- LOGIN
- MemberShip
- 2026-05-02 20:13:48
- Policy
- Countdown to initial non-face-to-face treatment
- by Lee, Jeong-Hwan Apr 05, 2023 05:48am
- Member of People’s Power Kim Seong-won (Photo: Congressman Kim Seong-won’s office blog)The Power of the People Together with Rep. Kim Seong-won, Democratic Party Rep. Kang Hoon-sik co-represented the National Assembly's startup research group'Unicorn Farm', which plans to propose a bill to amend the Medical Act to allow non-face-to-face treatment from the first visit on the 3rd. The representative initiative of the bill is Kim Seong-won (Dongducheon-si, Gyeonggi-do, Yeoncheon), and Rep. Kang Hoon-shik is listed on the list of co-sponsors. Rep. Kim Seong-won is active in the Industry, Trade, and Energy Small and Medium Venture Business Committee of the National Assembly. Rep. Kang Hoon-shik is the secretary of the Democratic Party of the Health and Welfare Committee. The core of the bill is to allow non-face-to-face treatment for patients specified by the Ordinance of the Ministry of Health and Welfare, regardless of whether they are first or re-examinations if it is possible to improve medical accessibility without harming the patient's health. The bill stipulates the scope of non-face-to-face treatment as continuous observation of health and disease, diagnosis, consultation, hospital visit guidance, and prescription. On this day, an official from Unicorn Farm explained in a phone call with Daily Farm, “There have been complaints from non-face-to-face treatment platform companies such as the Telemedicine Industry Council (hereinafter referred to as Wonsanhyup), and we will propose a bill for non-face-to-face treatment for first-time patients soon.” The official explains that the first-time non-face-to-face treatment bill is being legislated at the level of Unicorn Farm, a startup research group. Rep. Kim Seong-won, a member of the ruling party and a co-representative of Unicorn Farm, said that he agreed with the complaints regarding the non-face-to-face treatment regulation of Wonsanhyup, a member of which includes Dr. Rep. Kang Hoon-sik, a member of the opposition party, is also known to be in the position that, as a co-representative of Unicorn Farm, it is necessary to discuss and review the bill for first-time non-face-to-face treatment in the National Assembly. However, Rep. Kang Hoon-sik, secretary of the opposition party of the Welfare Committee, is fully aware of the fact that first-time non-face-to-face treatment is strongly opposed by the healthcare community, so he plans to put it on the discussion table to collect and deliberate various opinions from doctors, pharmacists, platform companies, and patients. Previously, Congressman Kang proposed a unicorn farm-level bill as a representative on the 13th of last month. The medical advertisement platform proposal, such as Gangnam Unni, was the starting point, and the amendment to the Medical Act that allows the Ministry of Health and Welfare to intervene in the review standards of the Medical Advertisement Deliberation Committee, which is composed of functional groups such as doctors, oriental doctors, and dentists, is that. This time, following Rep. Kang, Rep. Kim Seong-won accepted the proposal of a non-face-to-face treatment platform such as Wonsanhyup and introduced a bill to allow first-time non-face-to-face treatment. The first non-face-to-face treatment bill, which Congressman Kim is about to propose, allows non-face-to-face treatment from the first visit, except in cases where there are special restrictions. It is a legislative measure to address concerns that platform companies will go bankrupt if non-face-to-face treatment is allowed only for returning patients, and the scope of non-face-to-face treatment is specified in a negative regulatory manner rather than a positive regulation. When Kim's bill is proposed, for the first time, the National Assembly will review a first-time non-face-to-face treatment bill. All four non-face-to-face treatment bills proposed so far are bills centered on returning patients that do not allow first visits. Democratic Party lawmakers Kang Byung-won, Choi Hye-young, and Shin Hyun-young and People’s Power Congressman Lee Jong-seong have each proposed a representative initiative. An official from Unicorn Farm said, "It is a bill that allows non-face-to-face treatment for patients specified by the Ordinance of the Ministry of Health and Welfare if it can increase medical access without harming the patient's health." The scope of non-face-to-face treatment includes continuous observation of disease, diagnosis, consultation, hospital visit guidance, and prescription.” Meanwhile, the research purpose of Unicorn Farm, a research group of the National Assembly, is regulation rationalization, policy development, and related legislative measures to support the growth of startups and venture companies.
