- LOGIN
- MemberShip
- 2026-04-25 13:34:04
- "Pharma consignment business faces difficult times"
- by Chon, Seung-Hyun | translator | 2026-01-21 09:07:40
The survival of consignment production businesses for pharmaceutical companies is threatened. As joint development regulations and the tiered drug pricing system block the entry of late-comer generics, contract manufacturers are struggling to maintain their operations. Concerns are growing that the market will freeze further as the new drug price reform lowers the criteria for generic price calculation and strengthens the tiered pricing system. An increasing sense of poor treatment is being expressed as the government expands support for the biopharmaceutical Contract Development and Manufacturing Organization (CDMO) sector while neglecting synthetic drug consignment.

According to industry sources on the 19th, Korea's domestic pharmaceutical companies are facing concern that the upcoming drug price reform could determine the survival of their consignment businesses.
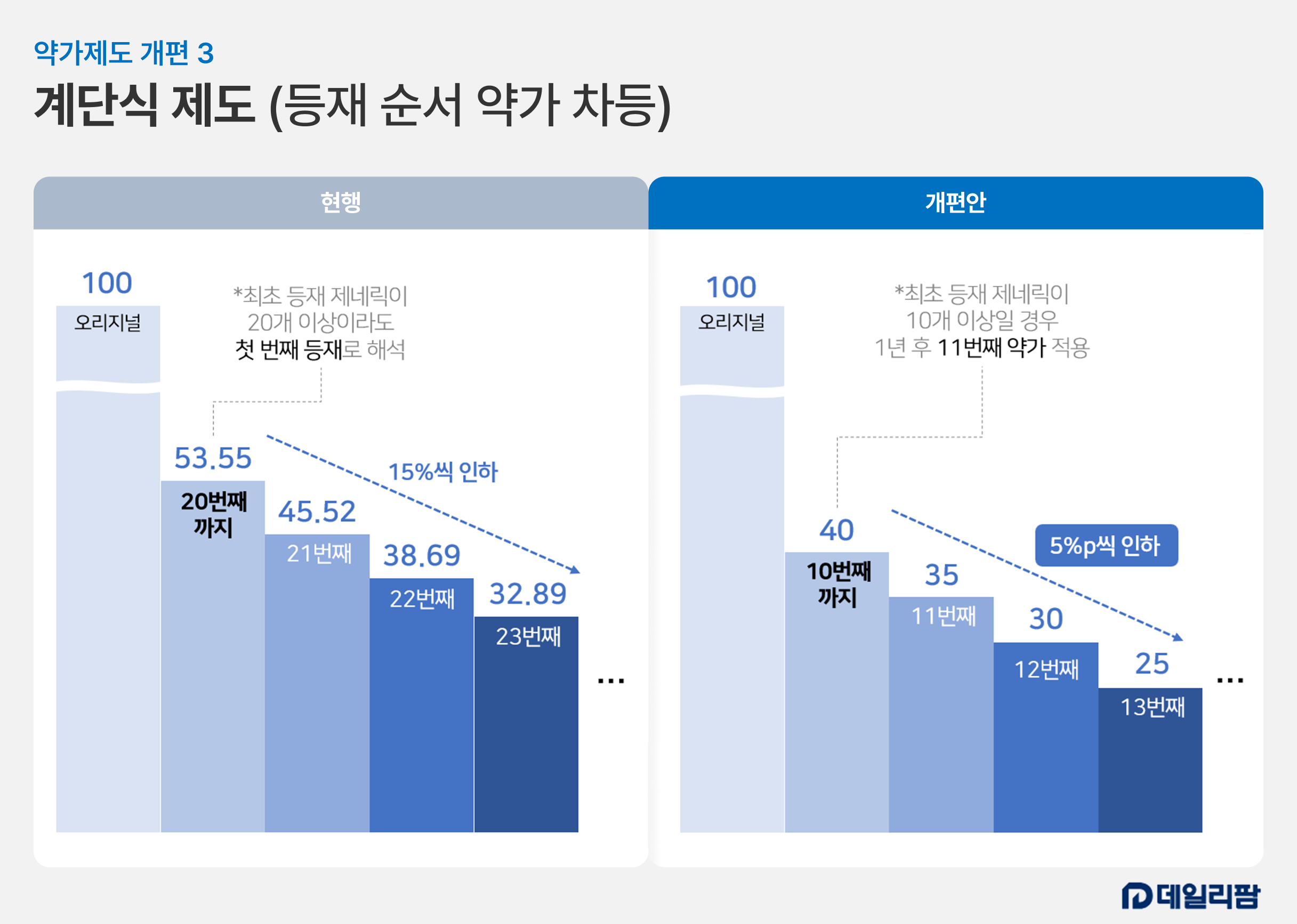
In the reformed system scheduled for implementation this July, the price calculation criteria for generics will drop from 53.55% of the original drug's price before patent expiration to about 40%. The final figure is expected to fall between 40% and 45%. If the maximum generic price drops to 40%, a 25% decrease in profit is expected.
