- LOGIN
- MemberShip
- 2026-04-24 08:27:16
- Policy
- Multiple myeloma drug 'Blenrep inj' seeking reimbursement
- by Jung, Heung-Jun Apr 08, 2026 07:47am
- GSK Korea's Blenrep inj (belantamab mafodotin), a new treatment for multiple myeloma, has officially entered the race for health insurance reimbursement listing.It has been just three months since receiving marketing authorization in December of last year as a designated new drug under the Global Innovative Product on Fast Track (GIFT) system.According to industry sources on the 8th, GSK Korea recently submitted an application to the Health Insurance Review and Assessment Service (HIRA) for the determination of reimbursement for Blenrep.Blenrep is a first-in-class Antibody-Drug Conjugate (ADC) targeting the B-cell maturation antigen (BCMA), which is overexpressed on the surface of multiple myeloma cancer cells. It induces cell death by releasing cytotoxic agents directly into the cancer cells. It has been approved as a second-line treatment for adult patients with relapsed or refractory multiple myeloma who have received at least one prior therapy.Blenrep holds clinical value by expanding treatment options for hematologic malignancies in an ADC market that has been focused on solid tumors in recent years, represented by drugs like Enhertu and Trodelvy.The approved indications in Korea are not for monotherapy but for combination therapies with ▲bortezomib and dexamethasone ▲ pomalidomide and dexamethasone.While the Central Pharmaceutical Affairs Council (CPAC) of the Ministry of Food and Drug Safety (MFDS) previously raised concerns regarding ocular toxicity, it concluded that the clinical benefits outweigh the risks. Consequently, the approval included management conditions, including regular ophthalmic examinations and the use of monitoring checklists.The approved dosage and administration also include requirements for eye exams and dose adjustments to manage adverse reactions. The upcoming reviews by the Cancer Disease Review Committee and the Pharmaceutical Reimbursement Evaluation Committee are expected to involve a thorough examination of reimbursement criteria and verification of cost-effectiveness in light of these requirements.Since this drug was approved as a second-line combination therapy to be used alongside existing anticancer drugs, analyzing the impact on National Health Insurance finances due to increased total treatment costs and negotiating Risk Sharing Agreements (RSA) are expected to be significant challenges in the listing process.
- Policy
- Government to prevent drug supply disruptions amid Middle East tensions
- by Jung, Heung-Jun Apr 07, 2026 07:22am
- Concerned that geopolitical risks in the Middle East could escalate into instability in the drug supply chain, the government is simultaneously pursuing regulatory improvements and conducting a survey of the current supply situation.This is because logistical disruptions involving raw materials and packaging materials could affect the domestic drug supply. In particular, there are concerns that shortages of petrochemical raw materials, such as naphtha, could exacerbate packaging supply issues.On the 3rd, the Pharmaceutical Supply Management Division of the Health Insurance Review and Assessment Service (HIRA) launched a survey on pharmaceutical supply status through the Korea Pharmaceutical and Bio-Pharma Manufacturers Association.The aim is to identify which drugs are being affected by import delays and supply disruptions due to risks in the Middle East. HIRA plans to use this information to proactively prepare countermeasures.HIRA is monitoring drug distribution inventory levels through the Korea Pharmaceutical Information Center. However, there is a limitation in that, while it has data on domestically distributed drugs, it is difficult to predict supply disruptions caused by import and logistics delays.HIRA is expected to compile information from pharmaceutical companies, including product names, packaging materials, and inventory levels, by next week and use this as a basis for formulating strategies such as future drug price management and securing alternative medications.If disruptions in the logistics networks for raw materials and packaging materials persist, the impact on domestic pharmaceutical companies’ production costs and supply stability could increase even further. Consequently, the government appears to be taking preemptive action before supply instability becomes a reality.The Ministry of Food and Drug Safety also discussed countermeasures last week with three companies supplying basic IV fluids and introduced unprecedented regulatory easing measures, including a fast-track system for approval changes.The measures were announced on the 3rd during a joint meeting of the Emergency Economic Measures Headquarters Meeting and Ministerial Meeting on Economic Affairs. The government announced regulatory easing measures. The MFDS lowered regulatory barriers to enable pharmaceutical and medical device manufacturers to diversify their supply chains for raw materials and packaging materials, such as IV solutions and syringe needles.First, in cases where changes to product approval are required due to shortages of petrochemical raw materials such as naphtha, a 'fast-track system' has been newly established to prioritize review over other items.Additionally, for medical devices, the agency allowed manufacturers to make changes to packaging materials to undergo document reviews instead of on-site GMP inspections.This is intended to reduce the one to two months it typically takes for pharmaceutical companies to complete product license amendment reviews. The measures to shorten review periods will be implemented immediately as part of the government’s proactive administration initiative.
