- LOGIN
- MemberShip
- 2026-05-06 15:22:55
- Policy
- Dong-A ST enters Phase III trial for its new OAB treatment
- by Lee, Tak-Sun Jan 20, 2022 05:55am
- Dong-A STNews of new drugs being developed by local companies are coming in one after another from early on this year. Dong-A ST, which had developed three new drugs including the erectile dysfunction treatment ‘Zydena (udenafil),’ antibiotic ‘Sivestro (tedizolid phosphate),’ and antidiabetic ‘Suganon (evogliptin),’ is also receiving attention with its new drug candidate entering final stages of commercialization. The Ministry of Food and Drug Safety approved the Phase III study protocol for Dong-A ST’s 'DA-8010,’ a new drug candidate in development for the treatment of overactive bladder syndrome. The Phase III trial will be conducted to test the efficacy and safety of DA-8010 in 595 patients with overactive bladder in Korea. The trial will be conducted at the Sinchon Severance Hospital. DA-8010 is a new antimuscarinic being developed by Dong-A ST. Antimuscarinics inhibit involuntary contraction of the bladder and reduce urinary urgency to increase bladder capacity by primarily acting in the urinary storage phase where parasympathetic nerves are not activated. Astellas ‘VESIcare (solifenacin)’ is a representative antimuscarinic drug, However, antimuscarinic drugs as such are associated with adverse events such as dry mouth and constipation. DA-8010 is expected to improve such side effects and have a more superior effect over existing products. Dong-A ST had started the development of the drug in 2010 last year and is now awaiting commercialization. In addition to Korea, the company has also completed a Phase I trial for the drug in Europe. Dong-A ST, which is famous for its energy drink ‘Bacchus,’ has been leading the R&D of new drugs in the domestic industry, succeeding in developing 3 new drugs. In particular, its ‘Zydena (erectile dysfunction)’ that was approved in 2005 and ‘Suganon (diabetes treatment)’ in 2015 both made great success in the market and were noted for their marketability as well. ‘4 new homegrown drugs’ approved last year… Daewoong and SK Bioscience expected to again receive approval for new drugs this year Yuhan Corp’s new NSCLC treatment that was approved in January last yearLast year, Korea celebrated the highest number of approvals of homegrown new drugs. Starting with Yuhan Corp’s anticancer drug ‘Leclaza’ in January, Celltrion’s COVID-19 treatment ‘Regkirona’ in February, Hanmi Pharmaceutical’s neutropenia treatment ‘Rolontis’ in March, and Daewoong Pharmaceutical's ‘Fexclu’ was approved in December last year. The companies are developing their drugs abroad as well to target the global market. In the case of Rolontis, the industry believes the drug may be approved by the US FDA within the year. Daewoong Pharmaceutical, which received approval for Fexclu tab in the end of last year, is also planning to introduce a new drug this year. The company plans to complete Phase III trials for the SGLT-2 inhibitor it is developing for the treatment of type 2 diabetes, 'DWP16001,' as a monotherapy and combination therapy. The COVID-19 vaccine candidate ‘GBP510’ that is being developed by SK Bioscience also completed patient enrollment for the Phase III trial by registering 4,000 patients and started the efficacy analysis. GBP5010 is expected to be commercialized in the first half of this year.