- Policy
- The severity level of COVID-19 lowered from May
- by Lee, Jeong-Hwan Mar 31, 2023 06:09am
- 한덕수 총리 The government announced plans to lower the severity level of COVID-19 to the alert from May and switch to a medium-level capital system. When the serious stage is lifted, the current 'temporary non-face-to-face treatment' by the current Infectious Disease Control Act is expected to end automatically. Today (29th), Prime Minister Han Deok-soo held a 'COVID-19 meeting' at the Seoul Government Complex and made this announcement. Prime Minister Han Deok-soo said, "Recently, the decline in the global COVID-19 epidemic is clear. The domestic quarantine situation is also being stably managed." We will finalize the action," he said. Prime Minister Han said, "We plan to change the obligation to quarantine and the obligation to wear masks, which remain, to a 'full recommendation'." We will maintain the protection system for the vulnerable,” he added. "The long fight against COVID-19, which has been going on for more than three years, is coming to an end," he said. "The government will do its best to protect the lives and safety of the people until COVID-19 is completely over."
- Policy
- Cold drugs subject to PVA will be adjusted
- by Lee, Tak-Sun Mar 31, 2023 06:08am
- The government is paying close attention to the correction plan for the price volume agreement (PVA) system that will be applied to respiratory drugs such as cold medicines whose usage has increased due to COVID-19. Therefore, the schedule the authorities had set to start PVA monitoring in April after finalizing the correction plan within the month is expected to be somewhat delayed. The health insurance authorities plan to make a final decision after calculating the number of products subject to PVA through simulation when it applies the correction plan. According to industry sources on the 30th, the National Health Insurance Service collected the opinion of the pharmaceutical industry and tentatively confirmed a correction plan. In the plan, the NHIS has been known to have widely reflected the opinions it had collected from the pharmaceutical industry. The correction plan will compare each drug’s use volume with the previous year after excluding their usage in specific months when the number of COVID-19 cases increased explosively. About 2,600 items are subject to corrections, including cold medicine and antibiotics that the Ministry of Food and Drug Safety encouraged the production of during the COVID-19 crisis. On the 7th, Sang-il Lee, Executive Director for Benefit at NHIS explained that the NHIS will complete discussions with the industry within the month, finalize the corrective measure, and then start monitoring drugs for PVA negotiations from April. However, it seems that it will take more than this month for the government to decide upon the final measure because of the government's cautious stance. The NHIS plans to apply its tentative correction plan to subject items and simulate how many products will be subject to mock negotiations, and then decide upon on a final draft with the Ministry of Health and Welfare mid-April. The government is focusing on minimizing the number of negotiation subjects in consideration of the opposition that will be raised by the pharmaceutical industry. An industry official said, “Since there is still time left until the government monitors PVA ‘Type C’ drugs, the government is cautiously preparing the correction plan. The government seems to be carefully considering the measure, taking into account the opposition that may arise in the pharmaceutical industry.”
