- LOGIN
- MemberShip
- 2026-04-24 08:27:14
- Policy
- Government busy cutting drug prices without volume control
- by Lee, Jeong-Hwan Mar 30, 2026 09:11am
- Health insurance drug expenditure is calculated by multiplying the reimbursement price by the prescription and utilization volume. No matter how much the government repeatedly lowers drug prices, it is difficult to achieve the policy goal of reducing pharmaceutical expenditure or rationally managing the health insurance budget unless physicians’ prescribing behavior and real-world usage are also controlled.In other words, even if the government lowers the generic pricing rate from 53.55% to 45% (a reduction of about 8.55 percentage points), failure to control prescription and utilization volume could paradoxically lead to higher drug spending than before the price cuts.If not handled carefully, this could lead to a worst-case scenario where the price cut creates an environment in which generics of lower quality than in the past are produced, while at the same time, some companies intensify their efforts to offer illegal rebates to increase prescription volumes in an attempt to mitigate the shock of price cuts.This is why, on the 27th, academia and the pharmaceutical industry urged the Ministry of Health and Welfare not to focus solely on the reform plan for generic drug price cuts, but to establish mechanisms to reasonably control prescription volumes.Academia “Government must lead generic competition through policies such as mandatory lowest-price substitution”Academia argues that attempting to reduce health insurance expenditure through price cuts alone without addressing volume control is fundamentally misguided.In particular, the prevailing view among academics is that while the Ministry’s efforts are needed to create an environment where pharmaceutical companies can compete to dominate the market through “low-cost generic” strategies, its repeated exclusion of this aspect is ultimately reinforcing an industry structure that leaves companies with no choice but to generate sales through illegal rebate competition.Criticism has been raised that the Ministry has virtually no plans for policies designed to enable low-cost generics to gain a competitive edge in the market, such as promoting the substitution of the lowest-priced generics, establishing a competitive landscape for generics based on international nonproprietary name prescriptions, or shifting the environment for generic selection through a Korean-style reference pricing system.Health economists emphasize that once bioequivalence is established, generics should be allowed to compete solely on price.Professor Hye-young Kwon of Mokwon University stated, “The conflict between the industry and the government over the generic drug pricing rate is meaningless. If drug prices fall, companies are more likely to increase prescription volume through rebates, which may maintain or even increase overall drug spending. Ultimately, cheaper generics must be prescribed more and sold more in order to achieve both health insurance savings and pharmaceutical industry development.”Professor Kwon added, "The Ministry of Health and Welfare must devise administrative measures to create a competitive market where pharmaceutical companies that lower prices the most for each ingredient dominate the market. A pharmaceutical company that wins on price in the domestic market has a good chance of succeeding in the global market as well. The government must work with doctors and patients to devise policies such as mandating the substitution of the lowest-priced generic, using the cheapest drug as a reference price, and providing incentives when that drug is used, or imposing disincentives when more expensive generics or originals are used.”She emphasized, “If we establish a generic price competition policy that goes beyond the government and pharmaceutical companies to include doctors, pharmacists, and patients, pharmacists and patients will be able to intervene in the decision-making process to ensure cheaper drugs are used, even if a doctor unilaterally prescribes a specific drug This would allow rationalization of health insurance finances without excluding physicians’ clinical judgment. The government needs to make a decisive move and establish the necessary framework.”Professor Seung-jin Bae of Ewha Womans University College of Pharmacy also believes that the Ministry of Health and Welfare has not given sufficient consideration to usage policies beyond generic price adjustment policies. In particular, the professor stated that administrative measures by the Ministry are needed to address the current reality where doctors are bound to brand-name prescriptions despite the government recognizing generics as therapeutically equivalent.Specifically, Bae suggested activating lowest-price substitution or implementing INN prescribing to enable genuine price competition among generics.Bae said, “We need policies that allow cheaper generics to expand market share. The issue of brand-name prescribing must be addressed first. It makes no sense for the government to acknowledge bioequivalence and still tolerate brand-name prescribing practices.”Bae emphasized, “We should not view this (the development of health insurance cost-saving policies) as a conflict between professions. Instead, we must consider measures such as substituting the lowest-priced generic to allow the market for chemical generics to be organized through price competition. Korea’s health insurance resources are not unlimited. In the case of biosimilars, there may be differing opinions regarding INN prescribing or substitution. However, for chemical drugs, we must take a more proactive approach from the perspective of health insurance finances.”The domestic pharmaceutical industry also maintains that, alongside price cuts, addressing physician prescribing behavior and patient overuse is essential to achieving effective cost savings. If the government pays attention to volume control, it could move away from its current administrative pattern of repeatedly relying on price cuts.An official from a leading domestic pharmaceutical company pointed out, “The government establishes policies claiming it will reduce drug costs, maintain generic drug quality, ensure a stable supply of medicines, develop innovative new drugs, and foster robust pharmaceutical companies, all within limited health insurance funds, but the conclusion always boils down to drug price cuts. It is time to consider the root causes of why we face criticism that medical institutions prescribe more drugs than necessary and why pharmaceutical companies are forced to boost sales through rebate competition.”