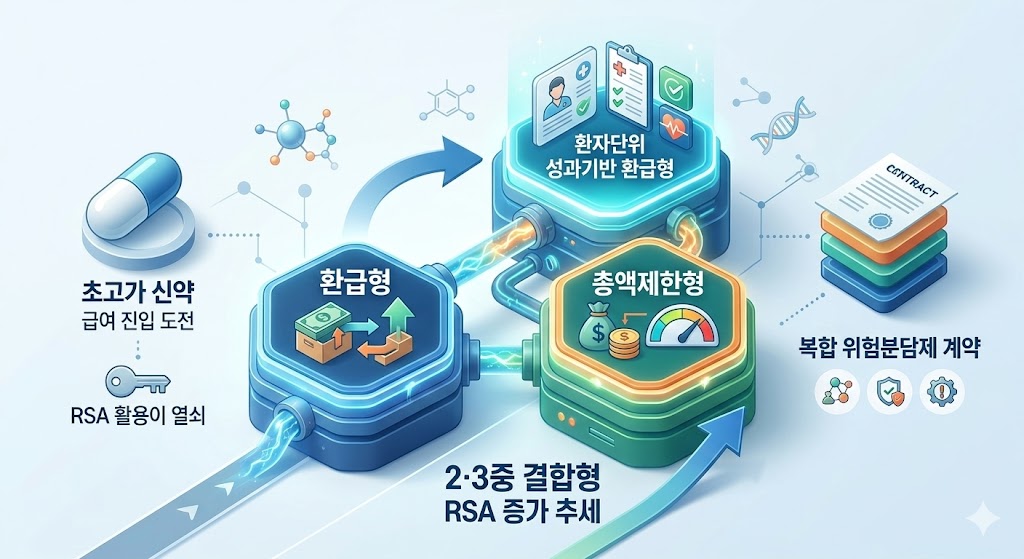
- Entering into RSA contracts is the key to high-cost drugs
- by Jung, Heung-Jun Mar 10, 2026 08:55am
- As the number of ultra-high-cost new drugs seeking insurance coverage grows, more medications are entering double or triple Risk-Sharing Agreement (RSA) contracts.There is a growing trend of triple RSA contracts that combine the standard RSA Refund model with Expenditure Caps and Patient-Level Outcome-Based Refunds.AI-generated imageAccording to industry sources on the 5th, ultra-high-cost drugs recently listed are entering the reimbursement bracket through contracts that combine existing Refund and Expenditure Cap models with Outcome-Based Refunds.The government is strengthening these safety measures to manage financial risks while ensuring access to new drugs; in effect, these hybrid models have become an essential option for crossing the reimbursement threshold.A majority of the new drugs listed or granted expanded coverage this month involved hybrid contracts. Antengene's Xpovio (selinexor) and AstraZeneca Korea's Imjudo and Imfinzi were all subject to both Refund and Expenditure Cap models.For Xpovio, the additional financial requirement due to expanded coverage was estimated at KRW 11.6 billion. However, the government determined that the actual financial impact would be lower when applying two types of RSA.GC Biopharma's Livmarli (maralixibat), a treatment for pruritus in Alagille syndrome, was subject to a triple contract: a Refund, an Expenditure Cap, and a Patient-Level Outcome-Based Refund.The Patient-Level Outcome-Based Refund model tracks treatment results; if pre-agreed targets are not met, the pharmaceutical company must refund a certain amount. It is reported that Livmarli passed the Drug Benefit Evaluation Committee and the Health Insurance Policy Deliberation Committee because GC Biopharma submitted a risk-sharing plan that included this outcome-based refund.Ipsen Korea's Bylvay (odevixibat), a treatment for pruritus in patients with cholestatic liver disease listed last October, is another case of a triple contract.During its cost-effectiveness evaluation, Bylvay was found to have higher annual costs than alternative treatments. However, it was able to clear the listing threshold through the risk-sharing types proposed by the pharmaceutical company. At the time, the estimated annual claim amount was KRW 118.4 billion, but the financial burden was deemed lower through the RSA.As the listing and expansion of reimbursement for immuno-oncology drugs and high-cost new drugs continue to increase, pharmaceutical companies are actively using combinations of Refund, Expenditure Cap, and Outcome-Based Refund models to obtain National Health Insurance coverage.