- Policy
- SK Bioscience has completed recruiting phase 3 participants
- by Kim, Jung-Ju Jan 20, 2022 05:55am
- The COVID-19 vaccine GBP510, which is being developed by Korean companies, is undergoing phase 3 global clinical trials. The recruitment of 4,000 people from six countries has been completed. Today (18th), the government held the 26th meeting of the pan-government TF to support clinical trials of COVID-19 vaccines and treatments to discuss the progress of the development of COVID-19 vaccines in Korea. The meeting was attended by Ryu Geun-hyuk, the second vice minister of the MOHW, related ministries, and private experts, and SK Bioscience President Ahn Jae-yong announced the progress of clinical trials for the COVID-19 vaccine currently under development. In Korea, eight companies are currently conducting clinical trials for the COVID-19 vaccine. Participating companies are SK Bioscience, EuBiologics, Geneone, Genexine, Quratis, HK inno.N, Cellid, Aijin. Among them, SK Bioscience's GBP510 is being developed the fastest with its entry into phase 3 clinical trials as of August 10 last year. Phase 3 clinical trials of GBP510 have recruited clinical participants from five foreign countries, including Korea, Thailand, the Philippines, Vietnam, Ukraine, and New Zealand, with a total of 3,990 subjects in five months since it began administration on August 30 last year. SK Bioscience announced that it plans to complete the development of vaccines in the first half of this year through rapid sample analysis and data acquisition for vaccines under development in the future. When the development of GBP510 is completed with CEPI support, it will be supplied to countries around the world through the COVAX Facility. Meanwhile, the government is also conducting a pre-purchase process for 10 million doses of domestic vaccines under development by SK Bioscience. Until now, the government has supported the recruitment of clinical participants, supported rapid clinical proceedings such as clinical permits through overseas missions in clinical countries, and supported rapid sample analysis led by the government for vaccine efficacy analysis through Korea National Institute of Health and the International Vaccine Institute. As of the 17th, a total of 2,163 cases have been received, and 1,764 cases have been analyzed, and the sample analysis is supported to proceed quickly as soon as the sample is received. In addition, a cross- and additional vaccination clinical trial under the supervision of KDCA is underway for the safe vaccination of the people after the development of domestic vaccines. Second Vice Minister Ryu Geun-hyuk said, "Even when it is difficult to recruit clinical participants due to COVID-19 vaccination, we were able to complete the recruitment of clinical trial participants for domestic vaccine development smoothly thanks to the public's high interest and active cooperation. After that, we will focus on support at the pan-government level so that sample analysis, approval, examination, and commercialization can proceed quickly. We will support the development of various domestic vaccines and treatments, including SK Bioscience, until the end."
- Policy
- Additional Paxlovid for 10,000 people will be introduced
- by Kang, Shin-Kook Jan 20, 2022 05:54am
- Prime Minister Kim Bu-gyeom visited the Residential Treatment CenterPrime Minister Kim Bu-gyeom announced that he would introduce an additional 10,000 servings at the end of this month as patients' condition improved after taking the COVID-19 treatment. Prime Minister Kim posted a message on his SNS on the 17th and said, "The prescription and administration of COVID-19 treatment began last week." He said, "I visited the residential treatment center in Jung-gu, Seoul a while ago and looked at the status of medication for eating treatments. "I met the elderly who were taking the medicine on the screen and asked how they were doing over the phone, and I heard that the elderly were taking the medicine well and are currently improving well without any adverse reactions." He said, "In addition to the elderly I called today, it is very fortunate that patients who were prescribed during home treatment are being treated without any major adverse reactions. There are drugs that patient shouldn't take together, so patient needs to be careful with prescriptions, and speed is important because he/she has to take the medication within five days of symptoms." He said, "Looking at the scene today, if the prescribed person is taking a medicine that should not be taken together, the notification function is immediately activated on the system. "We were well prepared for quick and safe administration through the world-class drug safety use service (DUR)." Prime Minister Kim said, "The introduction of treatments is also proceeding without much difficulty." He said, "Last week, Doses for 21,000 people were introduced, and an additional doses for 10,000 people will be arrived at the end of this month. If active prescriptions begin, the severity rate and mortality rate will be lowered through the treatment taken, which will be a good response to Omicron mutations, he predicted. In addition, he said, "The government will thoroughly manage the monitoring of adverse reactions of treatments to be taken and actively respond to side effects."