- Policy
- Silymarin to undergo reimbursement reevaluations this year
- by Jung, Heung-Jun Apr 07, 2026 07:22am
- The ingredient 'silymarin (milk thistle extract),' which was the subject of a lawsuit after the company contested the results of the 2021 reimbursement reevaluation, is finally being put to the test once again. It is scheduled to be included in this year’s list of items subject to re-evaluation, alongside ginkgo biloba and dobesilate, which were previously on the list.According to industry sources on the 6th, the Health Insurance Review and Assessment Service (HIRA) Drug Reimbursement Evaluation Committee recently discussed a proposal to add silymarin to the list of items subject to reevaluation.In the 2021 reimbursement reevaluation, which also included dried bilberry extract and Avocado-soya unsaponifiables, silymarin was deemed inappropriate for reimbursement. In November of that year, the Ministry of Health and Welfare announced its decision to delist.Pharmaceutical companies filed injunction requests and administrative lawsuits. While the first trial in November 2023 upheld the reimbursement withdrawal, the appellate court overturned the decision in December last year, ruling in favor of the companies. The ruling became final after the Ministry of Health and Welfare chose not to appeal.Based on a review of the appellate court’s ruling, it appears that the direction has been set to conduct a re-evaluation of reimbursement appropriateness rather than delisting.This is interpreted as the court having pointed out procedural flaws in the reevaluation process rather than having judged the clinical utility of the drugs.Seven ingredients, including ginkgo biloba extract and calcium dobesilate, had already been discussed for reevaluation this year. It is reported that there have been changes to some of the other items. Although the agenda for items subject to reimbursement reevaluation was submitted to the Drug Reimbursement Evaluation Committee last January, the Health Insurance Policy Deliberation Committee has not yet reached a decision.Generally, items for reimbursement reevaluation are decided by the Health Insurance Policy Deliberation Committee immediately after being submitted to the Drug Reimbursement Evaluation Committee, but the process has been delayed due to the drug pricing reform, which includes changes to the reimbursement reevaluation criteria.Since the drug pricing reform plan was finalized at the March Health Insurance Policy Deliberation Committee meeting, the items are expected to be submitted to the committee this month.Furthermore, the items to be reevaluated next year remain unclear. Because the drug pricing reform has changed the annual reimbursement reevaluation to be conducted on an as-needed basis, the decision on items for 2027 will require further observation.
