- LOGIN
- MemberShip
- 2026-04-25 01:06:18
- Policy
- Afilivu’s price cut and lists PFS formulation to chase Eylea
- by Jung, Heung-Jun Mar 24, 2026 08:16am
- Samsung Bioepis is stepping up its pursuit of Eylea by lowering the price of its macular degeneration treatment Afilivu and expanding into the prefilled syringe (PFS) segment.Next month, Afilivu Prefilled Syringe 6.6 mg/0.165 mL (aflibercept) will be listed for reimbursement, and the price of the existing Afilivu injection will be voluntarily cut to KRW 198,000, matching the market’s lowest price.According to industry sources on the 23rd, Samsung Bioepis is simultaneously pursuing both a price reduction and the addition of a new formulation to strengthen Afilivu’s competitiveness.Samsung Biologics expands Aflilivu's competitivity by introducing a PFS version with reimbursement. AI-generated imageThe ceiling price of Afilivu 40 mg, previously KRW 298,000, will be voluntarily reduced by KRW 100,000 to KRW 198,000.This matches the lowest price set by Sam Chun Dang Pharm’s Eylea biosimilar ‘Vgenfli Inj.’The Afilivu PFS formulation entering reimbursement this time has also been listed at the same price of KRW 198,000. Not only Eylea, but also Celltrion’s Eydenzelt and Sam Chun Dang Pharm’s Vgenfli already have PFS formulations in their reimbursement lineups.Celltrion also expanded its prescription lineup by listing the PFS formulation last November. Although the Afilivu PFS formulation is a later entrant compared to these, it appears the company will launch a full-scale market offensive by lowering its price.Sales of Afilivu in Korea were suspended early last year due to a preliminary injunction filed by the original developer, Regeneron, seeking to halt sales. However, after the injunction was overturned in December of the same year, domestic distribution and sales resumed. Samil Pharmaceutical holds the exclusive domestic sales rights.It is said to have shown a rapid recovery in sales since resuming distribution. Samil is expected to continue expanding prescriptions through sales activities that now include the PFS formulation.The domestic Eylea market is estimated at about KRW 100 billion. Samsung Bioepis, Celltrion, and Sam Chun Dang Pharm are all closely chasing Eylea and competing to expand market share.In particular, fierce competition is expected between Samsung Bioepis and Sam Chun Dang Pharm, as they are clashing with a lowest-price strategy.
- Policy
- Menarini withdraws approval for angina drug Ranexa
- by Lee, Tak-Sun Mar 24, 2026 08:16am
- ‘Ranexa (ranolazine), a new angina treatment with a novel mechanism of action that had garnered high expectations as the first of its kind in over 20 years, has given up on entering the Korean market five years after its local approval.Used as a second-line treatment for angina, the drug had remained approved but never been listed for reimbursement. As a result, it appears that the company decided to withdraw the product once the re-examination period expired.According to the Ministry of Food and Drug Safety, Menarini Korea voluntarily withdrew marketing authorization for three strengths of Ranexa extended-release tablets—375 mg, 500 mg, and 750 mg, as of the 23rd. Having obtained domestic approval in March 2020, the drug never made it to market and has now disappeared from the regulatory records after five years.Unlike existing beta-blockers or calcium channel blockers (CCBs), Ranexa garnered attention for its unique mechanism of action, which selectively inhibits sodium channels within cardiac muscle cells. Because it does not significantly affect blood pressure or heart rate, it has been considered an alternative for patients whose symptoms are difficult to control with existing medications.However, it never made it onto the national health insurance reimbursement list after approval. To make matters worse, the re-examination period granted at the time of approval reportedly expired on the 15th.Although three months remained before the application period for re-examination closed, the company appears to have chosen to withdraw the approval rather than pursue re-examination. Since no actual sales took place after approval, it is assumed that conducting a re-examination would also have been difficult.The drug was approved by the US FDA in January 2006. At the time, it was the first new angina treatment to gain approval in the United States in more than 20 years, drawing considerable attention.While it was approved in the US. as a first-line treatment for chronic angina, it was approved as a second-line treatment when it received approval in Europe. On July 9, 2008, the European Medicines Agency (EMA) approved it as an add-on therapy for first-line stable angina treatment.When it received domestic approval on March 16, 2020, the indications were also determined based on the European approval. Menarini Korea conducted bridging studies and other procedures to obtain domestic approval for this drug, ultimately receiving approval one year and eight months after filing.Low market potential is cited as the reason for withdrawing from the domestic market. With existing angina treatments already priced low, it is believed that Ranexa, as a second-line therapy, also faced an environment in which it would be difficult to secure a sufficiently high reimbursement price.Currently, there are no generics containing the same active ingredient available. Consequently, medications containing ranolazine are no longer available in Korea.An industry official said, “When a new drug gives up entering the Korean market because of marketability issues, it ultimately only lowers treatment accessibility for patients. When a new drug is withdrawn, it is necessary to closely examine whether there are any generics with the same ingredient or alternative medicines available.”