A decrease in the generic price criteria inevitably leads to a downturn in the consignment business. Contract manufacturers typically set their supply prices at a certain percentage of the product's insured drug price.
For example, if a product is priced at KRW 1,000, the supply price is set around KRW 300–500. Consequently, lower generic insurance prices result in lower supply prices for contract manufacturers. The more unfavorable the cost structure, the higher the supply price becomes.
If the insurance price of a generic supplied by a contract manufacturer drops, the consigning company is forced to demand a lower supply price due to its own falling profitability. However, pharmaceutical companies argue they have limited capacity for such cuts due to continuous prior price reductions.
Recently, as regulations on authorization and pricing have tightened, more companies are reportedly considering scaling back or abolishing their consignment operations.
The tiered drug pricing system, implemented during the 2020 reform, has blocked latecomer generics.
Under the tiered drug pricing system, the upper price limit decreases each month as a generic enters the market later. Reintroduced in 2020 after being abolished in 2012, it mandates a 15% price reduction for any generic entering a market with over 20 identical listed products. This makes it difficult for latecomers to enter the market at such low prices. Contract manufacturers, in turn, struggle to expand as they cannot recruit additional consigning companies for their current products.
Before the tiered drug pricing system was implemented, the first 20 companies to enter a market would quickly reach their full quota, leaving latecomers without any incentive to join.
Additionally, joint development regulations acted as a catalyst for the scale down. Since the revised Pharmaceutical Affairs Act took effect in July 2021, the so-called '1+3' regulation has limited the number of incrementally modified drugs (IMDs) and generics that can be authorized using a single clinical trial.
If a product is manufactured at the same site using the same formula and process as the drug used in the bioequivalence study, the bioequivalence data may be used only 3 times. This means only four generics (one original and three others) can be authorized per study. The same '1+3' rule applies to clinical trial data.
These regulations also apply to generics already authorized and on sale. After the regulation took effect, only up to three additional consigning products can be added. For instance, a contract manufacturer that previously produced 10 generics can add only 3 more, for a total of 13. Since contract manufacturers can only recruit new clients if an existing one leaves, expanding business regardless of production capacity has become nearly impossible.
A consignment manager at a pharmaceutical company commented, "In the past, the government actively encouraged consignment, expecting that specialized mass production would improve quality control," adding, "If they treat the consignment business as the cause of the proliferation of generics and pursue only restrictive policies, we will have no choice but to consider job cuts due to business scaling down or withdrawal."
The strengthening of the tiered drug pricing system in the upcoming reform is also cited as a factor that will aggravate these difficulties.
The Ministry of Health and Welfare (MOHW) has proposed a plan to reduce the price by 5 percent (p) for each subsequent item listed, starting with the 11th product of the same formulation. Because the stepped system will trigger at the 11th product instead of the 21st, the system for additional price cuts will activate much faster across the generic market.
Analysis suggests that changing the reduction standard from a flat 15% to a 5%p decrease will disadvantage latecomers in terms of drug pricing cuts.
For example, under the current system, if the maximum generic price is KRW 53.55, the 21st generic cannot exceed KRW 45.52 (a 15% drop). The 22nd and 23rd generics fall to KRW 38.69 and KRW 32.89, respectively. The 24th is KRW 27.95, and the 25th is KRW 23.76. The reduction amount decreases as more products enter.
Under the reformed system, if the maximum price is 40 KRW, the 11th and 12th generics would drop to KRW 35 and KRW 30 due to the 5%p reduction (representing cuts of 12.5% and 14.3%, respectively). The 13th generic would drop another 5%p to KRW 25, a 16.7% cut. Even at the third step, the rate of reduction is already higher than under the current system.
As the 14th and 15th generics drop to KRW 20 and KRW 15, the reduction rates accelerate exponentially to 20% and 25%. Even after only five steps, the price cap falls to about 15% of the original drug's pre-patent price, effectively abolishing the incentive for any further entries and blocking the expansion of consignment businesses.
The government's recent active support for the biopharmaceutical CDMO industry further increases the sense of poor treatment among those in the synthetic drug consignment.
On December 30 last year, the "Special Act on Regulatory Support for Biopharmaceutical Contract Development and Manufacturing Organizations" was announced. The core of this law includes establishing a registration system for biopharmaceutical export manufacturing, which was not defined in the Pharmaceutical Affairs Act, setting facility standards for export-specialized sites, and institutionalizing GMP certification and raw material certification for CDMO sites.
The Ministry of Food and Drug Safety (MFDS) plans to prepare detailed criteria for customized regulatory support, including simplifying import procedures for raw materials used by CDMOs, providing prior GMP consultations, and offering technical advice on manufacturing facilities.
An industry official stated, "Previously, there was a strong perception that the consignment business contributed to improving production capacity and creating new revenue streams, but business scaling down has become inevitable due to continuous regulations and price cuts," adding, "It is currently impossible to predict future profit changes, making even this year's business plans unclear."
-

- 1
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.