“If we merely suppress drug prices while neglecting the increase in usage, we cannot achieve fiscal savings. If we only cut drug prices, companies will find it difficult to avoid decisions such as increasing prescriptions for their own drugs through aggressive marketing using CSOs, or producing and supplying low-quality generics by cutting costs and reducing employment.”Calls grow for governance involving government, industry, and academia in drug pricing policyWhenever drug price reduction reform plans are discussed, some in the pharmaceutical industry offer a self-deprecating assessment that “pharmaceutical companies are always in the position of the weakest party.”This reflects the reality that pharmaceutical companies find it difficult to openly and freely express their opinions or arguments, as they must constantly gauge the reactions of both the Ministry of Health and Welfare, which seeks to lower drug prices, and doctors, who hold the power to prescribe medications.Accordingly, the pharmaceutical industry is requesting that, starting with this drug pricing system reform proposal, a governance structure be established where government agencies, the pharmaceutical industry, and academia can discuss and design drug pricing policies together.The intent is to ensure that the pharmaceutical industry is granted the authority to participate in the decision-making structure for establishing drug pricing policies, including drug price reductions and revisions to post-approval drug price management systems, both administratively and legally.In the recent reform process, since the Ministry’s proposal was announced on November 28 last year, the pharmaceutical industry has been limited to merely proposing amendments and improvements to the Ministry’s draft, rather than actively voicing its own opinions, from the time the Ministry’s revised proposal was submitted to the subcommittee of the Health Insurance Policy Deliberation Committee on March 11 of this year until its final approval at the plenary session on the 26th.In particular, since the Ministry’s proposal was not made public until just before it was reported to the HIPDC, submitted to the subcommittee, and finally approved at the plenary session, pharmaceutical companies’ drug pricing officials had no choice but to work tirelessly trying to interpret the government’s intentions.In contrast, advanced countries overseas guarantee the pharmaceutical industry’s right to submit opinions on the direction, implementation timeline, and detailed regulations of drug pricing system reform proposals through mandatory agreements or legal commitments at the executive and industry levels.France operates under a framework agreement between CEPS and the pharmaceutical industry association, while the UK mandates stakeholder participation in pricing and reimbursement reforms through NHS England and the Department of Health.Japan also determines pricing through the Central Social Insurance Medical Council under the Ministry of Health, Labour and Welfare.An official from a mid-sized pharmaceutical company stated, “This drug price reform should serve as an opportunity to prepare various measures for establishing public-private consultative governance and to submit industry-level opinions to the government and the National Assembly. Even if we cannot exert a substantial influence on the establishment of the system, wouldn’t this create a forum to publicly convey the pharmaceutical industry’s well-founded opinions to the government, academia, and the public?”Academics have also suggested that improving the operational standards of the Health Insurance Policy Deliberation Committee (HIPDC), which is already run by the government, could ensure transparency in the establishment of the drug pricing system and the national health insurance system.Professor Bae said, “Regarding the governance of the drug pricing system and national health insurance policies, there is a need to ensure transparency by disclosing meeting minutes and other details discussed within the HIPDC. National health insurance finances and policies are funded using premiums paid by the public. Since the system is funded by public contributions, citizens have the right to know what is discussed during the Health Insurance Policy Deliberation Committee’s proceedings.”He added, “Ultimately, for everyone to accept the health insurance policies decided by the government, we must improve the process so that it is clear who expressed what opinions at the committee, rather than suddenly bringing items to a vote as is currently done. Creating additional consultative bodies outside the Health Insurance Policy Deliberation Committee framework risks creating unnecessary layers of bureaucracy.”
- Policy
- Will only trivalent vaccines be available this season?
- by Lee, Tak-Sun Mar 27, 2026 08:33am
- AI 이미지 생성 활용With the WHO recommending a full shift back to trivalent influenza vaccines, supply suspensions of quadrivalent vaccines are continuing one after another.There is even a possibility that quadrivalent vaccines may disappear from the domestic market in the 2026–2027 season. In fact, only trivalent vaccines have been included in the National Immunization Program (NIP) since last year.According to industry sources on the 25th, Boryung BioPharma recently reported to the MFDS that it would discontinue the supply of Boryung Flu V Tetra Vaccine.Boryung Flu V Tetra Vaccine is a quadrivalent vaccine approved in 2016, designed to protect against four influenza virus strains.The company explained, “As the WHO’s recommended influenza strain composition for the 2026–27 season does not include quadrivalent influenza vaccines, we have decided to discontinue production and supply of this product.”It added, “We plan to produce and supply trivalent influenza vaccines instead, and there is no risk of supply shortages resulting from the discontinuation of this product.”Boryung Biopharma had already discontinued another quadrivalent vaccine, Boryung Flu VIII Tetra, last year in line with WHO recommendations.On the 12th, Seqirus Korea likewise reported to the MFDS that it would discontinue supply of its Fluad Quad Prefilled Syringe. This is also a quadrivalent influenza vaccine, and the decision was made in accordance with WHO guidance.The company stated, “We plan to supply the trivalent version of this vaccine, ‘Fluad Prefilled Syringe ((influenza surface antigen MF59C.1 adjuvanted vaccine),’ in the domestic market.”CSL Korea, on the 19th, obtained approval for a trivalent influenza vaccine, Flucelvax Prefilled Syringe. Flucelvax previously had an approved quadrivalent version, and this time an additional trivalent version has been approved. This is interpreted as the company obtaining approval for a new product to transition to a trivalent vaccine in accordance with WHO recommendations. Furthermore, a trivalent vaccine is now mandatory for pharmaceutical companies wishing to participate in the National Immunization Program (NIP).The WHO recommended a trivalent influenza vaccine in February of this year, following its recommendation last year, noting that not a single case of the B/Yamagata lineage virus has been detected worldwide since March 2020.In particular, the WHO determined that there is no longer a need to include a viral antigen that no longer exists in the vaccine. Consequently, it established the trivalent vaccine composition, which excludes the Yamagata antigen from the existing quadrivalent vaccine system, as the standard and recommended a complete return to trivalent vaccines.Following WHO guidance, the government already switched the NIP influenza vaccines from quadrivalent to trivalent last year. As a result, quadrivalent vaccines were sold only in the private market. However, some confusion arose as certain pharmaceutical companies continued promoting quadrivalent vaccines without clearly disclosing the WHO recommendation.This year, as pharmaceutical companies are gradually shifting back to the trivalent vaccine, there are predictions that the quadrivalent vaccine may completely disappear from the market.