- Policy
- CKD and Samjin win approval for 3mg donepezil 3 mg
- by Lee, Tak-Sun Mar 23, 2026 08:42am
- Leading generic manufacturers of the long-standing best-selling dementia treatment “Donepezil” are strengthening their product lineups by introducing a new 3mg low-dose formulation.Attention is focused on whether this will change a market structure that has so far centered on 5 mg and 10 mg products.According to the Ministry of Food and Drug Safety, following the approval of Chong Kun Dang’s “Neuropezil Tab 3mg” on the 18th, Samjin Pharmaceutical’s “Neutoin Tab 3mg” received marketing authorization two days later on the 20th.Neuropezil and Neutoin are leading donepezil generics that have recorded outpatient prescription sales of KRW 12.9 billion and KRW 5.2 billion, respectively, last year, based on Ubest data.The company that first opened the 3 mg donepezil market was Hyundai Pharm. On June 28, 2023, Hyundai became the first in Korea to win approval for ‘Hipezil 3 mg’, taking the lead in the low-dose market. Later, on February 27 last year, Myungin Pharmaceutical obtained the first generic approval with ‘Silvercept 3 mg.’ Silvercept also recorded KRW 5.3 billion in outpatient prescriptions last year, ranking relatively high among generics.At present, even Aricept, the original donepezil brand, does not have a 3 mg lineup in Korea. However, the fact that 3 mg products are already marketed and widely used in clinical practice in neighboring Japan became a key driver for Korean companies’ development efforts.The reason the pharmaceutical industry is focusing on the 3mg strength is safety. While donepezil is highly effective in alleviating dementia symptoms, a considerable number of patients report gastrointestinal side effects such as nausea, vomiting, and diarrhea during the initial stages of treatment.Korean companies, including Hyundai Pharm, developed 3 mg products so that the initial dose could be set at 3 mg for patients at high risk of gastrointestinal side effects or for elderly patients. During the approval process, two studies published in SCI-level academic journals were submitted as clinical evidence, and Japanese marketing authorization information was also used as reference material.An industry official stated, “Donepezil 3mg will serve as a useful alternative for high-risk patients who had to discontinue drug therapy due to side effects. With major pharmaceutical companies such as Chong Kun Dang and Samjin Pharmaceutical joining the field, the proportion of low-dose prescriptions in the dementia treatment market is expected to gradually increase in the future.”
- Policy
- Tightened rebate rules pressure innovative pharma certifications
- by Lee, Jeong-Hwan Mar 23, 2026 08:42am
- As the Ministry of Health and Welfare designs a reform of the innovative pharmaceutical company certification criteria, the pharmaceutical industry is raising concerns that the revised framework must include a system capable of reflecting companies’ self-regulatory efforts in compliance management, such as the prohibition of illegal rebates.The argument is that the reform plan must explicitly include provisions allowing the certification review process to consider a corporation’s efforts to prohibit rebates when illegal rebates are provided or detected due to individual misconduct by sales representatives, despite thorough management and supervision by the company itself.The logic behind this is that incorporating such provisions into the reform plan would prevent cases where certification is unfairly revoked due to rebates in which the corporation was not involved, while also creating incentives for pharmaceutical companies to operate internal compliance systems and lead self-policing efforts against rebates.According to the pharmaceutical industry and the ministry on the 20th, an amendment to the administrative notice improving the certification review criteria for Innovative Pharmaceutical Companies will soon be released for public comment.The MOHW plans to incorporate a revised statute of limitations for rebates, which would exclude illegal rebates occurring “more than five years prior” to the date of certification or recertification following the implementation of the reform plan.This is intended to encourage pharmaceutical companies’ commitment to developing innovative new drugs by not linking the revocation of Innovative Pharmaceutical Company certification to illegal rebates that occurred too long ago.However, the pharmaceutical industry argues that, in addition to this five-year rebate exclusion rule, the reform should also contain a mechanism to address the unintended consequence where pharmaceutical companies that have made sufficient efforts to prevent illegal rebates still face unconditional certification revocation, following the exemption for rebates older than five years.In other words, even where a company has established systems for monitoring and managing rebate prohibitions and has worked diligently