- Policy
- Adstiladrin receives expedited review in Korea
- by Lee, Tak-Sun Mar 09, 2026 08:53am
- The gene therapy for bladder cancer ‘Adstiladrin,’ developed by Swiss company Ferring Pharmaceuticals, has been selected for Korea’s GIFT program, enabling expedited regulatory review by the Ministry of Food and Drug Safety (MFDS).The drug is the world’s first gene therapy for bladder cancer and received U.S. FDA approval in 2022.The MFDS announced on the 6th that Adstiladrin (nadofaragene firadenovec) has been designated as a drug eligible for fast-track review. The designation date was February 4th.The therapy has been submitted for approval in Korea for the treatment of high-risk non–muscle-invasive bladder cancer (NMIBC) that is Bacillus Calmette-Guerin (BCG)-unresponsive with carcinoma in situ (CIS), with or without papillary tumors.The MFDS designated the drug for fast-track review, citing improved efficacy compared with existing treatments. It had previously also been designated as an orphan drug.Adstiladrin also underwent fast-track review during the U.S. FDA approval process, leading to its accelerated approval. The therapy exerts its antitumor effect through the expression of the interferon alpha-2b (IFNα2b) gene delivered via a non-replicating adenovirus vector, administered intravesically.The GIFT program is a Global Innovative products Fast-Track Review Program designed to support the development of innovative medical products in Korea and has been in operation since September 2022.Eligible products include innovative medicines targeting life-threatening diseases, rare diseases with no existing treatment alternatives, and new drugs developed by certified Korea Innovative Pharmaceutical Companies.The MFDS conducts a comprehensive evaluation of the candidates’ innovative therapeutic benefits, contributions to addressing public health crises, and the developer’s efforts, among other factors.Adstiladrin has been designated as GIFT No. 65. Among the 50 products approved under the GIFT program so far, 42 are orphan drugs.
- Policy
- Health and Welfare Committee to conduct inquiries on pending issues next week
- by Lee, Jeong-Hwan Mar 09, 2026 08:53am
- The ruling and opposition party leaders of the National Assembly's Health and Welfare Committee have agreed to hold a plenary session on the 10th of next week to receive this year's (2026) business reports and conduct inquiries on pending issues.The committee plans to hold the First and Second Subcommittees for Legislations on the 11th and 12th, respectively, to process pending legislation. Following this, the bills are expected to be approved during a plenary session on the 13th and forwarded to the Legislation and Judiciary Committee.Key issues to be highlighted during next week's plenary session include the findings regarding contaminated COVID-19 vaccines and the proposed restructuring of the drug pricing system, which includes price cuts for generics and preferential pricing for innovative pharmaceuticals.Other pressing topics include policies to strengthen regional, essential, and public healthcare, such as increasing medical school quotas, establishing new regional·public medical schools, and implementing a regional physician system.The opposition, led by the People Power Party, is expected to take issue with Minister of Health and Welfare Jung Eun Kyeong's failure to publicly announce cases of COVID-19 vaccines containing mold or foreign substances during her previous leadership as Commissioner of the Korea Disease Control and Prevention Agency (KDCA). They are likely to demand a parliamentary investigation.Rep. Kim Mi-ae, the executive secretary of the committee, along with Rep. Na Kyung-won and Rep. Shin Dong-wook, are strongly calling for Minister Jung’s immediate resignation. They are also demanding that the government drop its appeals against vaccine-related lawsuits and initiate a parliamentary investigation into the contaminated vaccines.Lawmakers from the Democratic Party also plan to question the announcement regarding the contaminated vaccine issue during next week's session. As the majority party, they are expected to counter the opposition's criticisms by emphasizing the necessity of vaccinations during the peak of the COVID-19 pandemic.Regarding the drug pricing restructuring, which the Ministry of Health and Welfare (MOHW) plans to implement in July following approval by the Health Insurance Policy Deliberation Committee this month, inquiries are expected to focus on whether the system adequately encourages and fosters innovation within the domestic pharmaceutical and biotech industry.In fact, Rep. Kim Yoon of the Democratic Party previously criticized the MOHW's plan as "too mechanical" and insufficient for rewarding the innovativeness of domestic pharmaceutical companies, and subsequently demanded the submission of a revised proposal.Despite persistent protests from the Korean pharmaceutical industry against the restructuring plan, the MOHW is maintaining its position in its policy to secure Health Insurance Policy Deliberation Committee approval in March.The MOHW plans to hold a one-point subcommittee meeting on the drug pricing restructure next week, followed by a final vote at the plenary session at the end of the month. Detailed calculation rates for generic drugs and preferential pricing measures for innovative pharmaceutical companies are expected to be finalized within this month.The MOHW has proposed reducing the generic pricing rate from the current 53.55% to the 40%, while the pharmaceutical industry views 48% as their bottom line or maximum acceptable cut.Furthermore, the industry argues that the criteria and mechanisms for preferential pricing must be significantly revised to reflect contributions to the development of the domestic industry and to the manufacturing, production, and distribution of high-quality medicines, in order to achieve the Ministry's policy goals.An official from a Health and Welfare Committee member’s office commented, "Since the New Year's business reports have been delayed and the bill subcommittees have not met properly for months, holding the standing committee in March is essential," adding , "The People Power Party has agreed to participate, and I understand that pending issues like the COVID-19 vaccine contamination played a role in that decision."