- Policy
- Complete revision to the "Innovative Pharma Company" certification
- by Lee, Jeong-Hwan Mar 27, 2026 08:32am
- The Ministry of Health and Welfare (MOHW) will make administrative move to encourage drug investment by raising the R&D expenditure requirement for "Innovative Pharmaceutical Company" certification.The MOHW will improve administrative fairness by revising the disqualification criteria related to illegal rebates. Specifically, "rebate violations that concluded more than five years before the point of certification or renewal evaluation" will now be excluded from the review process. The new policy aims to address the irrationality of revoking certifications based on violations that occurred in the distant past. However, the proposed "points-based scoring system" for rebate-related cancellations will not be introduced.Additionally, the certification track for foreign pharmaceutical companies will be newly established and revised. To encourage foreign firms to host research and production facilities in Korea, attract overseas capital, and engage in joint research and open innovation, the score weightings for these categories will be increased.Transparency in the certification process will also be enhanced. The minimum passing score for certification (65 points) will be explicitly stated in the official notice, and companies that fail to gain certification will be formally notified of the specific reasons for their disqualification.The Ministry of Health and Welfare (MOHW) revises the "Innovative Pharmaceutical Company" certification. 1. R&D expenditure ratio will be raised 2. "Rebate violations that concluded more than five years before the point of certification or renewal evaluation" will be excluded from review 3. Foreign company track for Innovative Pharmaceutical Company certificationOn the 26th, the MOHW issued a legislative and administrative notice regarding amendments to the Enforcement Decree, Enforcement Rules, and related notices of the "Special Act on Promotion and Support of Pharmaceutical Industry" (the Pharmaceutical Industry Act). The public opinion engagement remain open until May 6.R&D Expenditure Ratio to Rise by 2%pFirst, the R&D expenditure ratios required for the certification and renewal of Innovative Pharmaceutical Companies will be increased. While the ratio relative to pharmaceutical sales will rise by 2 percentage points across the board, the application of this new standard will be 'deferred for three years from the date of enforcement' to allow companies sufficient time to prepare.Through this administrative move, the MOHW aims to encourage a continuous expansion of R&D investment among certified firms.Furthermore, the system will now categorize firms into "Innovative Pharmaceutical Companies" and "Foreign Innovative Pharmaceutical Companies" to provide a regulatory system that accounts for the unique characteristics of multinational corporations. The track for foreign firms will be implemented immediately upon announcement.For companies holding cGMP (current Good Manufacturing Practice) or EU GMP certification seeking renewal under the relaxed "R&D expenditure-to-sales" ratio, they must now submit evidentiary materials prepared within 3 years of the certification's expiration date. This administrative move resolves the current lack of a specific timeframe for GMP-related documentation.The amendments to Enforcement Rules will take effect on the date of announcement and are expected to apply to renewal applications starting in the second half of this year.Revising 'Rebate Checking Policies' and 'Detailed Evaluation Criteria'Regarding illegal rebates, the MOHW has decided to exclude violations that occurred more than 5 years before the certification or renewal review.In cases where administrative appeals or lawsuits are filed, certification can be revoked within one year of the date of dismissal of the appeal or a final ruling against the company.By maintaining the current revocation structure rather than switching to a points-based system, and excluding violations older than five years, the MOHW aims to eliminate the irrationality of penalizing firms for old actions. This addresses repeated parliamentary criticism that legal stability and predictability for pharmaceutical companies were undermined when certifications were revoked for ancient violations.The current standards state that administrative actions for rebates under the Pharmaceutical Affairs Act or the Fair Trade Act that occurred five years before the certification review are excluded. However, if a lawsuit is filed challenging the administrative action, the date the judgment becomes final is treated as the date of the administrative action.Consequently, cases have occurred in which "Innovative Pharmaceutical Company" certifications were revoked due to rebate violations that occurred a long time ago. Criticisms rose from the National Assembly that the predictability and legal stability for pharmaceutical companies are being undermined.Furthermore, the detailed evaluation criteria for certification will also be improved and will be noted in the appendix. The total score will be adjusted from 120 to 100 points, and the number of evaluation items will be reduced from 25 to 17.Objectivity in certification standards will be enhanced by converting evaluation items, such as R&D investment, the number of clinical trials, and export volume, into quantitative indicators (4 out of 17 items).Notably, a new category will be established to recognize excellence in social responsibility activities, such as the production and supply of medicines that help stabilize the supply chain.For the Domestic Innovative Pharmaceutical Company certification standards, the score weightings for items such as partnership·collaboration activities, non-clinical·clinical trials, candidate development, and transparency in corporate management will be increased. Conversely, items such as research personnel, research/production facilities, and R&D strategies will be adjusted downward.Establishment of Certification Standards for Foreign Innovative Pharmaceutical CompaniesFurthermore, based on the types of Innovative Pharmaceutical Companies classified under the Enforcement Decree of the Pharmaceutical Industry Act, detailed certification review criteria for Domestic Innovative Pharmaceutical Companies and Foreign Innovative Pharmaceutical Companies will be separately regulated.Foreign pharmaceutical companies will be permitted to choose and apply under either the Domestic Innovative Pharmaceutical Company certification standards or the Foreign Innovative Pharmaceutical Company certification standards.The certification standards for Foreign Innovative Pharmaceutical Companies will have increased weightings for specific items to encourage foreign pharmaceutical firms to establish domestic research and production facilities, as well as to attract foreign capital, facilitate joint research, and promote open innovation.Given the characteristics of foreign pharmaceutical companies, in which the global headquarters typically holds the technology and patents, the score weightings for items related to the development of non-clinical and clinical trial candidates and to pharmaceutical patent technology transfer performance will be reduced.Finally, the MOHW will state the minimum passing score of 65 points and mandate that the reasons for failure be specified in notifications to companies. These changes will be reflected starting with new and renewal applications in the second half of this year.Meanwhile, the MOHW plans to systematically analyze the types and capabilities of Korean pharmaceutical and biotech companies and establish a "Strategy for Fostering National Pharmaceutical & Biotech-Industry" within this year.