to maintain compliance, it should be guaranteed the right to present a defense during the certification revocation review process if a rebate occurred due to the misconduct of an individual sales representative.On this, the industry said, “We are not asking for all rebates stemming from individual sales representatives’ misconduct to be automatically excluded from grounds for certification revocation. Rather, we are asking that an administrative track be established to allow for due consideration of the corporation’s efforts toward compliance-oriented management.”The reason for this is that if this provision is not reflected in the reform plan, pharmaceutical companies’ efforts toward compliance management to prohibit rebates will not be reflected at all during certification revocation reviews. This would increase the need to contest the unfairness of certification revocation in court, potentially leading to a significant rise in unnecessary administrative revocation lawsuits between pharmaceutical companies and the MOHW.In particular, pharmaceutical companies point out that the current regulations under review by the MOHW, which hold corporations jointly liable for the personal illegal acts of their internal sales representatives, while specifying that administrative sanctions for illegal rebates by Contract Sales Organizations (CSOs) should be imposed solely on the CSOs without linking them to the corporations, raise concerns that this could encourage pharmaceutical companies to abandon their own sales organizations and shift to CSO-based sales operations.In addition, as the ministry’s current drug pricing reform proposal moves in the direction of strengthening preferential measures based on whether a company is certified as an innovative company, there is concern that the management damage to companies whose certification is unfairly revoked will become far greater than before if no provision is included to reflect compliance management efforts.An industry official said, “If companies that have internal rebate-prevention systems and have made genuine efforts to control illegal conduct are judged under the same certification revocation standard as companies that do not implement compliance management, then the incentive for firms to eliminate illegal practices on their own disappears. There is also concern about the adverse effect that, as a disparity in administrative penalties arises between a pharmaceutical company’s internal sales staff and external CSO sales staff, if pharmaceutical companies decide to reduce the risk of administrative penalties by relying on CSO sales, the risk of circumventing regulations through backdoor rebate practices will increase.”Meanwhile, it appears unlikely that this reform proposal will include a provision to convert the revocation standard for innovative company certification in rebate-related cases into a demerit-point system.
- Policy
- Bispecific Ab 'Ziihera inj' for biliary tract cancer wins nod
- by Lee, Tak-Sun Mar 20, 2026 08:55am
- 'Ziihera inj'BeOne Medicines Korea's bispecific antibody for biliary tract cancer, 'Ziihera inj,' has obtained final approval. Analysis suggests that the expedited approval was made possible through the GIFT (Global Innovative product on Fast Track) review process. It is expected to provide new therapeutic opportunities for patients with biliary tract cancer.The Ministry of Food and Drug Safety (MFDS, Minister Oh Yu-Kyoung) announced that it has approved the imported orphan drug 'Ziihera inj 300mg (zanidatamab)' on the 19th.Ziihera is an orphan drug indicated for the treatment of adult patients with unresectable, locally advanced, or metastatic HER2-positive (IHC 3+) biliary tract cancer who have previously received at least one systemic therapy.HER2-positive (IHC 3+) refers to a state where HER2, a protein involved in regulating cell growth, is highly overexpressed as determined by an Immunohistochemistry (IHC) test.This drug is a bispecific antibody that simultaneously binds to two different extracellular domains (ECD4 and ECD2) of the HER2 protein. As an antineoplastic agent, its mechanism involves reducing HER2 expression to inhibit the growth of tumor cells. It is the first drug approved in South Korea for this specific indication. Consequently, expectations are high that it will offer a new treatment window for patients suffering from biliary tract cancer.Ziihera inj received expedited review after being designated as the 40th product under the 'Global Innovative product on Fast Track (GIFT)' system. GIFT is a support program designed to accelerate the commercialization of globally innovative medical products by assisting in the early stages of development (clinical trials).An official from the MFDS stated, "We will continue to ensure that new treatments for patients with rare and intractable diseases are supplied promptly, using our expertise in regulatory science, thereby expanding their opportunities for treatment."