- Policy
- INN prescribing bill may be resubmitted to April subcommittee
- by Lee, Jeong-Hwan Apr 06, 2026 03:51pm
- 소병훈 보건복지위원장Attention is mounting within the healthcare and pharmaceutical sectors on whether a bill mandating limited international nonproprietary name (INN) prescribing for essential medicines or drugs with unstable supply will be reviewed by the National Assembly’s Health and Welfare Committee’s legislative subcommittee in April.The bill was included on the agenda of the Health and Welfare Committee’s legislative subcommittee last March, but did not get a chance to be reviewed as it was pushed back by other bills.On the 3rd, lawmakers from the Democratic Party of Korea have emphasized the need to convene the subcommittee in April to discuss bills under their purview that were not reviewed last month.As it is highly unlikely that the subcommittee will convene in May, just one month before the June 3 local elections, the Democratic Party of Korea members of the Welfare Committee saw a strong need to expedite the review of bills this month.Currently, the Health and Welfare Committee is led by Rep. Byung-hoon So (Gwangju-gap, Gyeonggi Province; third-term), who took over as the new chair following the resignation of former Chair Rep. Ju-min Park, who stepped down to run in the Democratic Party’s primary for the Seoul mayoral race.Accordingly, Chair Byung-hoon So, Democratic Party Executive Secretary Soo-jin Lee, and People Power Party Executive Secretary Mi-ae Kim are expected to begin discussions on the schedule for the April subcommittee meeting.Once the subcommittee meeting is confirmed, the bill mandating the limited use of INN prescribing, which was not reviewed last month, is highly likely to be placed on the agenda.Last month, on the day of the subcommittee meeting, the Korean Medical Association (KMA) held a “Rally to Block INN Prescriptions” on the steps in front of the National Assembly Main Building to pressure the Health and Welfare Committee.At that time, KMA President Taek-woo Kim made it clear that the association would take strong collective action if the bill were to be formally introduced.KMA President Taek-woo Kim led the rally to block the INN prescribing bill on the steps in front of the National Assembly Main Building on the 11th of last month.In particular, President Kim maintains a hardline stance that the bill must not be tabled at the April subcommittee meeting. The KMA is currently using the threat of a general doctors’ rally as leverage to block the review of the limited mandatory INN prescribing bill.However, the Democratic Party argues that the INN prescribing bill is part of President Lee’s campaign pledge and a national policy agenda adopted after his inauguration. They maintain that legislation is necessary to improve public access to medicines.A committee official stated, “The Democratic Party is calling for the need to convene the April subcommittee, but whether it will actually take place has not yet been finalized. It will be determined soon following consultations between the ruling and opposition party floor leaders, taking into account the schedule for the June 3 local elections.”There are three bills related to limited INN prescribing currently pending in the National Assembly: an amendment to the Pharmaceutical Affairs Act proposed by Rep. Yoon Kim of the Democratic Party of Korea, and amendments to the Medical Service Act and Pharmaceutical Affairs Act proposed by Rep. Jong-tae Jang of the same party.
- Policy
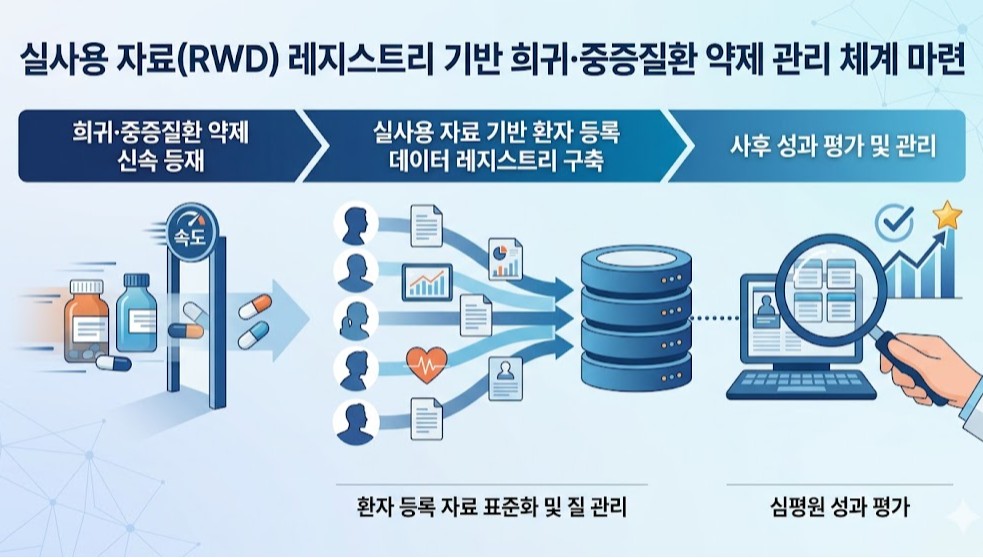
- Korea to use RWD for post-listing control of expedited drugs
- by Jung, Heung-Jun Apr 06, 2026 03:51pm
- A plan is being developed to utilize patient registry data, a subset of real-world data (RWD), to strengthen post-listing management of rare and severe disease treatments.This is an extension of the drug pricing system reform announced by the government last month through the National Health Insurance Policy Deliberation Committee. Previously, the government had revealed plans to expedite the listing of treatments for rare and severe diseases and then reevaluate their reimbursement status based on real-world data.According to industry sources on the 6th, HIRA and the Pharmacuetical Performance Assessment Department will conduct a study this year to establish an RWD registry-based management system. A registry refers to patient-level data collected by disease or drug.Although a