- Policy
- Industry calls for more proactive drug pricing incentives
- by Lee, Jeong-Hwan Mar 27, 2026 08:32am
- When asked what should be the top priority administrative action after the government fully implements the drug pricing reform plan in the second half of this year, the pharmaceutical industry immediately responded, “Establishing proper drug pricing incentive policies.”Given that the reform plan places significant emphasis on lowering generic drug prices, the industry is demanding proactive measures to design policies that either clearly favor generics, which have contributed to savings in the National Health Insurance budget, or ensure that no further price cuts are imposed.At the same time, the industry proposed conducting a proper post-evaluation of whether past policies, such as blanket price cuts for generics and differential pricing based on criteria, have actually achieved their intended goals of reducing NHI expenditure and addressing the excessive proliferation of generic products.Following the across-the-board price cuts in 2012, virtually all pharmaceutical companies decided to produce multiple generic products, triggering the first generic drug boom. The proposal calls for a thorough analysis of why a second boom, marked by a further increase in the number of generic drug items, occurred following the 2020 announcement of a tiered pricing system based on requirements for in-house bioequivalence testing and Drug Master File (DMF) registration, in order to set the direction for a new drug pricing system.On the 26th, the pharmaceutical industry criticized that government efforts to establish drug pricing incentives, which are closely tied to domestic companies’ investment in innovative drug R&D, remain significantly insufficient.It also criticized the government for repeatedly introducing price cuts without conducting follow-up administrative measures to evaluate policies in collaboration with the industry, despite the fact that a boom in the number of generics has occurred every time drug prices were revised.Domestic new drugs and generics lack sufficient incentives…“requires proactive administration”Domestic pharmaceutical companies argue that a multidimensional approach to pricing incentives is necessary to achieve the government’s stated goal of shifting toward a new-drug-centered innovative pharmaceutical industry structure.They criticize that the MOHW’s efforts are severely lacking, noting that corresponding preferential regulations are necessary to encourage pharmaceutical companies to develop globally competitive, first-in-class blockbuster drugs with no substitutes.Suggestions followed that it is urgent to design a drug pricing system that comprehensively recognizes the value of domestic pharmaceutical companies, including establishing exclusive preferential regulations for domestic new drugs, creating an exception track for post-listing price reductions for domestic new drugs, and reducing the price reduction rate for generics that have significantly replaced originals and contributed to savings in the national health insurance budget.In essence, they argue that the government must address the criticism that “there are plenty of policies that cut prices, but none that add value.”Criticism that the MOHW focuses solely on price reductions and neglects efforts to establish preferential pricing regulations for domestically developed new drugs persists even in this latest drug pricing reform plan.In the latest reform, the MOHW included provisions to delay or defer price cuts for already-listed generics for innovative and quasi-innovative pharmaceutical companies, and to apply a premium to their reduction rates. It also included preferential provisions for innovative and quasi-innovative companies regarding newly listed drugsNevertheless, the pharmaceutical industry remains steadfast in its position that there are no preferential policies that adequately compensate for the fair value of domestically developed new drugs.This is because, despite the MOHW’s stated intention to restructure the domestic pharmaceutical industry around innovative new drugs through the drug price reform, there are no preferential policies in place for new drugs first developed by Korean pharmaceutical companies.In fact, in response to requests from the National Assembly and the industry to introduce exclusive incentives for domestic new drugs, the MOHW has repeatedly stated that such measures must be carefully reviewed due to potential trade conflicts with countries such as the United States.In contrast, Japan applies innovation and utility premiums to domestically developed drugs with proven clinical value, and also provides a priority introduction premium for new drugs approved for the first time in Japan.The pharmaceutical industry is demanding that if preferential drug pricing (premiums) at the time of listing is realistically impossible, a track be created to grant them exceptional benefits when the post-listing price reduction mechanism is applied. In other words, they are asking that domestic new drugs be excluded from price-volume agreement negotiations, price reductions based on actual transaction prices, and price reductions due to expanded indications, or that the reduction rates be adjusted so that these drugs can derive tangible benefits.Furthermore, they argued that the domestic pharmaceutical industry’s innovation could be supported by recognizing the value of generics, which have a high substitution rate for expensive original drugs, and lowering their price reduction rates, in line with the goal of saving national health insurance finances, which is the very reason for the existence of generics.For generics with usage rates exceeding 80–90% in prescriptions, confirming clear contributions to NHI savings, the industry requested the government to explore proactive measures to lessen price reductions.The National Assembly is also demanding the establishment of a clear differential compensation mechanism for pharmaceutical companies that have demonstrated contributions to the development of the pharmaceutical industry and investments in the value of new drugs, in order to foster the pharmaceutical and biotech industries.Rep. In-soon Nam of the Democratic Party of Korea stated, “The current drug pricing system provides insufficient returns relative to R&D investment. A differentiated pricing system based on factors such as R&D investment ratio, successful new drug development, and global exports is needed, including expanded incentives at listing or exemptions and reductions in price cuts.Lack of post-evaluation in pricing reform also an issueThe pharmaceutical industry also urged the MOHW to reflect on the historical trajectory of Korea’s drug pricing system and jointly set the future direction.Korea established the framework of its pricing system with the introduction of the Positive List System (PLS) in 2006 and the price-volume agreement (PVA), and has since operated the system with incremental adjustments for nearly 20 years.In November 2011, the government reinstated the joint bioequivalence testing system and implemented a blanket price cut in April 2012, applying a generic pricing rate of 53.55%. In 2020, it introduced differential pricing based on direct bioequivalence testing and DMF requirements, along with the 1+3 joint bioequivalence rule.Then, in November last year, the MOHW presented a new reform plan, stating that distortions caused by the proliferation of generics under past policies had reached a point where they could no longer be ignored.The MOHW’s rationale is that the problem has become entrenched, with pharmaceutical companies uniformly launching dozens to hundreds of generics containing the same active ingredient and focusing their efforts on sales and rebate competition for generics rather than innovative drugs, leaving the MOHW with no choice but to intervene.While the domestic industry partially agrees with this assessment, it points out that the root cause of the current multi-product generic structure lies in past government policies on approvals and pricing.In fact, when joint bioequivalence testing and across-the-board drug price cuts were implemented simultaneously in 2012, pharmaceutical companies sought to overcome declining profit margins by rushing into multi-product generic production. This triggered the first generic drug boom, resulting in a significant increase in the number of products despite short-term cost-saving effects.Every time the government implemented reforms to the approval and drug pricing systems, the number of domestic generic drug products increased significantly. (Korea Pharmaceutical and Bio-Pharma Manufacturers Association)According to the MFDS, newly approved generics nearly doubled from 727 in 2012 to 1,283 in 2013, and further increased to 1,684 in 2014 and 1,914 in 2015. The number of reimbursed generics also doubled from 1,094 in 2012 to 1,717 in 2013 and 2,359 in 2015.The 2020 reforms, including differential pricing based on criteria and restrictions on joint bioequivalence (1+3), triggered a second generic boom.At the time of the blanket price cut, the MOHW promoted the policy as a way to elevate Korea’s pharmaceutical industry to a global level. However, in reality, it resulted in the unintended consequence of an explosive increase in the number of generic products.ccordingly, academia and the pharmaceutical industry are calling for post-evaluation and reassessment of the MOHW’s past pricing policies aimed at reducing drug expenditure, curbing generic proliferation, fostering industry development, and promoting innovation.Professor Jong-hyuk Lee of Chung-Ang University College of Pharmacy stated, “It has been 20 years since the introduction of the positive list system. The government’s direction on managing new drug reimbursement and generic pricing is not wrong, but while the goal of addressing excessive multi-product listings is valid, we need to analyze whether the current reform will actually lead to increased R&D investment or reduced NHI expenditure.”Jae-hyun Lee, head of the Pharmaceutical Research Center at Sungkyunkwan University, added, “Both the 2012 blanket price cut and the current reform plan link generics and new drugs. While it is true that the government mobilizes various systems to achieve policy goals, it is necessary to evaluate and reflect on whether these systems were designed and implemented in accordance with their original intentions, and to make revisions accordingly.”Director Lee added, “It is undesirable for policymakers to design a system in a way that expects secondary effects on new drugs through generic drug pricing or aims to achieve other outcomes, such as eliminating rebates as a side effect. If generic drug prices are to be adjusted, the system must remain faithful to its original intent by limiting reductions and surcharges to generics alone. It is contradictory to adopt a half-hearted approach that allows innovative pharmaceutical companies to have their generic drug prices cut by a smaller margin.”