- Policy
- Overseas clinical trial drugs for therapeutic purposes
- by Lee, Hye-Kyung Mar 31, 2023 06:08am
- An amendment to the Pharmaceutical Affairs Act passed the plenary session of the National Assembly on the 30th, including permitting the use of investigational drugs overseas for therapeutic purposes, establishing a basis for prompt action against online illegal activities of drugs, and strengthening education and publicity on the labeling of drugs for consumers. This amendment will be transferred from the National Assembly to the government and promulgated and will come into effect six months after the date of promulgation. The Ministry of Food and Drug Safety will expand the scope of investigational drugs that can be approved for use for treatment purposes not only domestically under clinical trials but also overseas clinical trials in the future so that patients with life-threatening serious diseases or alternative treatment is said to expand treatment opportunities for emergency patients without means. Allowing the use of overseas clinical trial drugs for therapeutic purposes is task number 22 of the 100 tasks for food and drug regulatory innovation announced by the Ministry of Food and Drug Safety in August of last year to support industrial development while putting public health and safety first. Although the legal grounds for immediate action against illegal acts such as online drug sales and advertisements were insufficient, the Ministry of Food and Drug Safety will in the future strengthen consumer safety by requiring information and communication service providers to take measures to notify consumers that they are advertisements for illegal sales. The Ministry of Food and Drug Safety can provide education and publicity on the sales, purchase, labeling, and advertising of pharmaceuticals to help consumers use medicines safely, conduct research and development to investigate the current status of illegal online drug sales, and prepare effective monitoring technologies and methods. be able to support "We will continue to do our best to improve related systems to protect the public by realizing the safety of warm food and medicine with social values, putting public safety first," said Minister Oh Yoo-gyeong of the Ministry of Food and Drug Safety. did. Major amendments to the Pharmaceutical Affairs Act can be found at http://mfds.go.kr or the National Assembly Security Information System.
- Policy
- Started a demand survey for drugs participating in the pilot
- by Lee, Tak-Sun Mar 31, 2023 06:08am
- Due to the burden of simultaneous data submission, there are likely to be fewer participating drugs. The government started a demand survey for drugs to participate in the parallel trial project of 'Permission Evaluation Negotiation', which is being pursued rapid reimbursement. The project was planned to drastically shorten the registration period by simultaneously proceeding with benefit evaluation and negotiations from the MFDS approval application stage. According to the industry on the 30th, the Ministry of Health and Welfare is conducting a drug demand survey to participate in a parallel pilot project in 2023, including application for permission, evaluation of benefits, and negotiation of drug prices, as part of a plan to improve access to treatment for expensive severe diseases and strengthen reimbursement management. Candidate drugs are scheduled to apply for approval from the Ministry of Food and Drug Safety in 2023 and are drugs aimed at treating diseases that threaten survival (life expectancy is less than one year) or rare diseases. If there is no existing treatment or if there is a clinically significant improvement in efficacy, etc. compared to existing treatment, participation is possible. The data to be submitted are related to approval: ▲the expected date of application for domestic approval, ▲effectiveness and effect among items of application for domestic approval (scheduled) , ▲countries approved in foreign countries and items permitted, ▲consistency between items approved in foreign countries and items scheduled for application for domestic approval. Regarding disease information of approved indications, ▲ disease severity, life expectancy, survival rate, progress, etc. ▲ existing treatment methods, and treatment performance data for the disease. Regarding clinical usefulness, ▲a clinical trial summary of the therapeutic confirmatory trial (or related evidence source), ▲the expected number of patients, ▲A8 listing status and listing price, ▲benefits evaluation result in excluding countries, ▲cost when applying for decision Effectiveness application track expected data (PE, medication cost comparison, etc.) can be submitted. The data will be used as basic data for selecting drugs for pilot projects. Applications must be submitted to KPBMA by April 4th. The government plans to promote the pilot project in earnest from the second half of the year. On the 28th, Yoo Mi-young, head of the Pharmaceutical Management Office of the HIRA, said at a meeting with the Korea Special Press Association, “We are currently discussing the target selection and related procedures with the relevant departments of the Ministry of Food and Drug Safety to promote the pilot project.” Explained. It is expected that there will be not many drugs to participate in the pilot project. Since the evaluation methods and conditions of the three agencies - MFDS, HIRA, and NHIS - are different, related pharmaceutical companies are also burdened with preparing data at the same time. It is interpreted that the first step was to investigate demand before starting the pilot project in earnest in the second half of the year.