- Policy
- Bill for Rotarix·RotaTeq to be covered by NIP
- by Lee, Jeong-Hwan Jan 19, 2022 06:06am
- A bill to promote the health of infants and young children while reducing consumer burden by including rotavirus vaccines in the National Immunization Program (NIP) is being promoted. If passed, the bill will turn GSK’s Rotarix and MSD’s RotaTeq that are already being used in Korea into free national vaccinations. Since the WHO recommends mandatory vaccination of the rotavirus vaccine and the nature of the virus that is highly infectious among infants and young children, attention is focused on the legislative direction of the bill. On the 18th, NA member Bae Hyun-jin of the People Power Party announced that she had submitted a bill as representative for the partial amendment of the ‘Infectious Disease Control And Prevention Act’ to incorporate such changes. The incidence of rotavirus infection is very high among children aged under 5 to the extent that most of these infants are infected at least once with the virus. NA member Bae Hyun-jin expressed concerns that some parents give up rotavirus vaccinations due to the high cost even though rotavirus vaccinations are recommended for all newborns after 6 weeks of age. According to the KDCA’s Immunization Registry System, 7.9%, 21,728 of the 274,221 newborns eligible for vaccinations did not receive rotavirus vaccines in 2020. Statistics over the past 5 years show that 14.8% of the infants and young children eligible for vaccinations – 222,565 – did not receive rotavirus vaccines. Rotavirus vaccines are relatively high-priced, costing around ₩200,000-₩300,000. The two rotavirus vaccines currently sold in the market are GSK’s Rotarix and MSD’s RotaTeq. Rotarix is given in 2 doses, RotaTeq in 3. The two vaccines cost ₩70,000-₩100,000 and ₩100,000-₩130,000 per shot, respectively. In total, Rotarix costs around ₩200,000-₩260,000 and RotaTeq ₩210,000-₩300,000 to get fully vaccinated. The issue regarding the price of rotavirus vaccines has risen several times intermittently. GSK and MSD had raised the price of Rotarix and RotaTeq by 12% and 17% respectively, increasing the burden borne by consumers. Rotavirus vaccines are considered essential for newborns, and therefore its consumers are highly sensitive to their change in price. As a result, some local governments have implemented policies to provide free rotavirus vaccines for infants. Bae believes that the government should pay for the cost of the rotavirus vaccine inoculations to prevent enteritis in infants and young children, contribute to promoting public health, and reduce the burden borne by the consumers. Therefore, the bill contains a plan to include rotavirus infection as a disease eligible for regular vaccinations. Bae said, “The reality is that the need for rotavirus vaccination has been discussed for a long time but was excluded from support due to its high cost. However, no infants nor young children should be left out, unable to receive essential vaccinations due to financial reasons.”
- Policy
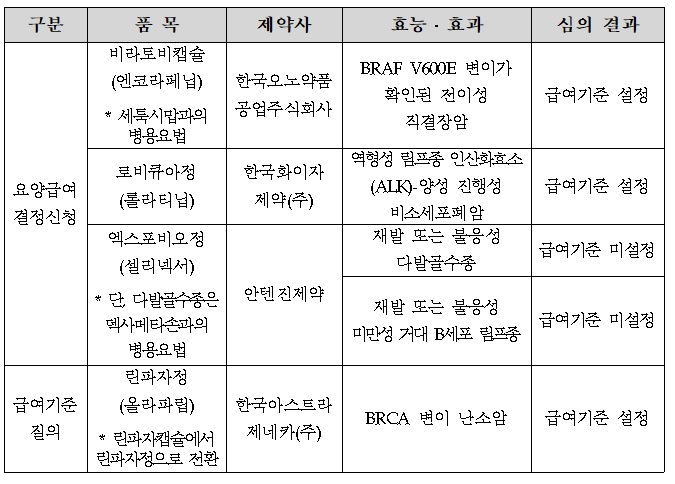
- The benefit standard for Braftovi & Lorviqua was set
- by Lee, Hye-Kyung Jan 19, 2022 06:06am
- At the first Severe (Cancer) Disease Review Committee held this year, the benefit standards for Braftovi, a direct bowel cancer treatment of Ono Pharmaceutical Korea, and Pfizer's Lorviqua, were set. The HIRA (Director Kim Sun-min) today (12th) released the "Result of Review on benefit Standards for Drugs for Cancer Patients" reviewed by the 1st Cancer Disease Review Committee in 2022. Today's cancer screening was newly participated by members of the 9th committee formed in November last year, and Braftovi and Lorviqua, who applied for medical care benefit decisions, set benefit standards, and Antigene's Xpovio did not set standards. Braftovi, which standard was set, was approved by the MFDS on August 19 last year for the use of combination therapy with Erbitux in adult patients with direct bowel cancer who had previous treatment experience and confirmed BRAF V600E mutation. Since Lorviqua was designated as a rare drug in March 2020, it is approved in Korea in July 2021 for use in cases treated with Xalkori and at least one other ALK inhibitor, or with a primary ALK inhibitor as a monotherapy for the treatment of adult patients with ALK-positive progressive non-small cell lung cancer. Xpovio has not established a benefit standard for both permits, such as recurrent or refractory multiple myeloma and recurrent or refractory giant B cell lymphoma. The standard was set for Lynparza tab's standard. Meanwhile, in accordance with Articles 5 and 5-2 of the National Health Insurance Medical Care Benefit Standards, the HIRA may publicly announce drugs prescribed and administered to severely ill patients after deliberation by the Severe Disease Review Committee. The benefit criteria for the drug may be set differently according to clinical literature, domestic and foreign guidelines, and expert opinions within the scope of the efficacy and effectiveness of the MFDS' permission, and the benefit criteria may change during the follow-up procedure.