call for proposals has not yet been issued, the research is scheduled to be completed by the end of this year.Following last year’s study on RWE (Real-World Evidence) guidelines for drug performance assessment, the government is now moving to establish a management system based on RWD registries.This research is significant as it moves beyond the conceptual definition stage towards building an actionable infrastructure. It also serves as a key follow-up measure for implementing the government’s drug pricing system reform.This is because a patient data management system must be established to implement measures such as performance-based reimbursement, adjustments to reimbursement scope, or drug price based on post-marketing evaluation results using real-world data.A HIRA official stated, “Building a registry is essential for managing rare disease treatments. We will conduct policy research on how to actually build such a system. I expect results to be available by the end of the year, following approximately 6 months of research.”The official further explained, “While this can be seen as an extension of the drug pricing system reform, there has long been a consensus that a registry is essential for establishing a management system for rare and severe disease drugs.”However, patient or disease-specific registration data in clinical settings have not yet been standardized, and concerns regarding proper quality control remain unresolved. These issues are expected to be key points of contention during the process of establishing a registry-based management system.An HIRA official stated, “We plan to commission the research soon. We ask researchers to show strong interest, given the high accessibility and utility of the registry data.”
- Policy
- Study to improve ICER threshold underway
- by Jung, Heung-Jun Apr 02, 2026 08:46am
- With the government announcing plans to raise the ICER threshold next year, detailed reform measures are expected to be prepared by October this year.For this, research aimed at ensuring fiscal sustainability while realigning cost-effectiveness evaluation criteria to become more realistic will be conducted over approximately 6 months.On the 31st, the Health Insurance Review and Assessment Service (HIRA) issued a request for proposal for a research project titled “Establishing rational cost-effectiveness evaluation criteria for listing new drugs under the national health insurance system.”This study was launched in response to criticism that the current ICER threshold fails to reflect ▲rising national income and inflation ▲demands for enhanced patient coverage ▲trends in innovative drug development.Through this research, a model for calculating the ICER threshold that incorporates socioeconomic factors such as income and inflation will be developed. A weighting system that takes disease severity, therapeutic benefits, and fiscal impact into account will also be designed.The study will also explore ways to systematize and categorize ICER thresholds and develop models for applying evaluation criteria to reimbursement decisions through pilot implementation.Additionally, the government will review the introduction of a system for periodically adjusting ICER thresholds by referencing major countries. It also plans to gather input from stakeholders, including the pharmaceutical industry, patient groups, and academia.HIRA plans to sign the research service agreement between March and April, conduct an interim review, and complete the study by October. The total budget for the project is KRW 60 million.Through this study, the agency expects to establish rational cost-effectiveness evaluation criteria, thereby improving patient access to treatment and incentivizing new drug development. It also aims to build trust among pharmaceutical companies, patients, and the government by establishing clear standards and procedures.Previously, the Ministry of Health and Welfare announced plans to raise the ICER threshold next year through reforms to the drug pricing system. It decided to introduce a weighting system to allow for the flexible application of ICER values. The ministry plans to conduct policy research this year and implement a rational plan in 2027 based on the results.The government is expected to finalize detailed implementation plans based on the research results, which are due around October, and proceed with full-scale adjustments to the ICER threshold sometime next year.