- Policy
- Can cutting generic prices really create new drugs?
- by Lee, Jeong-Hwan Mar 26, 2026 09:29am
- "It is unclear whether the policy objective of this drug price reform plan is to foster the domestic pharmaceutical industry or to rapidly introduce global new drugs from abroad. Will savings generated by lowering generic drug prices actually be used for domestic new drugs and robust domestic pharmaceutical companies, or end up flowing into the high-priced new drugs of multinational pharmaceutical companies? Can the government’s policy, which aims to create domestic pharmaceutical companies capable of producing blockbuster drugs by adjusting generic drug prices, truly be realized?"Although the Ministry of Health and Welfare has proposed a drug pricing reform aimed at shifting the domestic pharmaceutical industry toward innovation-driven new drug development by lowering generic prices and differentiating pricing for newly listed drugs by company, academia is questioning both its credibility and effectiveness.The concern is that if the government repeatedly implements across-the-board price cuts while merely touting the promotion of the pharmaceutical industry and the creation of domestic new drugs without presenting a detailed blueprint for where, how much, and how to allocate limited health insurance funds, it may ultimately result in savings from generics being used to reimburse high-cost global innovative drugs.At the same time, critics argue that the policy paradigm itself is flawed, as it attempts to generate innovative drugs that are rooted in basic and advanced science through partial adjustment of pricing policies for generics, which is a manufacturing-based industry.It is suggested that the MOHW must embark on a shift in its administrative paradigm, considering the need to design a two-track system that separates and distinguishes generic and new drug policies, in order to move closer to a rational policy that minimizes areas of conflict.On the 24th, the public health community criticized the ongoing backlash from the domestic pharmaceutical industry regarding the drug pricing reform plan announced by the MOHW for implementation this year, calling it “the result of an administration that has abandoned predictability.”While the MOHW cited “pharmaceutical industry structural innovation” as the rationale and justification for its drug pricing reform plan, which includes a drastic reduction in the pricing calculation rate for pre-listed generic drugs from 53.55% to the 40% range, the prevailing view among academics is that it is difficult to predict whether this will succeed.“The government’s concept of ‘industry promotion’ lacks clarity in both philosophy and target”Health economists and pharmaceutical pricing experts are questioning whether the MOHW has a clear philosophy for the rational management of drug expenditures within the national health insurance budget, and what the specific direction of its policy goal to foster the pharmaceutical industry actually is.They argue that it is unclear whether the “development of the pharmaceutical industry” mentioned by the MOHW refers to fostering the domestic pharmaceutical industry or to improving patient access through the rapid inclusion of original new drugs, many of which are produced by multinational pharmaceutical companies, for reimbursement in the national health insurance system.They also point out that the MOHW has not clearly defined how much pharmaceutical spending should be allowed within the insurance budget, how that spending should be allocated between prelisted generics and innovative new drugs, or how to address growing demands for coverage of ultra-high-priced drugs.Many scholars argue that tinkering with the drug pricing system in this manner without clarity makes it difficult to set precise policy goals, inevitably leading to the vague and ambiguous slogans of “fostering the pharmaceutical industry” and “reducing health insurance costs.”This is why concerns are being raised that the drug pricing reform plan recently proposed by the MOHW is disproportionately disadvantageous to domestic generic-focused pharmaceutical companies, while favoring multinational companies.Professor Jong-hyuk Lee of Chung-Ang University College of Pharmacy noted that the MOHW’s reform plan failed to align the levels of price reduction regulations imposed on generics and original drugs to a similar degree, despite its stated goal of addressing fairness between domestic and multinational companies.He argued that it is necessary to question whether the ministry is consistently lumping together generic drug regulations, which affect only domestic pharmaceutical companies, and patient access issues, which are directly linked to improved reimbursement rates for multinational companies’ new drugs, under the guise of “fostering the pharmaceutical industry.” The point is to demand transparency regarding the policy philosophy.“The Ministry’s definition of the pharmaceutical industry is vague. Domestic and multinational sectors are being lumped together. In reality, this reform reduces the profits of domestic companies through generic price cuts, potentially reallocating those resources to reimburse multinational companies’ innovative new drugs.”He continued, “While it is not desirable to draw a line between domestic and multinational companies, this drug pricing reform plan contains virtually no provisions that would cause losses for off-patent original drugs, and instead tends to include numerous regulations that are disadvantageous to domestic companies, such as generic price cuts. Even regarding post-listing control, the timing is being consolidated and adjusted, which will likely lead to situations where original drug prices are cut less frequently. Consequently, multinational companies are more likely to reap the actual benefits than domestic firms.”“The government must clearly present its philosophy on how pharmaceutical spending will be allocated and what industry fostering and development concretely means. It’s unclear how much savings will be generated from generic price cuts, and whether those funds will be reinvested in domestic companies or used to expand access to innovative drugs. This is precisely why the industry is calling for joint research with the government, to clarify these uncertainties.”Linking generic drug prices to new drug development the right approach?… time to rethink the administrative paradigm”Some point out that the MOHW’s current administrative approach, which links generic drug pricing policies with the creation of innovative new drugs, is bound to lead to inevitable contradictions.The argument is that generics should be viewed from the perspective of protecting manufacturing, a traditional industry, and public health security, while new drugs should be supported through an environment that fully fosters basic and advanced science; however, linking the two often leads to conflicts.Rather than simply maintaining the current approach, where the MOHW adjusts generic drug prices to encourage more pharmaceutical companies to focus on innovative drug development, the government needs to consider a paradigm shift, such as adopting a two-track administrative system that separates policies for generics and innovative drugs.In essence, the purpose of generic drug pricing is to ensure a stable supply of cost-effective medications to the public within the constraints of limited national health insurance funds. Cost control through price reductions and management, along with the stabilization of quality and supply, are the core elements of the system.Conversely, fostering the development of new drugs hinges on pharmaceutical companies making bold investments in high-risk, high-return clinical trial R&D and strengthening the fundamental capabilities of basic life sciences, such as chemistry, biology, and genetics.Accordingly, the academic community urges the government to establish a nuanced drug pricing system that allows the generic industry to grow, based on its understanding of the value of generics, while simultaneously implementing a separate administrative framework that fully supports the creation of innovative new drugs, independent of the generic sector.Jae-hyun Lee, Director of the Pharmaceutical Regulatory Science Center at Sungkyunkwan University, stated, “The government needs to better understand the role and value of the generic industry. Generic pricing policy should focus on creating an environment where companies can sustainably produce high-quality, affordable generics. Trying to promote innovation through generic pricing is fundamentally contradictory.”Lee explained, “This means the drug pricing system should focus solely on creating an environment where pharmaceutical companies strive to manufacture generics properly. Instead of the current approach of uniformly lowering drug prices, we need a system that differentially adjusts prices by comparing the average price per therapeutic class or ingredient with international averages.He also criticized the reform for lacking predictability from the perspective of pharmaceutical companies.