- Policy
- Xtandi to list tablet form to defend market ahead of patent expiry
- by Jung, Heung-Jun Mar 20, 2026 08:55am
- Astellas Korea’s prostate cancer drug Xtandi Tab (enzalutamide) is expected to be listed for reimbursement next month. With 3 months remaining before the drug’s patent expiry, the company appears poised to defend its market share by adding the tablet formulation to its reimbursement lineup.This move appears to be an effort to secure a foothold in the tablet prescription market, where no generics have yet been approved, in preparation for generic competition following patent expiry.According to industry sources on the 19th, Astellas will add tablets to Korea’s reimbursement list next month, following the listing of its soft capsule formulation.Unlike the soft capsules approved in 2013, the tablet formulation was approved in December 2024. This means the tablets are entering the reimbursement list approximately 1 year and 3 months after approval.In the case of Xtandi soft capsules, multiple generics have already received approval and are awaiting the expiration of the substance patent on June 27.Alvogen Korea’s Anamide Soft Capsules, Daewon Pharmaceutical’s Enzadex Soft Capsules, Hanall Biopharma’s Enzaloo Soft Capsules, and Menarini Korea’s EnzalX Soft Capsules are expected to be launched.Generic competition is also expected to intensify for the tablet formulation. Companies including Hanmi, Chong Kun Dang, Alvogen Korea, JW Pharmaceutical, and GL Pharma have won passive scope confirmation trials on Xtandi’s composition patents.However, since no generics have yet received marketing approval, entry into the tablet market is expected to lag behind capsules.The newly listed Xtandi Tab will be available in 40 mg and 80 mg strengths, expanding the reimbursement lineup at the same price level as the capsule formulation.According to the market research institution UBIST, Xtandi recorded KRW 38 billion in sales last year, up 26% from KRW 30.2 billion the previous year.However, it appears difficult to sustain this sales growth trend starting in the second half of this year due to the market penetration of generics.
- Policy
- Twice-yearly injectable HIV drug Sunlenca nears approval in Korea
- by Lee, Tak-Sun Mar 20, 2026 08:55am
- AI-generated imageGilead Sciences’ multidrug-resistant HIV treatment Sunlenca (lenacapavir sodium) is reportedly nearing marketing approval in Korea.The drug was designated in March last year for the MFDS GIFT (Global Innovative product on Fast Track) program, and it has recently been reported that the safety and efficacy review has been completed.According to industry sources on the 19th, the MFDS has completed its safety and efficacy assessment of Sunlenca. Once a product passes this stage, only the final approval process typically remains. In addition, products that have undergone safety and efficacy reviews can apply for reimbursement to the Health Insurance Review and Assessment Service (HIRA) under the approval–reimbursement assessment linkage system.Observers predict that if Sunlenca has received a positive determination in the safety and efficacy review, it is highly likely to receive marketing authorization approval in the near future.Sunlenca is a first-in-class capsid inhibitor that targets multiple stages of the HIV replication cycle. It is primarily aimed at patients with multidrug-resistant HIV who have developed resistance to existing treatments and have no further treatment options.It inhibits HIV-1 at multiple points throughout the viral lifecycle, including by interfering with the capsid-mediated nuclear entry of the pre-integration complex and impairing virion production and proper capsid core formation.A key feature of the drug is its twice-yearly injectable dosing, offering improved convenience.This is because it is a “long-acting” formulation that requires only a single injection every 6 months (26 weeks) during the maintenance phase, after an initial phase combined with oral therapy. As a result, it is widely recognized for significantly reducing the psychological burden and inconvenience previously experienced by patients who had to take medication daily.An industry official stated, “Sunlenca is of great clinical significance in that it provides strong viral suppression through a completely new mechanism for these patients. In particular, the fact that it is a long-acting injectable administered once every six months can maximize patient adherence.”Sunlenca has already been approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) and is currently in use. Recently, it has also demonstrated exceptional efficacy as a pre-exposure prophylaxis (PrEP), drawing attention as a potential game-changer, set to reshape the HIV treatment market.The FDA approved Sunlenca in December 2022, following fast-track, breakthrough therapy designation (BTD), and priority review (PR) designations. The EMA approved it earlier in August 2022, and Japan’s PMDA approved it through priority review in August 2023.In Korea, it was designated an orphan drug in December 2024, and on March 4 last year, it was designated as GIFT product No. 46, reflecting the lack of existing treatment options.The indication submitted for its use in Korea is as combination therapy with other antiretrovirals for the treatment of HIV-1 infection in hin adults with multidrug-resistant HIV-1 infection who have a history of treatment failure with current antiretroviral therapy due to resistance, intolerance, or safety issues.