- Policy
- Hair loss drug finasteride, sexual dysfunction-linked suicidal ideation warning
- by Lee, Tak-Sun Apr 02, 2026 08:46am
- AI-generated imageThe labeling for finasteride 1mg tablets, used in the treatment of hair loss, will now include warnings regarding sexual dysfunction side effects.For dutasteride, another hair loss treatment, a new "General Precautions" section will be added to include information on conditions such as depression.The Ministry of Food and Drug Safety (MFDS) recently announced that it has prepared these proposed label changes based on European Medicines Agency (EMA)'s results of a safety information review and has requested feedback from companies by the 10th.According to the revised labeling, a new statement will be added to the warnings section for finasteride 1mg tablets as "Sexual dysfunction that may affect mood changes, including suicidal ideation, has been reported in some patients. Patients should be advised to seek medical consultation if sexual dysfunction occurs. Consideration should be given to whether treatment should be discontinued."While the existing warnings section already mentioned reports of depression, it did not include specific details such as suicidal ideation resulting from sexual dysfunction, as specified in the newly added text.For dutasteride formulations, information regarding 'mood changes and depression' was added to the "General Precautions" section, which is a lower-tier alert than a warning.The newly added information states: "Mood changes (including depressed mood, depression, and rarely, suicidal ideation) have been reported in patients treated with other oral 5-alpha reductase inhibitors. Patients should be advised to seek medical consultation if such symptoms occur."For finasteride 1mg tablets, sexual dysfunction-related adverse events such as decreased libido and erectile dysfunction have already been frequently reported. They are specified in the "Adverse Reactions" section of the labeling. However, details regarding suicidal ideation linked to sexual dysfunction were previously absent.The target products for this label change include 94 items of finasteride 1mg, including the original drug Propecia 1mg.Additionally, for dutasteride, 98 items are subjected to changes, including the original drug Avodart Soft Cap 0.5mg.Meanwhile, in May of last year, the EMA specified suicidal ideation as a new adverse effect in the product information for finasteride. The EMA stated, "Cases have been included where patients complained of suicidal ideation alongside sexual dysfunction such as depression, decreased libido, and erectile dysfunction."The EMA recommended adding a warning statement regarding mood changes and suicidal ideation to dutasteride, a 5-alpha reductase inhibitor of the same class, as a precautionary measure. The EMA stated that, for dutasteride, the evidence does not currently provide a clear causal relationship, unlike that of finasteride.
- Policy
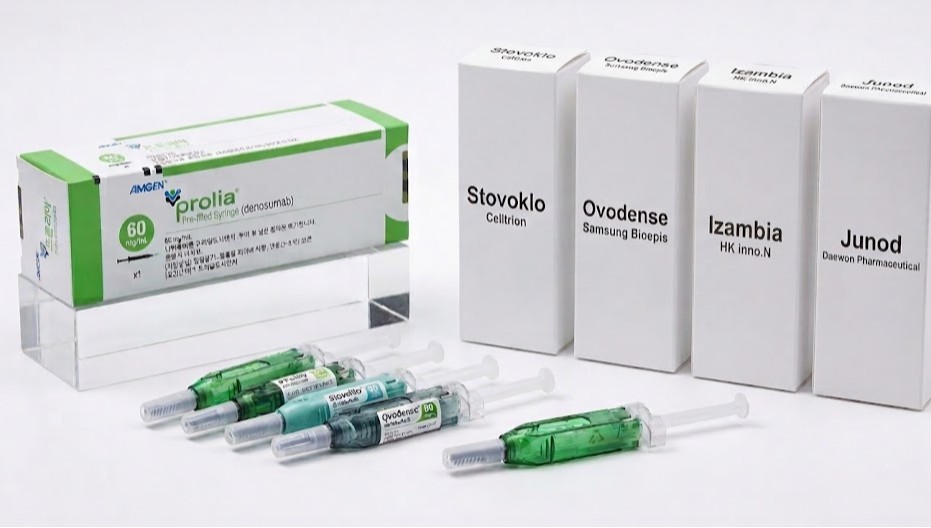
- Daewon Pharm joins the Prolia biosimilar competition
- by Lee, Tak-Sun Apr 01, 2026 08:15am