- Policy
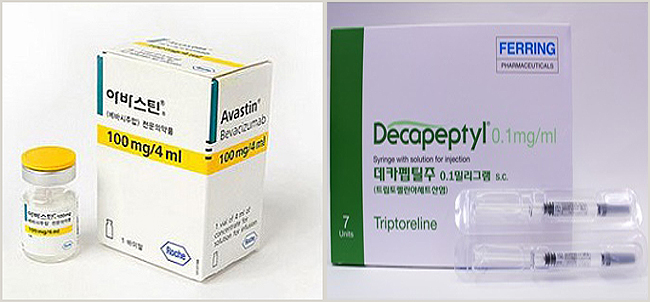
- About 180 clinics without IRB used diagnostic drugs
- by Park,Yang-myeong Mar 30, 2023 05:42am
- Avastin and Decapeptyl, are drugs that allow the use of drugs beyond the scope of approval without the IRB. How much is it used in front-line medical institutions? About 230 macular degeneration treatment drugs Avastin and precocious puberty drug Decapeptyl injection were found to be using these drugs without IRB at 180 hospitals and clinics. The Pharmaceutical Management Office of the Health Insurance Review and Assessment Service disclosed the current status at a meeting with the Special Press Association held on the 28th. In 2019, the relevant notice was changed so that drugs exceeding the permitted scope could be used without reimbursement even without an IRB, but it was only last year that the system was actively applied to the field. The first drug approved by an institution not implemented by the IRB is Avastin, which is used for ophthalmic diseases. Avastin, as a targeted anticancer drug, has been used for various cancers, including colorectal cancer, breast cancer, non-small cell lung cancer, renal cell cancer, ovarian cancer, peritoneal cancer, and cervical cancer. In February 2020, the Korean Academy of Ophthalmology applied for the use of Avastin over the permitted scope to the ophthalmologist, and the HIRA prepared guidelines after discussing infection control measures. The guidelines include measures to prevent infection during the dispensing process. The HIRA received applications for use from non-IRB organizations since February of last year, and until last year, about 230 places used Avastin for ophthalmologic diseases. The second approved drug is Decapeptyl 0.1mg, which is used to diagnose precocious puberty. Decapeptyl is a prescription drug approved for hormone-dependent prostate cancer, endometriosis, uterine fibroids, and central premature onset of puberty in girls under 9 years old and boys under 10 years old. It was not approved as a diagnostic reagent for precocious puberty, but as the supply of the only diagnostic reagent was stopped, medical institutions are continuing to apply for approval for excessive use. It was applied by the Korean Hospital Association in October 2021, and it was confirmed that about 180 hospitals and clinics without an IRB applied for use between July last year and just five months. On the other hand, for the use of drugs over the scope of permission by institutions that have not implemented the IRB, the Korean Medical Association, the Korean Hospital Association, the Korean Dental Association, and related academic societies for each specialty apply for use over permission, and the HIRA reviews for approval. ▲ More than one-third of approved drugs compared to all drug clinical trial institutions for the same case, ▲approval of more than 3,000 drugs for the same case based on the previous year, ▲ Need for expansion Approval can be made if it meets at least one standard, such as medicines for rare diseases and pediatric diseases that are recognized.
- Policy
- New Pompe disease drug Nexviazyme is approved in Korea
- by Lee, Hye-Kyung Mar 30, 2023 05:41am
- The long-term enzyme replacement therapy for Pompe disease, Nexviazyme Inj (avalglucosidase alfa-ngpt), has received marketing authorization in Korea. On the 29th, the Ministry of Food and Drug Safety (Minister Yu-Kyung Oh) announced that it had approved Sanofi-Aventis Korea’s Nexviazyme, a treatment for a rare condition called Pompe disease. Nexviazyme is a recombinant enzyme used as a long-term enzyme replacement therapy in patients confirmed with Pompe disease, which is caused by a genetic deficiency or dysfunction of the lysosomal enzyme acid alpha-glucosidase (GAA). Pompe disease is a rare genetic disorder that causes progressive weakness in the heart and skeletal muscle, leading to respiratory failure and cardiomyopathy. It is a rare condition reported in 1 in 40,000 around the globe and affects about 1,300 people in Korea. The disease presents in two main forms: infantile-onset Pompe disease (IOPD) which presents during infancy, and late-onset Pompe disease (LOPD) which is characterized by onset at all ages and progressively weakens the patients’ muscles. The muscle damage also leads to heart failure, respiratory failure, movement disorders, and sleep disorders. In particular, as Pompe disease is caused by a disorder in the GAA gene, a drug that targets the mannose-6-phosphate (M6P) receptor that allows the transportation of GAA enzymes into the lysosome is administered for its treatment. The drug is a biobetter that has improved dosage and administration over the company’s previous Pompe disease treatment, ‘Myozyme (alglucosidase alfa).’ The company had changed the sugar structure of Myozyme to improve cell absorption of Nexviazyme's active pharmaceutical ingredient. Patients who did not see an effect with Myozyme may use Nexviazyme Inj and see an effect. The MFDS said, “We will continue to make efforts to allow the prompt provision of treatments with confirmed safety and efficacy based on regulatory science."