- Policy
- Daewoong tx are being developed for skin side effects
- by Lee, Tak-Sun Jan 18, 2022 06:06am
- Daewoong PharmaceuticalDaewoong Pharmaceutical has started to develop a treatment for skin side effects caused by anticancer drugs. This treatment is known as a stem cell-based drug. The MFDS approved the phase 2 clinical trial plan for DWP708 submitted by Daewoong Pharmaceutical on the 17th. This clinical trial is conducted to explore the efficacy of DwP1708 and evaluate safety for the treatment of EGFR inhibitor-related skin side effects. The test methods are multicenter, randomized, double-blinded, placebo-controlled, therapeutic search phase 2. It will be held at Gyeongsang National University Hospital and Dong-A University Hospital for 96 patients in Korea. EGFR inhibitor is mainly used in the treatment of arsenic lung cancer. It has a mechanism to reduce cancer cell proliferation by inhibiting EGFR, and types of anticancer drugs include treatments containing Erlotinib, Cetuximab, and Gefitinib. These anticancer drugs, however, are known to have frequent serious skin side effects. 50-90% of patients administered EGFR anticancer drugs suffer from skin side effects. Mainly, skin rashes, follicles, and dry skin occur in the face and upper body. Treatment of skin side effects is said to be managed with steroids or antibiotics, but the effect is not significant. Accordingly, Daewoong Pharmaceutical has been conducting research on related drugs, believing that EGFR inhibitory anticancer drugs block normal cell proliferation such as keratinocytes, causing skin side effects. According to the list of Daewoong's R&D tasks, DWP708 is indicated as a stem cell treatment. The study started in 2007 and received approval for phase 2 clinical trials this time and will begin full-scale human application research. The new drug candidate is known to be the first anticancer drug to be applied as a skin side effect treatment. Daewoong Pharmaceutical expects that it will contribute to controlling the dose of anticancer drugs and reducing the discontinuation rate due to skin side effects as a mechanism to suppress skin epithelial regeneration and inflammation. It is not possible to determine the exact market size as there are no anticancer drug skin side effects treatments, but if the drug is commercialized, it is expected to significantly improve the quality of life of cancer patients administering EGFR treatments.
- Policy
- Discord in system hinder coverage of rare disease drugs
- by Lee, Jeong-Hwan Jan 18, 2022 06:06am
- A suggestion has been raised that it is irrational for a drug that received an orphan drug designation to be ineligible for insurance benefits that were set for rare disease treatments to improve patient accessibility because its disease was not designated as a rare disease or set for special exemption of calculation. In other words, the treatment and disease burden born by the patients are worsening because the tools for orphan drug reimbursement evaluations such as the risk-sharing agreement system (RSA) and the pharmacoeconomic evaluation exemption system cannot be applied due to discord between the designation of orphan drugs and rare diseases, and this issue must be resolved. In particular, the benefits of reimbursement tools such as the RSA are too concentrated on anticancer drugs, leaving orphan drugs to be neglected from the system. Criticism also followed that the national orphan drug policy is more focused on providing convenience to the administrative system rather than being patient-focused in its operations. On the 17th, Jong Hyuk Lee, Professor of Chung-ang University’s College of Pharmacy, wrote so in a special column of ‘Angel Spoon,’ a magazine published by the Korean Organization for Rare Disease. Professor Lee argued that Korea should spend more on rare drugs as the current rate falls much below the global rate. As of 2018, Korea spent 370 billion won on rare disease drugs, which is 2.1% of its total drug expenditure. The global rate of expenditures on rare disease drugs exceeds 14%. Lee added that there is a need to improve the reality that stops patients from benefitting from systems that can reduce their medical costs, such as the RSA and the pharmacoeconomic evaluation exemption system that are in place in Korea. Patients cannot receive these benefits if their condition is not designated as a rare disease. Even if a drug is designated as an orphan drug used to treat rare diseases, the drug may not go through the RSA or pharmacoeconomic evaluation exemption track when undergoing evaluation for reimbursement benefits if its indicated disease is not recognized as a rare disease. This in turn triggers failure in reimbursement and increases the burden borne by patients. Also, the operation of the NHI coverage enhancement policy is too focused on anticancer drugs, harming the reimbursement of rare disease drugs in the process. 