“Government policy must ensure transparency and predictability. Without predictability, companies will resort to workarounds—such as increasing prescription volume to offset price cuts. Japan gradually reduced prices to around 40% over a decade, allowing companies time to adjust. Korea should also adopt a similar phased approach and give companies time to prepare.”Industry voices the government’s lack of understanding on the value of genericsDomestic pharmaceutical companies also argue that the government significantly undervalues the role of generics.They complain that every pricing reform is built on the assumption of across-the-board generic price cuts, inevitably leading to repeated government and industry conflict.They further point out that generics play a critical role in healthcare security and should be actively supported by the government, rather than subject to one-sided cost-cutting policies that fail to adequately consider the difficulties faced by the pharmaceutical industry, including rising manufacturing costs, such as raw materials and labor expenses.An administration focused solely on generic drug regulations, such as price cuts, will ultimately encourage the use of cheap, low-quality raw materials and lead to problems such as the abandonment of production of essential drugs with low profitability, thereby deepening reliance on imported generics.Young-joo Kim, Chair of the Policy Planning Committee for the Emergency Task Force on Drug Price System Reform, stated, “Since generics account for about 50% of South Korea’s pharmaceutical and biotech industry, lowering drug prices will immediately trigger a massive decline in sales. This threatens the survival of pharmaceutical companies. leading to reduced R&D investment due to declining profitability, difficulties in retaining top-tier research talent, and workforce restructuring resulting from production cuts.”“Korea currently manufactures and supplies most generics domestically, which ensures quality control and supply stability while also funding R&D. In contrast, countries that aggressively cut generic prices now rely heavily on imports, leading to various issues such as frequent stockouts, quality control issues leading to fatalities, and supply instability.”A pricing official from a leading domestic pharmaceutical company added, “The government should move beyond abstract slogans like ‘industry advancement’ and properly recognize the value of domestic generics in its pricing policies. There needs to be clear criteria to reward companies that consistently produce and supply high-quality generics, along with a governance system that works in partnership with the industry,” he said.The official concluded, “The negative effects of blanket price cuts are already evident, such as the collapse of the domestic API manufacturing industry. If the government insists on linking generics and innovation, then companies that demonstrate true innovation should be fully exempt from price cuts. Without such changes, conflicts between the industry and the government will persist.”
- Policy
- Criticism over MFDS’s delayed crackdown on oral albumin
- by Lee, Tak-Sun Mar 26, 2026 09:29am
- Although the Ministry of Food and Drug Safety announced a planned inspection targeting false and exaggerated advertising of ‘oral albumin’ products, the market is criticizing the move as a ‘belated response’ that came only after consumer harm had already occurred.At the time the issue was first raised earlier this year by media outlets and civic groups, sales had already peaked, and illegal advertising intensified further during the Lunar New Year holiday season. Critics argue that the MFDS should have officially announced the inspection earlier through media channels to curb illegal market practices.AI-generated imageMFDS’s “Emergency Response Team for Unfair Food Practices,” which officially launched on the 24th, announced a focused inspection of unfair advertising for albumin-containing foods as its first initiative. Although albumin derived from eggs or milk is broken down into amino acids upon consumption and does not directly increase blood albumin levels, false advertisements have been rampant on home shopping channels and social media, claiming it has the same effects as the pharmaceutical ‘serum albumin.’In particular, as of December last year, albumin products ranked first in the number of home shopping health product broadcasts, and some products sold out more than 10 times, showing explosive sales. Despite being a high-priced product costing around KRW 100,000 per month, it successfully targeted consumers eager to recover from fatigue and boost their immunity.In January, Dailypharm had already published an in-depth report on the harms of oral albumin supplements, followed by similar media coverage.As the MFDS response remained insufficient despite media reports, civic groups and the medical community called for stronger action. On the 20th, the consumer advocacy group Consumers Together issued a statement calling it “a nationwide fraud that packages ordinary food without proven medical efficacy as if it had therapeutic effects. We ask the MFDS to conduct a full investigation into false advertising and impose strict penalties under a zero-tolerance principle.”Earlier, the Korean Medical Association also raised concerns, stating that “there is no clinical evidence supporting oral albumin.” It further indicated that it is considering referring so-called ‘show doctors’ who appeared in misleading advertisements by exploiting their professional authority, to the Ethics Committee and recommending disciplinary action.“Only acting after the peak?”… industry criticism growsIt was only after the KMA and consumer groups issued strong demands that the MFDS officially announced its first planned inspection of oral albumin products through its Emergency Response Team for Unfair Food Practices. However, the pharmaceutical sector and related industries are questioning the practicality of this planned inspection, as large-scale sales had already taken place during peak seasons such as the Lunar New Year.According to the distribution industry, many companies have already generated substantial profits and entered a discount phase to clear remaining inventory following the crackdown announcement. A pharmacist who requested anonymity criticized, “By the time the MFDS declared enforcement, companies had already sold most of their stock,” calling it “a textbook case of a belated response.” An industry official also pointed out that “At the very least, targeted inspections should have been conducted before the Lunar New Year, when sales peak.”In response to criticism of delayed action, the MFDS stated, “We have continuously conducted crackdowns on misleading advertisements for oral albumin products. The launch of this emergency response team is aimed at tackling overall unfair food practices, and is not a targeted inspection specifically for oral albumin.”However, criticism has emerged that there have been no officially planned inspections regarding false advertising of edible albumin products to date, and that existing crackdowns have relied primarily on online post-hoc monitoring. This suggests that there are limitations in responding to real-time advertisements on home shopping channels or social media.While the MFDS aims to overcome these limitations through the new emergency response team, it is expected to face growing accountability issues over consumer damage that has already occurred.