- AI-generated image (Prolia, Stoboclo, Obodence, Izambia, Junod)A series of biosimilars with the same active ingredient as Amgen’s osteoporosis drug Prolia are being approved. With Daewon Pharm joining the race, 5 products have now received marketing authorization in Korea.Prolia is an osteoporosis treatment administered as a subcutaneous injection once every 6 months, offering strong bone density improvement and fracture prevention effects.In Korea, it has dominated the market with annual sales of approximately KRW 180 billion through copromotion with Chong Kun Dang.On the 31st, the Ministry of Food and Drug Safety approved Daewon Pharma’s Prolia biosimilar, ‘Junod Prefilled Syringe.’This product is indicated for: ▲ the treatment of postmenopausal women with osteoporosis, ▲ the treatment of men with osteoporosis to increase bone density, ▲ the treatment of glucocorticoid-induced osteoporosis, ▲ the treatment of bone loss in patients with non-metastatic prostate cancer receiving androgen deprivation therapy, and ▲ the treatment of bone loss in women with breast cancer receiving adjuvant aromatase inhibitor therapy.It is administered via subcutaneous injection 6 six months in the upper arm, upper thigh, or abdomen, identical to Amgen’s Prolia. The product demonstrated equivalence to Prolia through Phase I and Phase III trials.Juno is a biosimilar developed by Gedeon Richter, headquartered in Budapest, Hungary.Gedeon Richter is a multinational pharmaceutical company with a global presence.In addition to Junod, Daewon Pharm has also introduced the Forteo biosimilar ‘Terrosa Inj’ through an agreement with Gedeon Richter. Terrosa was developed by Richter-Helm Biotec, a joint venture between Gedeon Richter and the German company Helm.With Daewon Pharm obtaining approval for Junod, there are now 5 Prolia biosimilars available in the Korean market.Celltrion’s ‘Stoboclo Prefilled Syringe’ was the first biosimilar approved in November 2024, followed by Samsung Bioepis’ ‘Obodence Prefilled Syringe,’ Meditip’s ‘Meditip Denosumab Prefilled Syringe,’ and HK inno.N’s ‘Izambia Prefilled Syringe,’ all of which have obtained marketing approval.Meditip Denosumab Prefilled Syringe was approved in Korea through a regulatory affairs agency, and Novartis has been identified as the contract manufacturer. There is a possibility that Novartis will handle domestic sales in the future.HK inno.N’s Izambia was developed by Spain-based mAbxience. HK inno.N has also signed a contract with mAbxience to introduce a nivolumab (brand name: Opdivo) biosimilar in addition to this denosumab biosimilar.Meanwhile, Celltrion’s Stoboclo is being co-marketed by Daewoong Pharmaceutical, and Samba’s Obodence is being co-marketed by Hanmi Pharmaceutical.The denosumab biosimilar market is rapidly becoming a battleground among major domestic pharmaceutical companies.Currently, Celltrion’s Stoboclo and Samsung Bioepis’ Obodence are listed for reimbursement. Full-scale competition is expected once the remaining approved products are also listed. All patents for Prolia listed with the MFDS expired as of March 17 last year.