- Policy
- Obizur will be evaluated for reimbursement upon approval
- by Lee, Tak-Sun Mar 30, 2023 05:41am
- Obizur, a treatment for acquired hemophilia A for adults, approved on the 20th, immediately began a benefit evaluation. This drug is attracting attention as it is the first treatment to replace blood coagulation factor 8 with an indication for acquired hemophilia A. According to the industry on the 29th, the HIRA received an application for Obizur benefits and entered the evaluation. Obizur received approval from the MFDS on the 20th. This drug was designated as an orphan drug in July 2021. US FDA approval was obtained in 2014. Acquired hemophilia A is a rare disease in which autoimmune antibodies cause antibodies to coagulation factor VIII, and bleeding cannot be stopped like in hemophilia patients. The prevalence rate is 0.2 to 1.48 per year per 1 million people, and it is known that there are about 60 patients in Korea. Takeda explains that Obizur is a genetically engineered product made by removing the B-domain from pig coagulation VIII, which is similar to human, and replaces inactivated human VIII, which is not easily recognized by autoimmune antibodies, to help blood coagulation and help control bleeding. did. In phase 2/3 for 28 patients with acquired hemophilia A, Obizur stopped or reduced bleeding in all early bleeding episodes, showed clinical improvement, or showed VIII activity above the target 24 hours after the first administration. The treatment success rate at the time of final administration was 85.7%, and the success rate was higher in the patient group using Obizur as the first treatment at 94%. The patient group using the second-line medicine showed a 73% treatment success rate. It seems that Obizur applied for reimbursement to the HIRA immediately after passing the safety and efficacy verification of the Ministry of Food and Drug Safety.
- Policy
- Govt seeks to improve the drug price reduction system
- by Lee, Tak-Sun Mar 29, 2023 06:02am
- The Korean government will be working together with the pharmaceutical industry to come up with plans to improve the price reduction system that discounts the actual transaction price of drugs. The Health Insurance Review and Assessment Service plans to hold a roundtable meeting with the pharmaceutical industry to improve the actual drug transaction price reduction system based on the recently disclosed research service results. However, whether the improvements will be received with content by both the authorities and the industry remains in question as some of the research service results may be difficult for the pharmaceutical industry to accept. According to industry sources on the 21st, the results of the 'Study to Prepare a Comprehensive Improvement Plan through an Effect Evaluation of the Transaction Drug Price Reduction System' that was led by principal investigator Jin-Hyun Kim, a Seoul National University professor, was disclosed through the public institution management disclosure system on the 9th. The research team proposed short-term, mid-term, and long-term measures for improvement in its final report. As for the short-term measures, ▲abolishing the 10% upper limit in the price ceiling system and ▲reflecting the low purchasing price paid by national and public hospitals in the drug price cuts were proposed. As mid-term measures, ▲ identifying the actual transaction price including rebates ▲ tougher punishments for false reporting, ▲ real financial savings, and ▲ promoting low-price purchases, etc. were proposed. As long-term measures, ▲ switching to a list price reimbursement system rather than the currently ineffective actual transaction price reimbursement system, ▲reducing the insurance price ceiling based on the actual transaction price, ▲changing the drug price structure including the dispensing fee, and ▲ maximizing pharmaceutical expense saving effect on outpatient pharmaceutical expenditures were proposed. As the proposed mid-to long-term measures are difficult to apply immediately, the short-term plans proposed by the research team are highly likely to be discussed with the pharmaceutical industry as a promotion task. However, predictions are that it will be difficult to reflect the low purchasing price paid by national and public hospitals in the drug price cuts due to strong opposition from the industry. Therefore, realistic measures such as the abolition of the 10% upper limit in the price ceiling system are expected to be discussed. A HIRA official said, “The actual drug transaction price reduction system has been in effect for a long time, and there had been criticism on its low effect, therefore, we plan to come up with an improvement plan. If discussions progress well, measures may even be derived within the year.” The reductions to the actual drug transaction price had been carried out every year in line with the separation of prescribing and dispensing system in 2000 to reflect the actual transaction price of drugs in the drug price, and then has been conducted every two years since 2016. However, the prevailing opinion was that an improvement was needed as its drug cost reduction effect was poor and it was difficult to identify the actual transaction price. This was why HIRA set out to derive an improvement plan through a research service last year.