- Policy
- Pharma companies may suffer KRW 600B annual loss shock
- by Lee, Jeong-Hwan Mar 25, 2026 07:21am
- The Ministry of Health and Welfare (MOHW)'s 'drug pricing system reform plan' to raise the low-price purchasing incentive rate for hospitals and pharmacies from 20% to 35%. Concerns about pharmaceutical industry shock.The government's 'drug pricing system reform plan' to raise the low-price purchasing incentive rate for hospitals and pharmacies from 20% to 35% is facing criticism. Critics pointed out that it is an administrative move that will increase pharmaceutical companies' losses.The pharmaceutical industry is already enduring approximately KRW 350 billion in annual revenue losses under the 20% incentive system. Concerns are rising that if the government transitions to a market-linked actual transaction price system by expanding the incentive rate to 35%, annual losses will exceed KRW 600 billion, inevitably causing market contraction.Given that the government already implements price cuts for existing generic drugs, analysis suggests that raising the low-price purchasing incentive rate will further worsen pharmaceutical companies' profitability, potentially threatening the production and stable supply of essential medicines.On the 24th, the Ministry of Health and Welfare (MOHW) announced a regulation raising the incentive rate paid to medical institutions and pharmacies for low-priced purchasing from 20% to 35% as part of the actual transaction price-based price reduction method. This reform aims to restructure the current system into a market-linked actual transaction price model.The low-price purchasing incentive is a system in which, if a medical institution or pharmacy purchases a drug from a pharmaceutical company at a price below the government-set insurance ceiling price, a portion of those savings is returned to the institution as an incentive.Under the current 20% rate, if a hospital purchases a drug at KRW 100 below the reference price (ceiling price), it receives a KRW 20 incentive.While systemically designed to improve purchasing efficiency and save the National Health Insurance budget, the system has been criticized for pressuring pharmaceutical companies to reduce prices.The pharmaceutical industry estimates that current losses amount to KRW 350 billion annually under the 20% rate. Based on this, pharmaceutical industry argues that a 35% incentive rate would result in annual drug revenue losses to KRW 600 billion or more.Furthermore, assessments suggest that if the price-reduction mechanism for actual transaction price investigations fails to operate properly despite the 35% rate, pharmaceutical companies could face actual losses exceeding these calculations.Concerns are rising that the overall profit base of the pharmaceutical industry could be destabilized if additional price pressures, such as the increased incentive rate, are added to the Ministry's general policy of cutting prices for listed drugs.The MOHW's proposal to raise the incentive rate (from 20% to 35%) is seen as a measure that strengthens the mechanism for transferring price-cutting pressure to manufacturers. Hospitals and pharmacies will have a greater incentive to purchase drugs at the lowest possible prices to secure larger rewards, which may put pressure on pharmaceutical companies to lower their supply prices.An official from a mid-sized Korean pharmaceutical company stated, "While the official insurance drug price may remain the same, the structure forces actual transaction prices to keep falling. If the incentive rate is raised, hospitals, which have more authority, will demand stronger price cuts. Pharmaceutical companies may face significant burdens."The official added, "The current actual transaction price reduction system and the 20% incentive rate must be maintained so that it can alleviate the excessive burden on the industry," and emphasized, "Raising the incentive rate exacerbates the issue of increased losses, especially when a pharmaceutical company sells more. This may ultimately force certain companies to abandon the stable supply of essential medicines."
- Policy
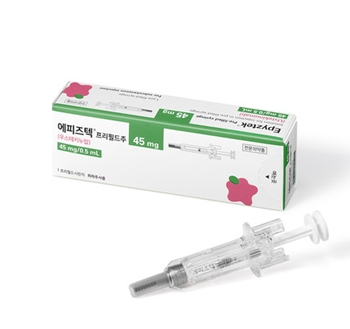
- Stelara biosimilar ‘Epyztek Pen’ listed for reimb next month
- by Jung, Heung-Jun Mar 25, 2026 07:21am
- The pen formulation of Samsung Bioepis’s autoimmune disease treatment Epyztek (ustekinumab) is scheduled to become the first reimbursed product of its ingredient class next month.Samsung Bioepis appears to be pursuing a differentiation strategy with a pen formulation that even the original product, Janssen Korea’s ‘Stelara,’ has neither received approval nor reimbursement for.According to industry sources on the 24th, Samsung Bioepis will expand its reimbursement lineup next month with a prefilled pen formulation of its Stelara biosimilar Epyztek.Like Stelara, Epyztek is an autoimmune treatment indicated for plaque psoriasis, psoriatic arthritis, Crohn’s disease, and ulcerative colitis. In South Korea, it competes in the market alongside Celltrion’s biosimilar ‘Steqeyma’ and others.The pen formulation, scheduled for listing next month, allows patients to administer the medication conveniently and accurately, giving it a competitive edge in self-administration settings.Stelara is currently approved only in intravenous, subcutaneous, and prefilled syringe formulations, making the prefilled pen the first of its kind among products with the same active ingredient. Samsung Bioepis secured its place on the reimbursement list just 3 months after receiving approval from the MFDS last January.This preemptive addition of a new formulation is expected to continue its offensive not only against biosimilar competitors but also in the battle for market share against the original product.Samsung Bioepis is targeting the market with a price lower than the original. The newly listed Epyztek Prefilled Pen 90 mg/1 mL is expected to receive an insurance ceiling price in the KRW 1.27 million range.The original Stelara Prefilled Syringe 45 mg/0.5 mL is priced at KRW 1,745,600, while Epyztek Prefilled Syringe at the same dose is about 30% cheaper at KRW 1,233,376. The pen formulation offers double the dosage but is priced about 27% lower.Leveraging its lower price and the addition of new formulations, the company is expected to begin a full-scale push to expand its market share starting in the second quarter of this year.Samsung Bioepis sells Epyztek (known as Pyzchiva in the U.S. and Europe) not only in Korea but also in Europe and the U.S. through its partner, Sandoz.