- Policy
- Premiums for incrementally modified drugs remain uncertain
- by Jung, Heung-Jun Mar 31, 2026 08:45am
- As the government has finalized the overall framework of the drug pricing system reform, discussions are expected to move into detailed areas, including premiums for incrementally modified drugs.The core of this drug pricing system reform is the innovative level of companies. However, decisions regarding product-level premium measures, such as those for incrementally modified drugs, have been left out.According to industry sources on the 27th, since the premium rate for incrementally modified drugs was not clearly determined at this Health Insurance Policy Deliberation Committee meeting, follow-up discussions are expected to continue.The reform plan discussed at the Health Insurance Policy Deliberation Committee meeting last November had tentatively decided to maintain the current premium system for “incrementally modified drugs, incrementally modified combination drugs, and biosimilars.”However, this wording was deleted at the latest HIPDC meeting. With the addition of a new “quasi-innovative company” category, the drug price premium scheme has been significantly revised. The disclosed premium preferential measures consist solely of a 60% premium for innovative drugs, a 50% premium for quasi-innovative drugs, and preferential treatment for pharmaceutical companies and drugs that ensure supply stability.Under the current pricing calculation system, incrementally modified drugs receive premiums on top of the base price. For salt/formulation changes after original patent expiry, the price is set at 70% with a premium from the base price (53.55%). The price is set at 77% with a premium from the base price (58.9%). The premium is generally applied for 1 year after listing, but can be extended up to 3 years if there are three or fewer generic manufacturers listed for reimbursment.The key question is whether the government will maintain these premium rates. Since the base calculation rate has been lowered, there is a possibility that the premium rates will also be readjusted. If the government attempts to lower the premium rates for incrementally modified drugs in line with the reduced base rate, strong industry backlash is expected. The prevailing view in the industry is that if the government attempts to adjust the premium rate as well, it will dampen the motivation to develop incrementally modified drugs.There is also the possibility of reforming premiums for incrementally modified combination drugs. Currently, their price is calculated as the sum of 53.55% of each component drug’s pre-patent-expiry price. In this case, innovative pharmaceutical companies receive preferential treatment with a 68% sum, while general pharmaceutical companies receive a 59.5% sum.Given that drug price premium tiers are currently divided into innovative, quasi-innovative, and non-innovative categories, changes are needed in the calculation rates for incrementally modified drugs as well.In particular, since the “quasi-innovative” category is a newly established preferential pricing bracket, discussions must also address whether to apply differential premium rates within this category.Industry insiders agreed that, as the basic calculation rates and drug price premiums had not been finalized until now, concrete discussions must begin immediately.A pricing manager at a pharmaceutical company stated, “Until now, the focus was on establishing the overall framework, so detailed discussions were not possible. Now, discussions on incrementally modified drug premium rates must begin.”Another industry official added, “The content announced at this Health Insurance Review and Assessment Service (HIRA) meeting only covers the broad framework, so significant detailed adjustments are needed. The same applies to the premium rate for incrementally modified drugs. While each pharmaceutical company may focus on different aspects of the reform, since the base calculation rate has decreased, it is necessary to ensure that premiums are maintained as much as possible.”