- Policy
- "MOHW will design the drug pricing policy considering NHI cost"
- by Lee, Jeong-Hwan Mar 19, 2026 12:38pm
- Lee Hyung-hoon, Second Vice Minister of Health and Welfare"We recognize that the pharmaceutical industry has varying opinions regarding government policy. However, the government designs policy by looking at where the public's expectations lie and what their interests are. The necessity of fostering the pharmaceutical industry and the fact that domestic generic drug prices are high are areas where society relatively shares a consensus, and social agreement is required. We are not setting a single specific drug pricing reform plan and trying to force it through. We will repeatedly collect and discuss opinions from the pharmaceutical industry. Since policy can change at any time, there is room to modify the reform plan."Lee Hyung-hoon, Second Vice Minister of Health and Welfare (60, Yonsei University), has drawn significant attention by highlighting the difficulties of reconciling diverse interests in a single policy for drug pricing reform, which primarily focuses on lowering the prices of previously-listed generic drugs.Lee stated that, given South Korea's adoption and operation of a single-payer National Health Insurance system, writing a reform plan that simultaneously achieves the goals of fostering the pharmaceutical industry and ensuring efficient, rational national pharmaceutical expenditures requires considering the interests of both the industry and the public as beneficiaries of health insurance.Meeting with the Korea Special Press Association on the 18th, Vice Minister Lee promised, "Rather than taking the stance of a policy authority, the Ministry of Health and Welfare designs policy by examining public interests. However, we have not finished setting a single drug pricing reform plan. Policy is open and can continue to change. We will communicate with the pharmaceutical industry."This was the Vice Minister's response when asked how the Ministry intends to resolve the industry's backlash against the drug pricing system reform plan announced on November 28 last year.Lee stated that, while agreeing on the necessity of fostering the South Korean pharmaceutical and biotech industry on a global scale to strengthen national competitiveness, the final reform plan would be finalized after collecting opposing opinions from the industry on uniform price cuts.The Ministry intends to listen to the industry's voice and to prepare a revised proposal until the Health Insurance Policy Deliberation Committee deliberates on the reform plan on the 26th.Since releasing the initial draft of the reform plan on November 28 last year, the Ministry has presented a revised draft to the industry on March 11 at the subcommittee. However, the domestic pharmaceutical industry continues to criticize the process as 'closed administration,' citing the failure to specify clear generic price calculation rates, mentioning only a range of 'low-to-mid 40%,' the lack of specific timing and methods for price cuts per item, and the absence of detailed criteria and periods for preferential drug pricing.Lee repeatedly stated that he would gather industry opinions before the committee's final decision. He emphasized that, because pharmaceutical expenditures are funded through the health insurance budget, the government views the drug pricing system considering the efficiency of public insurance premiums."South Korea operates the drug pricing system based on the health insurance finances funded by the mandatory premiums paid by citizens," Lee stated. "Because medical fees and drug prices for both generics and new drugs are operated through health insurance finances, we value the National Health Insurance cost, which represents public expectations."Lee also noted that he recognizes the domestic pharmaceutical industry as a partner in South Korea's efforts to advance to one of the top five global pharmaceutical and biopharmaceutical powers. This is interpreted as a necessity for the administration to communicate and make adjustments to the aspects the industry hopes will be reflected in the reform plan.Lee stated, "Since the Ministry of Health and Welfare also aims to leap into a top-five pharmaceutical and bio-powerhouse as a national task, the pharmaceutical industry is an important partner," Lee explained. "We are discussing the reform plan with the industry. There is a backlash because the industry has more favorable, explicit expectations, but we will continue to communicate. There is room for modification within the system."Lee added, "There may be differing opinions because the industry has its own perspective and expected levels. While the Ministry has its position as a policy authority, we work by looking at where the public views, perspective, and interests lie."Lee stated, "We are open to differing opinions on Ministry policy or points raised by the media. We are not setting one thing and trying to force it through," adding, "Policy can continue to change."Lee concluded by saying, "The need to foster the pharmaceutical industry and the fact that generic prices are high are areas where there is a relatively social consensus. We have carefully considered the drug pricing policy. We are listening to the pharmaceutical industry's opinions," adding, "We fully recognize, acknowledge, and respect the necessity for innovative pharmaceutical companies and the domestic industry to possess national competitiveness and move toward becoming a bio-power. We support and share with the industry's aspiration to expand to a larger global stage."