- Policy
- Anticancer drugs account for record high share of pharma expenditures
- by Jung, Heung-Jun Mar 24, 2026 08:16am
- Claims for anticancer drugs are rising steeply within Korea’s national health insurance pharmaceutical expenditures. It exceeded KRW 3 trillion the year before last, surpassing anti-atherosclerosis drugs for the first time.While total drug expenditures rose by 5.6%, claims for anticancer drugs climbed by 15%, the largest increase by far among the 5 five therapeutic classes in terms of spending.Anticancer drug claims exceed KRW 3 trillionAccording to the 2024 Status of Expenditures on Reimbursed Drugs Report released by the National Health Insurance Service on the 23rd, health insurance drug expenditures totaled KRW 27.6625 trillion, a 5.6% increase from the previous year’s KRW 26.1966 trillion.Drug costs accounted for 23.8% of total healthcare expenditure of KRW 116.2375 trillion. As of 2023, the proportion of drug expenditures in Korea’s current health expenditure was 19.4%, which is 5 percentage points higher than the OECD average of 14.4%.Among major overseas countries (A8) that Korea uses for drug price references, Korea’s spending ratio was also higher than Japan (17.6%), Germany (13.7%), and the UK (9.7%).◆ Anticancer drug claims reach KRW 3.1432 trillion... share hits record high of 11.4%Claims and spending share for anticancer agents reached all-time highs. Claims totaled KRW 3.1432 trillion in the year before last, up 15% from KRW 2.7336 trillion the previous year.The top 5 therapeutic classes accounted for 40.4% of total drug expenditure, with anticancer drugs holding the largest share at 11.4%. Until now, anti-atherosclerotic drugs had previously held the highest claim amount and market share, but anticancer drugs rose to the top spot for the first time.Drug costs for cancer patients totaled KRW 4.2958 trillion, an 11.9% increase from the previous year. This marks the largest growth rate in the past 5 years.◆ By ingredient, “ezetimibe + rosuvastatin” ranked highest…up 16% to exceed KRW 700 billionBy ingredient, spending on ezetimibe + rosuvastatin rose 16.3% year on year, reaching KRW 704.6 billion.Choline alfoscerate came in second at KRW 557.6 billion, down 1% from the previous year. Atorvastatin also declined slightly, with claims of KRW 554.3 billion, down 0.8%. The antiplatelet agent clopidogrel reached KRW 441.8 billion, up 5.7%.◆ Decline in original drug claims...generic share rises to 44.4%Original drug claims showed a declining trend, while generic claims increased. In the year before last, spending on original drugs amounted to KRW 15.3434 trillion, accounting for 55.6% of total pharmaceutical expenditure, down from 59.1% the year before.In contrast, generic drug claims totaled KRW 12.2591 trillion. This accounted for 44.4% of total drug expenditure, an increase from 40.9% the previous year.Since 2021, the spending share of original drugs has continued to decline while that of generics has risen, narrowing the gap in reimbursement claims between the two categories.An NHIS official said, “In line with government policy direction, we will further refine implementation measures and actively support the execution of tasks for the benefit of the public and the development of the pharmaceutical industry, while making every effort to ease patients’ drug cost burdens and ensure the financial sustainability of the National Health Insurance system.”
- Policy
- Will Mifegyne gain momentum for Korean entry?
- by Lee, Jeong-Hwan Mar 24, 2026 08:16am
- The domestic introduction of Mifegyne, the abortion drug whose marketing approval by the Ministry of Food and Drug Safety has been delayed for a long time, is expected to gain momentum.This follows recent remarks by Yong-jin Park, vice chair of the Regulatory Reform Committee, who identified the delay in Mifegyne’s approval as a prime example of irrational administrative regulation and announced his intention to work toward a solution.Vice Chair Park is reportedly determined to ensure domestic approval through administrative action even before legislation is enacted, given that the introduction of Mifegyne is regarded as a matter of national policy.Vice Chair Yong-jin ParkVice Chair Park views the issue of Mifegyne’s introduction from a global standards perspective. He argues that the situation where only Korea is blocking a drug already permitted in over 100 countries worldwide, including OECD nations, solely due to administrative procedures, represents an irrational regulation unique to Korea.Vice Chair Park believes that the reality where women are forced to purchase the drug through underground channels despite its globally proven safety due to administrative barriers threatens women’s health rights and must not be ignored.The MFDS has maintained a cautious stance on approving Mifegyne on the grounds that follow-up legislation has not yet been made since the Constitutional Court’s 2019 decision declaring the abortion law unconstitutional.Vice Chair Park believes that if the MFDS takes a more forward-looking approach to the regulatory barriers it has tied itself to, a solution can be found.This effectively suggests the need to expedite the timing of domestic approval and introduction through proactive government action, even before the legislature completes the relevant process.Attention is now focused on whether the drug could be approved before the National Assembly passes a proposed revision to the Mother and Child Health Act, introduced by Democratic Party of Korea lawmaker In-soon Nam and others, that would allow medication-induced abortion.If the MFDS adopts administrative regulations or establishes a separate approval process, the official introduction of Mifegyne in Korea will gain momentum.An industry official stated, “If Vice Chairman Park’s plan to ease administrative regulations is effectively implemented, the MFDS will be able to exercise proactive administration even amid a legislative vacuum. It would become a representative example of the government putting forward a preemptive alternative at the administrative level.”