78%, or 32 of the 41 drugs that are applied RSA, a system that plays the biggest role in enhancing coverage of new drugs, were anticancer drugs. Another issue that was raised was that the system for rare disease drugs was designed to convenience government ministries rather than the patients. Orphan drug designation/approval is carried out by the Ministry of Food and Drug Safety; rare disease designation/ insurance registration/ special exemption of calculation by the Ministry of Health and Welfare and operated by the Korea Disease Control and Prevention Agency, the Health Insurance Review and Assessment Service, and the National Health Insurance Service; medical expenses support by each city/town/district; and the catastrophic medical expense support project operated by NHIS, rendering the processes too complicated for patients to follow. In the column, Lee emphasized that we need to expedite the approval of rare disease treatments to increase accessibility for the patients and increase special systems like the pharmacoeconomic evaluation exemption system for such diseases in the course of granting their reimbursement. Also, Lee added that social discussion should be held on diversifying finances for the coverage of rare diseases that are mainly sourced by NHI finances by raising funds for rare diseases, etc. Lee said, “We need to carefully examine whether any patients are left neglected due to institutional issues that create gaps between the designation of rare diseases and orphan drugs. The entities that operate such systems for rare diseases vary and the procedures are also very complicated. Therefore, the system should move away from such convenience-focused administrative practices and become more patient-centered.”
- Policy
- The gov will push for a refund on losses from suspension
- by Kim, Jung-Ju Jan 18, 2022 06:05am
- The government will push for a revision of the law that will put a brake on the suspension of execution filed by pharmaceutical companies in drug price lawsuits. If companies fight against the government over drug prices and finally win the case, the insurer will refund the drug cost equivalent to the loss so far. This is a revision of the legal unit, so it is highly likely to take effect immediately. The revision bill will three times shorten the negotiation period for the reduction of the original drug price of patent expiration, and includes amendments to omit drugs with a history of negotiations in the future. The MOHW announced today (17th) the "Partial Amendment to the Rules on the standards of national health insurance medical benefits" and began inquiring opinions. The revision is a plan already reported by the government to the Health Insurance Policy Deliberation Committee late last month, and is a follow-up to calls by the National Assembly to prevent abuse of execution and rationalize industry negotiations. ◆Introduction of the loss refund system according to the outcome of the dispute =The government explained that the purpose of the system is to protect the rights and interests of drug manufacturers and prevent financial losses in health insurance by applying for suspension of execution when pharmaceutical companies file drug lawsuits. The main contents of the amendment stipulate that the chairman of the National Health Insurance Service pays the manufacturer, etc. the loss if the drug manufacturer or the administrative trial's citation ruling is confirmed on whether the Minister of Health and Welfare is eligible, adjustment of upper limit price, and changes in details. On the contrary, if the government wins, a revision is currently pending in the National Assembly to recover the insurer's loss to the pharmaceutical company that filed the lawsuit. ◆Regulation of negotiation period and procedure for registered drugs and follow-up measures = The maintenance and measures were designed to prevent confusion in the pharmaceutical industry and secure predictability caused by unclear negotiation procedures and follow-up measures since October 2020. The main contents include allowing negotiations to be omitted for drugs with a history of existing negotiations and reasonably reducing the negotiation period from the current 60 days to the next 20 days in consideration of health insurance finances when the government adjusts the original drug ex officio. However, if the negotiations break down, renegotiation will be allowed after deliberation by the Drug Benefit Evaluation Committee, and if the negotiations break down, the drug will be excluded from medical care benefits. The MOHW plans to conduct an opinion inquiry until March 18 and implement it as originally planned if there is nothing special.