- Policy
- 87.5% of anticancer/rare drugs, evaluated as drugs w/o PE
- by Lee, Tak-Sun Mar 29, 2023 06:01am
- Yoo Mi-yeong, head of the HIRAAs the PE drug standard expands, the HIRA is seeking a reasonable management plan. This year, we plan to improve and supplement the PE system by deriving detailed plans through research services. Yoo Mi-young, head of the Pharmaceutical Management Office at the Health Insurance Review and Assessment Service, explained this at a meeting with the Professional Reporters Association held on the 28th. "In the case of anti-cancer drugs and rare disease drugs that were listed as new drugs last year, 87.5% of the total were evaluated as drugs without PE," said Director Yoo. We plan to prepare management plans based on this.” The research service is planned to start in April and end around the end of the year, and it is a policy to come up with a reasonable plan to improve health insurance financial sustainability and patient accessibility. The system that allows PE data submission to be omitted began in May 2015 with drugs for treating rare diseases for which there is no alternative or that threatens survival, and anticancer drugs, and in October 2020, it was expanded to drugs for tuberculosis, antibacterial drugs, and emergency antidotes among essential national medicines. From January of this year, it has been applied to drugs that have been proven to improve the quality of life of pediatric patients with rare diseases. Accordingly, CRYSViTA, a pediatric rickets treatment, falls under this category, passed the HIRA review stage without PE, and is currently negotiating with the NHIS. This PE omission system is being used to advance the drug evaluation period in order to increase patient access to treatment. Starting in the second half of this year, full-scale pilot projects linking permission-evaluation-negotiation For the same purpose, the MFDS, the HIRA, and the NHIS are starting this year to shorten the registration period for treatments for severe diseases such as cancer and rare diseases. are promoting Director Yoo explained, "We are currently in discussions with the MFDS related departments on target selection and related procedures to promote the pilot project." However, since the three institutions have different evaluation methods and conditions, and the beneficiary pharmaceutical companies also have burdens in preparing data at the same time, it is expected that only a few pharmaceutical companies will actually benefit from the system. Participation in the NHIS Pharmaceutical Evaluation Committee, there are concerns that fairness and objectivity issues may be raised. On the 7th, Lee Sang-il, executive director of the NHIS, said at a Korea Special Press Association meeting, “Secure the benefit adequacy and consistency of financial impact when listing a new drug, and evaluate the uncertainty of risk-sharing new drugs through organic linkage of negotiations. In order to support the decision, participation in the Pharmaceutical Reimbursement Evaluation Committee of the NHIS is necessary, so we plan to review a plan for NHIS members to participate in the Pharmaceutical Reimbursement Evaluation Committee through consultation with the Ministry of Health and Welfare and related organizations such as the HIRA." Director Yoo said, "Currently, the NHIS attends and monitors every meeting, and regularly holds meetings with the Ministry of Health and Welfare and the NHIS to discuss the contents of the committee's deliberations and issues for each individual agenda, and shares related data from time to time." Negative views were made on the need for NHIS to participate in the committee.