- Policy
- GSK’s Nucala-Omjjara enters pricing negotiations for reimb in Korea
- by Jung, Heung-Jun Mar 09, 2026 08:53am
- [GSK Korea’s new myelofibrosis drug Omjjara (momelotinib) and the eosinophilic disease treatment Nucala Autoinjector (mepolizumab) have entered drug price negotiations with the National Health Insurance Service (NHIS).In addition, Janssen Korea’s new multiple myeloma treatment, Darzalex SC injection (daratumumab), is also proceeding with price negotiations, the final stage of reimbursement listing.According to industry sources on the 6th, new drugs that passed the Drug Reimbursement Evaluation Committee (DREC) in January have entered the negotiation process with the NHIS.Both Omjjara and Nucala passed the DREC with the condition that reimbursement would be accepted only if the price is set below the committee’s evaluation amount. AI-generated imageOmjjara Tab (100, 150, and 200 mg) was deemed adequate for reimbursement as a treatment for intermediate- or high-risk myelofibrosis in adults with anemia. Nucala Inj was deemed adequate for reimbursement as an add-on maintenance therapy for the treatment of severe eosinophilic asthma in adults and adolescents that is inadequately controlled with existing treatments.With GSK receiving reimbursement approval for two new drugs simultaneously, expectations are rising on the possibility of their concurrent listing.Omjjara passed the Cancer Disease Deliberation Committee review last March but faced delays in reaching the DREC due to issues such as the selection of comparator drugs, requiring a resubmission. It cleared the DREC hurdle approximately 10 months after resubmission.Meanwhile, the Nucala Autoinjector, a new formulation of the drug, is a self-injectable device that allows patients to administer the treatment at home. If reimbursement coverage is expanded to include the self-injection formulation, Nucala’s prescription market presence is expected to grow.At the previous DREC meeting, both Omjjara and Nucala were approved with the condition that reimbursement is appropriate only if the company accepts a price below the evaluated amount. Since the pharmaceutical company has accepted this condition, negotiations with the NHIS are expected to focus on details such as the projected claims amount.Meanwhile, Janssen Korea’s Darzalex SC Inj was recognized as appropriate for reimbursement for ‘combination therapy with bortezomib, cyclophosphamide, and dexamethasone in newly diagnosed light-chain amyloidosis patients.’Unlike Omjjara and Nucala, Darzalex did not receive the “below evaluation price” condition from DREC, allowing it to proceed directly to price negotiations.Following its approval by the CDDC in the latter half of last year, the subcutaneous injection formulation of Darzalex is steadily gaining access to tertiary hospitals in Korea.
- Policy
- Pharmaceutical Act prohibiting non-face-to-face wholesaling
- by Lee, Jeong-Hwan Mar 09, 2026 08:53am
- The amendment to the Pharmaceutical Affairs Act, prohibiting non-face-to-face care platforms from concurrently operating as pharmaceutical wholesalers, is expected to be passed in its original form without revisions.Analysis suggests that related personnel, including Representative Choo Mi-ae, Chairperson of the Legislation and Judiciary Committee, and Representative Han Jeoung-ae, Chair of the Democratic Party's Policy Committee, have maintained a firm commitment to the original bill, which had been stalled at the plenary session for several months after being initially scheduled for processing late last year.The legislation had previously passed the Health and Welfare Committee and the Legislation and Judiciary Committee with agreement between both parties. However, it was repeatedly excluded from the plenary agenda due to opposition from certain lawmakers and the Ministry of Small Businesses and Startups.According to officials from the National Assembly and the pharmaceutical industry, the Pharmaceutical Act prohibiting non-face-to-face care platforms from concurrently operating as pharmaceutical wholesalers is likely to pass the National Assembly plenary session this month.The amendment is aimed at preventing conflicts of interest that arise when a non-face-to-face platform also acting as a wholesaler. If a platform acts as an intermediary for non-face-to-face treatment while also functioning as a wholesaler, a conflict of interest can arise. This is because the platform would be directly involved in prescribing, preparing, and distributing the medications it manages. Additionally, this setup raises concerns that the system could be misused as a method for providing rebates on illegal medications.Some Representatives characterized the bill as the "Doctor Now Prevention Act" and argued it would stifle innovation in the telemedicine sector. Accordingly, this bill was delayed for the National Assembly consideration.In this process, a sharp difference in positions between the relevant government agencies, the Ministry of Health and Welfare (MOHW) and the Ministry of SMEs and Startups, emerged, leading to suggestions that mediation by the Prime Minister's Office and the Office of the President was necessary.However, Ministry of Health and Welfare Minister Jung Eun Kyeong maintained firmly on passing the original version to ensure institutional safety and block market distortions. Within the Democratic Party, voices grew to maintain the original bill, with leadership emphasizing that revised proposals should not compromise the legislative process.In fact, ruling party lawmakers, including Democratic Party Chairman Jung Cheong-rae and Rep. Choo Mi-ae, visited the General Assembly of the Korean Pharmaceutical Association and promised to pass pharmacy-related legislation, including a bill to prevent intermediary platforms from concurrently operating wholesale businesses. Consequently, it is likely that the bill will be considered and processed during this month's plenary session.An official from a Democratic Party lawmaker’s office commented, "From the start, the bill that passed the Legislation and Judiciary Committee was not a matter where amendments should have been discussed, or its tabling in the plenary session was repeatedly excluded without a specific reason." The official added, "While there were some differing opinions within the party, the floor leadership stayed committed to passing the original version."