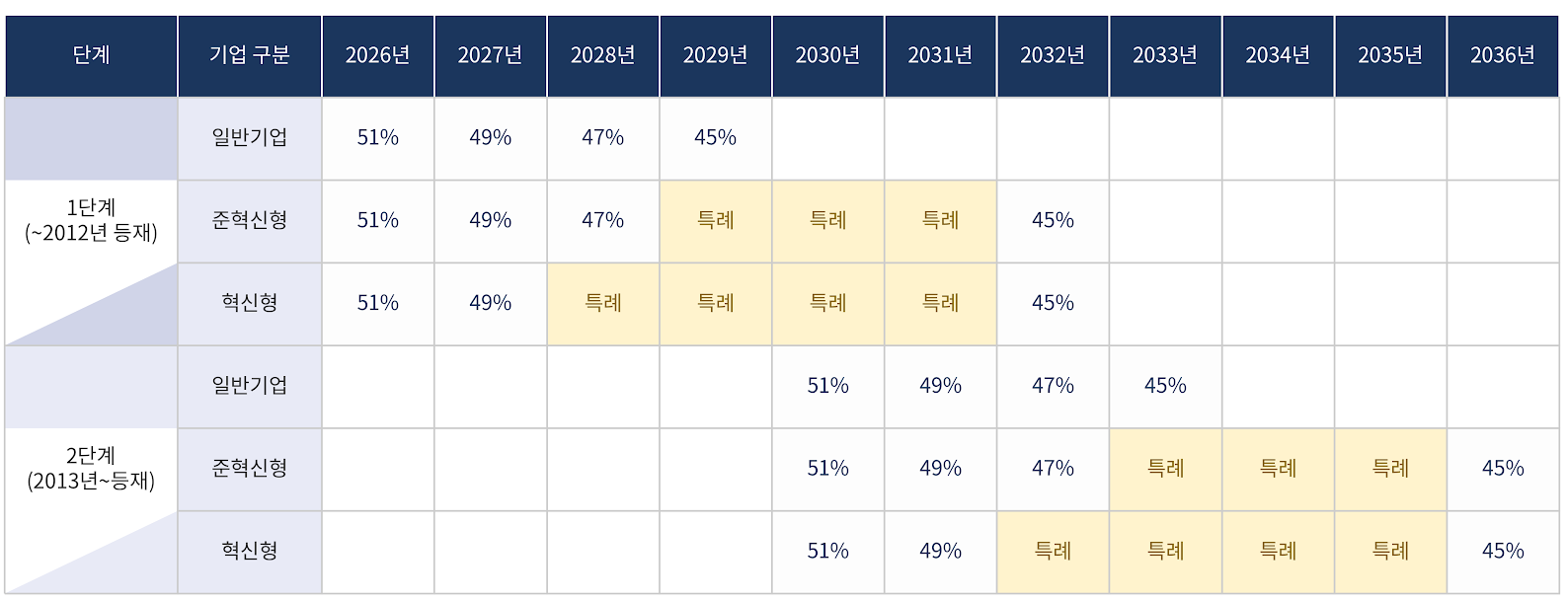
- Policy
- Generic drug price for non-innovative firms set to 45% by 2029
- by Jung, Heung-Jun Mar 30, 2026 09:12am
- The government will implement price cuts for already-listed drugs, including those of non-innovative pharmaceutical companies, over a period of up to 10 years. With a gradual annual reduction of 2%, the drug price of innovative and quasi-innovative companies is expected to reach a rate of 45% by 2032, while non-innovative companies will reach the same level by 2029.Additionally, for products listed after 2013, gradual reductions will begin in 2030. Non-innovative companies will converge to 45% by 2033, while innovative and quasi-innovative companies will do so by 2036.The aim is to mitigate the shock of a sudden drop in sales for the pharmaceutical industry. Under this structure, if pharmaceutical companies fail to generate new revenue, the scale of their sales decline will gradually increase over the 10-year period.Price reductions for already-listed drugs will be implemented over a maximum of 10 years, including for non-innovative pharmaceutical companies.According to the government’s finalized plan announced on the 26th, the timing of reaching the 45% pricing rate will differ depending on ▲the listing date ▲whether special provisions apply.There are also conditional clauses for drugs with the same active ingredient, those failing to meet requirements, combination drugs, and data-submitted products. This means that even if a product was listed in 2013, it may still be included in the first-stage reduction.First, even general pharmaceutical companies that are not classified as innovative will undergo price cuts over 4 years. Prices will fall to 49% next year, then to 47% in 2028, and to 45% in 2029. Compared to innovative firms, they will reach the 45% level three years earlier.Innovative companies will be given a four-year grace period, and quasi-innovative companies a three-year grace period, reaching 45% by 2032. For innovative firms, the pricing rate will remain at 49% until 2027 and then drop directly to 45% in 2032.Drugs listed from 2013 onward will start at 51% in 2030 and decrease by 2% annually. General pharmaceutical companies will reach 45% in 2033, while innovative and non-innovative companies will similarly receive a 3- or 4-year grace period, with the 45% rate applying in 2036.A key feature is the classification of drugs listed by 2012 and those listed from 2013 into two stages (Stage 1 and Stage 2). However, drugs with the same active ingredient are classified into the same group based on the timing of the first generic entry.For example, even if a drug was listed in 2013, if the first generic for that active ingredient was listed in 2012, it falls under the first stage price reduction. ‘In addition, drugs that do not meet requirements, such as those lacking in-house bioequivalence testing or registered active pharmaceutical ingredients, will be subject to the 45% pricing rate. The same applies to combination drugs and data-submitted drugs that are essentially based on off-patent, generic substances.This means that even if a drug is a combination drug or a data-submission drug, if its active ingredients are equivalent to off-patent generics, it is subject to the 45% adjustment without exception.