- Policy
- A plan to expand patient access to Kymriah should be passed
- by Kim, Jung-Ju Jan 17, 2022 05:51am
- While the agenda for the health insurance registration of the ultra-high-priced innovative drug Kymriah and the expansion of immuno-cancer drug Keytruda benefits have been in full swing since the afternoon of today (13th), patient groups gathered in front of the Kukje Electronic Center's Smart Work Center. Patient maximum co-payment of 1 million won Of the patients' Union including patient maximum co-payment of 1 million won told the Drug Reimbursement Evaluation Committee, that the committee should pass the Kymriah insurance registration agenda and the expansion of Keytruda benefit standards, and presidential candidates insisted on the introduction of a "fast new drug health insurance system" and "Patient maximum co-payment of 1 million won." Acute lymphoblastic leukemia, lymphoma - car kymriah t treatment is Novartis Korea by taking advantage of the 'drug approval - patent linkage system' for listing on March 3, health insurance last year and on October 13, nearly seven months after conditionally cancer drugs benefit appraisal committee through. Kymriah, an acute lymphocytic leukemia and lymphoma CAR-T treatment, applied for health insurance registration on March 3 last year using the "Drug Approval-Patent Linkage System" and passed the Cancer Drugs Benefit Appeal Committee on October 13, about seven months later. No longer there is no cure, recurrence or refractory acute lymphocytic leukemia and lymphoma patients about 200 new cases annually. Kymriah's treatment effect was a one-time treatment, with 8 out of 10 patients with end-stage acute lymphocytic leukemia and 4 out of 10 patients surviving for a long time, which is a "one-shot treatment" with only one dose, but the disadvantage was about 460 million won. In addition, Keytruda, a non-small cell lung cancer immuno-cancer drug among lung cancer, has to be used from the first treatment to obtain better treatment effects, and thousands of patients are required to pay from 70 million won to 100 million won per year. Therefore, Kymriah has been delayed in health insurance registration for 11 months, and Keytruda has not been able to expand its health insurance standards to primary treatments for 4 years and 4 months. Patient groups appealed, "Thousands or tens of thousands of terminal acute lymphocytic leukemia and lymphoma patients and stage 4 non-small cell lung cancer patients, who had been eagerly waiting for the coverage of Kymriah and Keytruda health insurance, eventually died because they could not afford the drug price." These groups also argued in connection with the recent pledge of the ruling party's presidential candidate to apply health insurance to hair loss drugs. Patient groups said, "Whether hair loss drugs are covered is a matter of comprehensive judgment considering the priority of financial use, Korea's health insurance situation, patient burden of drug prices, and cost-to-treatment effects, but if Presidential candidate Lee Jae-myung actively reviews life-related health insurance coverage, he should soon." In particular, these groups pressured the National Human Rights Commission of Korea's review of the petition filed against the Minister of Health and Welfare in October last year by some patient guardians preparing for leukemia patients and Kymriah treatment. Finally, they urged the government ▲to push for patient maximum co-payment of 1 million won system to ensure rapid access to Kymriah and Keytruda to about 200 terminal acute lymphocytic leukemia and lymphoma patients and thousands of stage 4 non-small cell lung cancer patients, ▲ to be held today, drug reimbursement evaluation committee the agenda through, and ▲the government should introduce a quick registration system for new drug health insurance directly related to life.
- Policy
- The ultra-high-priced new drug Kymriah passed the Committee
- by Lee, Hye-Kyung Jan 14, 2022 05:50am
- Kymriah(Tisagenlecleucel) of Novartis Korea, dubbed an ultra-high-priced "one-shot treatment" with a cost of 500 million won per dose, passed the committee. The HIRA (Director Kim Sun-min) held the first Drug Reimbursement Evaluation Committee in 2021 on the afternoon of the 13th and conducted a review on the appropriateness of medical care benefits for five items such as Nasa Care Realtris Nasal Spray 18mL or 31mL. At that time, the deliberation committee was slightly delayed until the agenda was presented. ▲ Higher levels of risk sharing by pharmaceutical companies considering the level of overseas drug prices, ▲ In the case of diffuse giant B-cell lymphoma, which has insufficient clinical performance compared to acute lymphoid leukemia, the risk-sharing system of the performance-based payment model according to treatment performance is added on a patient-by-patient basis, and ▲ Kymriah While attaching clues such as setting the total amount of expenditure. While the committee decided on the adequacy of Kymriah benefits, it attached conditions for applying DLBCL and Expenditure Cap. Until now, the NHIS has been holding continuous meetings with the MOHW regarding financial sharing ahead of the Kymriah price negotiations, and will use RSA/Expension Cap for 60 days to come up with financial sharing plans between the government and pharmaceutical companies. In addition, five items, including lesinowon, which were reviewed, were also recognized for their appropriateness of benefits, but NASA Care Realtris Nasal Spray can take the stage of listing salaries if they are accepted below the committee's evaluation amount.