- Policy
- MOHW drug pricing reform to likely bypasses NA briefing
- by Lee, Jeong-Hwan Mar 18, 2026 09:11am
- Joomin Park, chair of NA Health and Welfare CommitteeThere is a growing likelihood that the Ministry of Health and Welfare’s drug pricing system reform plan, the first in 14 years, will be finalized and implemented without a formal briefing to the National Assembly.Although Joomin Park, Chair of the National Assembly Health and Welfare Committee from the Democratic Party of Korea, instructed Health and Welfare Minister Jung Eun-kyung to provide a separate briefing on the reform plan, it has been confirmed that the ruling and opposition floor leaders on the committee are not actively coordinating a schedule for a plenary session.On the 17th, an official from an opposition lawmaker’s office on the National Assembly’s Health and Welfare Committee explained, “We have heard absolutely nothing regarding a plenary session for a separate briefing on the drug pricing reform plan.”If this explanation is correct, it means that despite Chair Park’s remarks on the need for a separate briefing on the drug pricing system, negotiations between Democratic Party floor leader Sujin Lee and People Power Party floor leader Miae Kim have come to a complete standstill. If an additional plenary session for the briefing is not held, the Ministry of Health and Welfare will proceed with the drug pricing reform plan, which has faced backlash from the pharmaceutical industry, without reporting to the National Assembly.The need for an additional National Assembly briefing on the drug pricing reform plan first arose during the plenary session held on the 10th for the annual policy briefing by relevant government ministries, when Rep. Sun-min Kim of the Rebuilding Korea Party raised the need through a procedural intervention.Rep. Kim criticized the ministry for attempting to push forward rapidly with the drug pricing reform—centered on across-the-board generic price cuts—despite opposition from the pharmaceutical industry, while including no update at all on the reform plan among the key issues in its official policy briefing.At the time, Committee Chair Park agreed with Kim and told Minister Eun-Kyoung Jeong, “Because this is a very important matter, it would be a good idea to provide an additional briefing at a plenary session once the procedures related to the reform plan are completed.”Nevertheless, the reason why the convening of the Welfare Committee’s plenary session remains a distant prospect appears to be rooted in the Ministry of Health and Welfare’s passive attitude and the suspension of consultations between the ruling and opposition parties.The Ministry of Health and Welfare has no incentive to take the lead in convening a plenary session at the administrative level, free from the burden of a one-point briefing, as the reform plan to lower the generic drug pricing rate from 53.55% to the 40% range has met with opposition from the pharmaceutical industry.As for the ruling party, given the ongoing fierce criticism from the opposition regarding the national vaccination campaign for COVID-19 vaccines found to contain foreign substances, which proceeded without proper procedure, the party has no reason to hold an additional plenary session that would expose it to further attacks from the opposition.In fact, People Power Party lawmakers on the Legislation and Judiciary Committee have decided to file a complaint accusing Minister Eun-kyoung Jeong, who served as Commissioner of the Korea Disease Control and Prevention Agency during the COVID-19 pandemic, of alleged dereliction of duty over the administration of 14.2 million doses of contaminated vaccines.Time constraints also make an additional plenary session difficult. The ministry plans to finalize the details and implementation schedule of the drug pricing reform at the HIPDC meeting on the 26th, and physically, it may be impossible to hold an additional National Assembly briefing before then.If the additional plenary session of the Health and Welfare Committee fails to materialize, the ministry is expected to have the reform plan approved at the HIPDC without first reporting it to the National Assembly.The Ministry of Health and Welfare plans to finalize the specific details and implementation date of the reform plan at the full HIPDC meeting on the 26th, following a subcommittee meeting on the 11th focused solely on the drug pricing reform plan and another subcommittee meeting on the 18th.An official from an opposition party lawmaker’s office on the Health and Welfare Committee said, “It is uncertain whether a plenary session for an additional briefing on the drug pricing reform plan will be held. Time is tight physically, and I understand that both the ruling party and the ministry are passive about holding one. The conflict between the ruling and opposition parties over the contaminated vaccine issue and calls for Minister Jeong’s accountability are also affecting the failure to agree on the briefing for the drug pricing reform plan.”The official continued, “In a situation where the opposition is demanding a hearing on contaminated vaccines and the indictment of Minister Jeong, an additional briefing on the drug pricing reform plan, which is already drawing strong resistance from the pharmaceutical industry, would naturally be burdensome for the ruling party. Although the pricing reform is an important policy that will shape the future of the pharmaceutical industry, and although Chair Park has requested a report on it, the government and ruling party seem to lack the will to proceed.”An official from the office of Democratic Party floor leader Sujin Lee stated, “Given the timing, it seems difficult to hold an additional plenary session on the drug pricing system, but we plan to hear the opinions of the opposition party’s floor leadership. If an additional briefing proves difficult, we will also consider a plan for ruling party lawmakers to receive individual briefings on the reform plan from the Ministry of Health and Welfare.”
- Policy
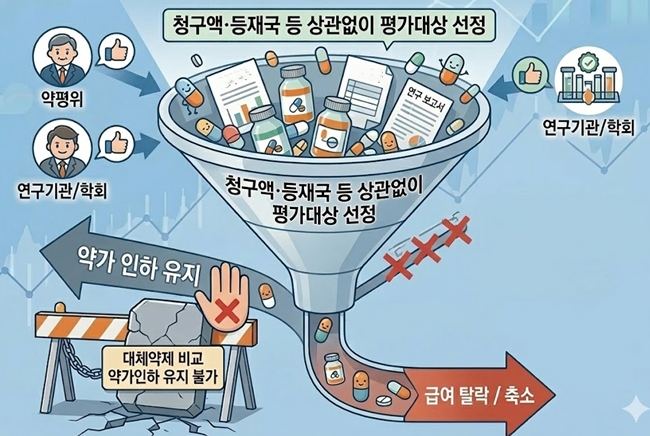
- Scope expanded, outcomes tightened for reimbursement reassessment in Korea
- by Jung, Heung-Jun Mar 18, 2026 09:11am
- The government is moving to specify implementation criteria as part of its efforts to reform the reimbursement adequacy reassessment system. It appears that ingredients for which the Drug Reimbursement Evaluation Committee (DREC), research institutes, academic societies, and others raise the need for reassessment will undergo reevaluation regardless of claim volume or listing-country criteria.In addition, the option of maintaining reimbursement through price cuts by comparing a product with its alternatives is expected to disappear going forward.The government is specifying the implementation criteria for reimbursement adequacy reassessment. AI-generated image.According to industry sources on the 17th, the government is specifying the implementation requirements for reimbursement reevaluation reassessments ahead of deliberation by the Health Insurance Policy Deliberation Committee (HIPDC) later this month.Until now, reassessments have been conducted on ingredients among older listed drugs that met the claim volume and countries of listing criteria.The reform plan announced last November signaled a shift away from criteria based on claim volume and listed countries, instead focusing on: ▲ ingredients for which national health authorities in A8 countries have initiated clinical or reimbursement adequacy reassessment; ▲ cases where data or clinical evidence contradicting previously reported efficacy has been published; and ▲ drugs for which the need for reassessment has been recommended by academic societies or experts.Recently, ingredients deemed necessary by DREC through monitoring, as well as those evaluated by research institutions or public agencies, have been subject to additional review.This means the committee will be able to proactively examine and designate a broader range of candidates for reassessment. As a result, concerns are emerging within the industry that the number of drugs subject to reassessment may increase.A pharmaceutical industry official said, “There had already been a tendency to broaden the criteria and expand the scope of targets. The intent is to cut drug spending by strengthening reassessment. Since the idea is that reassessment will proceed whenever there is a perceived need, I believe the number of targets will likely increase.”In particular, because the results of reassessment are being simplified into either delisting or positive listing, the weight of target selection becomes even greater. The option of comparing a product with alternative therapies and maintaining reimbursement through price cuts will disappear.Another industry official said, “It is difficult to predict how things will unfold next year. Depending on the new implementation criteria, the number of targets could either increase or decrease. However, considering the practical work involved in reassessment, the authorities cannot simply increase the number of ingredients indiscriminately, so the annual number of reassessment targets may not change significantly.”